Multiple Centers, Exceptions, and Polarity
advertisement

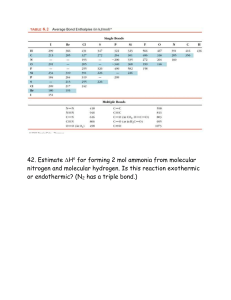
Multiple Bonds, Multiple Centers, Exceptions, and Polarity Objective Today I will be able to: Summarize the exceptions and inner workings of molecular geometries Draw Lewis Structures to represent the valance electrons of atoms Predict the molecular shape of a molecule using the VSEPR theory Evaluation/ Assessment Informal assessment – Listening to group interactions as they complete the illustrating ionic and covalent compounds practice and the molecular shapes lab. Formal Assessment – Analyzing student responses to the exit ticket and the practice worksheets. Common Core Connection Make sense of problem and persevere in solving them Reason abstractly and quantitatively Use appropriate tools strategically Look for and make use of structure Warm – Up Explain: Exceptions and Polarity notes Explore and elaborate: Molecular Shapes Lab ◦ Informal Assessment Evaluate: Exit Ticket • Formal assessment Today I will be able to: ◦ Summarize the exceptions and inner workings of molecular geometries ◦ Draw Lewis Structures to represent the valance electrons of atoms ◦ Predict the molecular shape of a molecule using the VSEPR theory STEM Fair ◦ In Class Presentations Wednesday January 22 Warm-Up Exceptions & Polarity Notes Molecular Shapes Lab Exit Ticket Which shape has the largest bond angle? How many lone pairs and bonds formed are in the trigonal pyramidal shape? Draw the molecular shape for phosphorus pentachloride. Sample Problem 1 PROBLEM: Writing Lewis Structures for Molecules with One Central Atom Write a Lewis structure for CCl2F2 Sample Problem 2 PROBLEM: Writing Lewis Structures for Molecules with More than One Central Atom Write the Lewis structure for methanol (molecular formula CH4O), an important industrial alcohol that is being used as a gasoline alternative in car engines. Multiple Bonds If there are not enough electrons for the central atom to attain an octet, a multiple bond is present. Step 5: If the central atom does not have a full octet, change a lone pair on a surrounding atom into another bonding pair to the central atom, thus forming a multiple bond. Sample Problem 3 PROBLEM: Writing Lewis Structures for Molecules with Multiple Bonds Write Lewis structures for the following: (a) Ethylene (C2H4), the most important reactant in the manufacture of polymers (b) Nitrogen (N2), the most abundant atmospheric gas Exceptions to the Octet Rule Molecules with Electron-Deficient Atoms B and Be are commonly electrondeficient. Odd-Electron Species A molecule with an odd number of electrons is called a free radical. Exceptions to the Octet Rule Expanded Valence Shells An expanded valence shell is only possible for nonmetals from Period 3 or higher because these elements have available d orbitals. Sample Problem 4 PROBLEM: Writing Lewis Structures for Octet-Rule Exceptions Write a Lewis structure and identify the octet-rule exception for (a) SClF5; (b) BFCl2. How to draw and decipher Figure 10.12 The tetrahedral shapes around the central atoms and the overall shapes of ethane (A) and ethanol (B).Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. ethane ethanol CH3CH3 CH3CH2OH Sample Problem 5 PROBLEM: Predicting Molecular Shapes with More Than One Central Atom Determine the shape around each of the central atoms in acetone, (CH3)2CO. Molecular Shape and Molecular Polarity Overall molecular polarity depends on both shape and bond polarity. The polarity of a molecule is measured by its dipole moment (μ), which is given in the unit debye (D). A molecule is polar if - it contains one or more polar bonds and - the individual bond dipoles do not cancel. The orientation of polar molecules in an electric field. Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Molecules are randomly oriented. Molecules become oriented when the field is turned on. Bond Polarity, Bond Angle, and Dipole Moment Example: CO2 The DEN between C (EN = 2.5) and O (EN = 3.5) makes each C=O bond polar. Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. CO2 is linear, the bond angle is 180°, and the individual bond polarities therefore cancel. The molecule has no net dipole moment (μ = 0 D). Bond Polarity, Bond Angle, and Dipole Moment Example: H2O The DEN between H (EN = 2.1) and O (EN = 3.5) makes each H-O bond polar. Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. H2O has a V shaped geometry and the individual bond polarities do not cancel. This molecule has an overall molecular polarity. The O is partially negative while the H atoms are partially positive. Bond Polarity, Bond Angle, and Dipole Moment Molecules with the same shape may have different polarities. CCl4 Bonds are polar, but individual bond polarities cancel. CHCl3 Bond polarities do not cancel. This molecule is polar (μ = 1.01 D). Sample Problem 7 Predicting the Polarity of Molecules PROBLEM: For each of the following use the molecular shape and EN values and trends to predict the direction of bond and molecular polarity, if present. (a) Ammonia, NH3 (b) Boron trifluoride, BF3 (c) Carbonyl sulfide, COS (atom sequence SCO) Finish up and complete the polarity section now! Determine the shape CH3Cl CH2O BeCl2 BCl3 PF5 SF6 XeF4 SF4 ICl3 O2K+ ClO2-1 CN-1 CO32SO42- Draw the Lewis Structure for CO2 Determine the shape and bond angles of CO2 according to the VSEPR theory.