Metabolic
advertisement

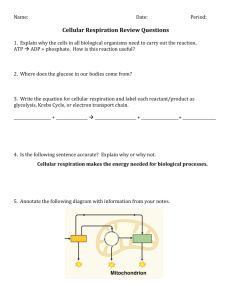
Lecture 2 Outline (Ch. 8, 9) I. Energy and Metabolism II. Thermodynamics III. Metabolism and Chemical Reactions V. Cellular Energy - ATP VI. Enzymes & Regulation VII. Cell Respiration A. Redox Reactions B. Glycolysis C. Coenzyme Junction VII. Preparation for next Lecture Energy What is Energy? Where does energy on earth come from originally? [equivalent of 40 million billion calories per second!] Metabolism and Energy Metabolism Metabolism –chemical conversions in an organism Types of Energy: - Kinetic Energy = energy of movement - thermal - Potential = stored energy - chemical Thermodynamics Thermodynamics – study of energy transformation in a system Potential energy can be converted to kinetic energy (& vice versa) Potential Energy Kinetic Energy Thermodynamics Laws of Thermodynamics: Explain the characteristics of energy 1st Law: • Energy is conserved • Energy is not created or destroyed • Energy can be converted (Chemical Heat) 2nd Law: • During conversions, amount of useful energy decreases • No process is 100% efficient • Entropy (measure of disorder) is increased Energy is converted from more ordered to less ordered forms Potential vs. Kinetic Energy Metabolism Metabolic reactions: All chemical reactions in organism Two Types of Metabolic Reactions: Catabolic = breaks down molecules Anabolic = builds up molecules Chemical Reactions Chemical Reaction: • Process that makes and breaks chemical bonds + + Reactants Products Two Types of Chemical Reactions: 1) Exergonic = releases energy 2) Endergonic = requires energy Metabolism Metabolic reactions: • Chemical reactions in organism Two Types of Metabolic Reactions: Catabolic = break down Exergonic = release energy Anabolic = build up Endergonic = requires energy Chemical Reactions Glucose CO2 + H20 CO2 + H20 Glucose -ΔG +ΔG (or 0) release energy intake energy spontaneous non-spontaneous • Exergonic reaction • Endergonic reaction Question/Recall: Which has more order? Stores more energy? Polymer or Monomer, Diffused or Concentrated H+? What is relationship between order and energy? What type of energy is stored in a covalent bond? A. B. C. D. E. Kinetic energy Diffused energy Heat energy Potential energy Conventional energy Cellular Energy - ATP • ATP = adenosine triphosphate • ribose, adenine, 3 phosphates • last (terminal) phosphate - removable Be able to diagram ATP! Cellular Energy - ATP • ATP hydrolyzed to ADP • Exergonic ATP + H2O ADP + Pi • Energy released, used in another reactions (endergonic) Cellular Energy - ATP • ATP regenerated • cells power ATP generation by coupling to exergonic reactions Like cellular respiration! ATP Cycle Making ATP from ADP + Pi is… A. B. C. D. Exergonic because it releases energy Endergonic because it requires energy Exergonic because it requires energy Endergonic because it releases energy Chemical Reactions Chemical Reactions: • Like home offices – tend toward disorder Chemical Reactions Chemical Reactions: • Endergonic – energy taken in • Exergonic – energy given off Exergonic Endergonic Self-Check Reaction Breaking down starches to sugars Building proteins Digesting Fats Exergonic or Endergonic? Chemical Reactions Activation Energy: Energy required to “jumpstart” a chemical reaction • Must overcome repulsion of molecules due to negative charged electrons Nucleus Repel Nucleus Activation Energy Nucleus Repel Activation Energy Nucleus Chemical Reactions Exergonic Reaction: – Reactants have more energy than products Activation energy: Make sugar and O2 molecules collide sugar + O2 water + CO2 “Downhill” reaction Respiration (ch. 9) preview Cellular Respiration Equation: C6H12O6 + O2 CO2 + H2O You will need to KNOW this equation. Chemical Reactions and Enzymes Enzymes • lower activation energy only for specific reactions • cell chooses which reactions proceed! enzymes: cannot make rxns go that wouldn’t otherwise Cannot change endergonic into exergonic rxns Do speed up rxns that would occur anyway Enzymes • Enzymes – control rate of chemical reaction • sucrase – enzyme sucrose breakdown • sucrase – catalyst “-ase” enzyme -speed up rxn, but not consumed Enzymes Enzymes • enzyme – specific to substrate • active site – part of enzyme -substrate • binding tightens fit – induced fit • form enzyme-substrate complex • catalytic part of enzyme: converts reactant(s) to product(s) Enzymes • Enzymes lowers EA by: -template orientation • substrate(s) enter -stress bonds -microenvironment • enzyme reused • product(s) formed • What factors might affect enzyme activity? Enzymes • inhibitors: binds & blocks active site • Drug – blocks HIV enzyme at the active site binds allosteric site – alters conformation If a competitive inhibitor is in an enzyme reaction, can you reverse the inhibition by adding more substrate? A. B. C. D. Yes No I’m not sure Wait, what’s a competitive inhibitor? Cellular Respiration Overall purpose: • convert food to energy • animals AND plants • complementary to photosynthesis Cellular Respiration Cellular Respiration: (Exergonic) • catabolizes sugars to CO2 • requires O2 • at mitochondrion Redox Reactions • as part of chemical reaction, e- are transferred • e- transfer = basis of REDOX reactions (reduction) (oxidation) Redox Reactions Use “H rule” for reactions in this class Reactant with more H’s = e donor, will be oxidized Reactant with more O’s = e acceptor, will be reduced ZH2 + O2 yields ZO + H2O • follow the H, e- move with them Self-Check Reaction ZH2 + O2 yields ZO + H2O CH4 + 2O2 yields CO2 + 2H2O C6H12O6 + O2 yields CO2 + H2O Molecule Reduced Oxygen Molecule Oxidized ZH2 Redox Reactions Equation for respiration Redox Reactions • transfer of e- to oxygen is stepwise Redox Reactions • e- moved by NAD/H (from niacin/vit B3) • NADH carry e- (reduced!) • NAD+ not carrying e- (oxidized!) Where do e- come from? Where do e- go? • glucose NADH ETC O2 (makes H2O) In this equation is NAD+ to NADH oxidized or reduced? NAD+ + H+ + 2e- NADH A. B. C. D. Reduced, it gained electrons Oxidized, it gained electrons Reduced, it lost electrons Oxidized, it lost electrons Steps of Respiration • Steps of respiration: 1. glycolysis - cytosol Coenzyme Junction 2 CO2 2. Citric acid cycle - mitochondrial matrix 3. ETC - inner mitochondrial membrane 4. Chemiosmosis - inner membrane to intermembrane space 4 CO2 Cellular Respiration • Stages of respiration: 1. Glycolysis – prep carbons Cellular Respiration 1. Glycolysis • 1 glucose (6C) 2 pyruvate (3C) • Keep track of: - inputs - ATP - NAD+/NADH - CO2 and H2O - outputs • eukaryotes AND prokaryotes Glycolysis Glucose Glucose-6-phosphate ATP 1 2 ADP Glucose-6-phosphate Fructose-6-phosphate Glycolysis ATP ADP Fructose1, 6-bisphosphate 4 5 Dihydroxyacetone phosphate Glyceraldehyde3-phosphate Glycolysis 2 ADP 2 ATP 2 Phosphoenolpyruvate 2 ADP 10 2 ATP 2 Pyruvate How many NET ATP are produced by glycolysis? A. B. C. D. E. one two four six eight Glycolysis Cellular Respiration -inputs: 1 Glucose 2 ATP -outputs: 2 pyruvate 4 ATP (2 net) 2 NADH CO2 = none yet (2 H2O) Where do the outputs go? Energy production Mitochondria • energy from nutrients ATP Cellular Respiration Coenzyme Junction • 2 pyruvate (3C) 2 Acetyl CoA (2C) • pyruvate joins coenzyme A (from vitamin B5) • 2 carbons lost (as CO2) • 2 NAD+ NADH Things To Do After Lecture 2… Reading and Preparation: 1. Re-read today’s lecture, highlight all vocabulary you do not understand, and look up terms. 2. Ch. 8 Self-Quiz: #1-6 (correct answers in back of book) 3. Read chapter 9, focus on material covered in lecture (terms, concepts, and figures!) 4. Skim next lecture. “HOMEWORK” (NOT COLLECTED – but things to think about for studying): 1. Describe the relationship between exergonic/endergonic, catabolic/anabolic, and “uphill”/”downhill” chemical reactions 2. Diagram one molecule of ATP and how ADP is different 3. Cut apart the boxes on the previous sheet – match up three (name, energy balance, basic reaction) for glycolysis and three for the coenzyme junction 4. Place the following molecules in order for when they are used/created during glycolysis: fructose-6-phosphate, glucose, glucose-6-phosphate, pyruvate, glyceraldehyde-3-phosphate Self-check at home