020507

Chapter 1
• Foundations of biochemistry
– Biological
– Chemical
– Physical
– Genetic
– Evolutionary
• Focus on chemical and physical
– Need to know biological!
Hierarchy of biology
Biosphere
Ecosystem
Organism
Organ systems
Organs
Tissues
Cells
Organelles
Molecules
Physical and chemical properties that define their function
Cells are transducers of energy
• First law of thermodynamics
SOLAR
CHEMICAL (C-C Bonds)
CHEMICAL (ATP)
Cells are transducers of energy
• Synthesis of macromolecules
• Mechanical work
• Osmotic/electrical/chemical gradients
• Light production
• Genetic information: transfer/maintenance
• Heat
Second law of thermodynamics
• Disorder always tends to increase
– ENTROPY
• Life requires order
• Creating/maintaining order requires work, therefore energy
Gibbs free energy
(G)
• Amount of energy in a system available to do work
• “Force” that drives chemical reactions
• A system always strives to achieve minimum free energy
System strives to reach lowest energy
G = H - TS
• T = temperature
– In vivo ~constant
– Becomes important in vitro
• H = enthalpy
– Heat/thermal energy
– Chemical bonds
• Break bonds: add heat
• Form bonds: release heat
• S = entropy
– Disorder
– Randomness
– Freedom
G = H - TS
– More degrees of freedom: higher entropy
Which has more entropy?
vs.
Which has higher entropy?
Liquid water Solid water vs.
G = H - TS
• Chemical reaction: Reactants and products have different Gs
• Change of free energy: D
G
D
G =
D
H – T D
S
• Negative D
G: exergonic
• Positive D
G: endergonic
A + B ↔ C + D
Lower free energy is ‘better’
D
G < 0 (-
D
G)
-reaction is spontaneous (no energy input necessary)
-proceeds from L to R
D
G > 0 (+
D
G)
reaction isn’t spontaneous
-proceeds from R to L
D
G = 0
-both reactions are spontaneous
-reaction at equilibrium
Two ways to “manipulate” D
G
• Energy payoff: D
H vs.
D
S
• Cooperation: couple unfavorable and favorable reactions
The energy ‘payoff’
• Decreased enthalpy ( D
H < 0) favored
• Increased entropy ( D
S > 0) favored
• But, overall D
G has to be negative
– If D H is positive, reaction can be ‘driven’ by entropy
Energetic coupling
Unfavorable reactions can be coupled to favorable reactions to make an overall favorable reaction
Glucose + phosphate → glucose-6-phosphate
D
G > 0
ATP → ADP + phosphate D
G << 0
Glucose + ATP → glucose-6-phosphate + ADP
D
G < 0
Chemical equilibrium
3. Products become reactants exactly as fast as reactants become products
Equilibrium – “Steady State” G < 0)
At steady state:
D
G = 0
Equilibrium is related to
D
G °
a A + b B ⇌ c C + d D
High K eq
, neg
D G: reaction ‘favors’ products
Low K eq
(<1): reaction ‘favors’ reactants
Biological reaction: sugar + oxygen ↔ CO
2
+ water
High energy “Transition state”
Intermediate between R & P
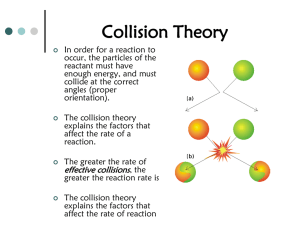
Activation energy
Reactants (R)
E
A
Products (P)
D
G < 0 reaction
Reaction should be spontaneous
REACTION PROGRESS
Equil should favor products