超臨界流體萃取及層析
advertisement

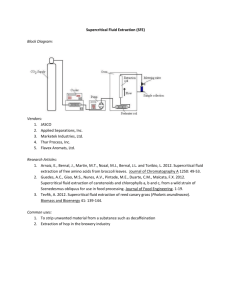
超臨界流體 萃取,層析及其他應用 Supercritical Fluid Extraction, Chromatography and Other Applications 林華經 引光生物科技有限公司 What is a Supercritical Fluid ? When the pressure and temperature of a substance is raised above its critical pressure and critical temperature (the critical point) the substance enters the supercritical state. A Supercritical Fluid is a substance with both gas- and liquid-like properties. Critical Temperature and Pressure The Critical Temperature (Tc) is the maximum temperature at which a gas can be converted to a liquid by increasing the pressure. The Critical Pressure (Pc) is the maximum pressure at which a liquid can be converted to a gas by increasing the temperature. Pressure Phase Diagram of Carbon Dioxide Solid Liquid Gas Triple Point Temperature Supercritical Fluid Critical Point 31.3 oC, 1072 psi Physical Properties of CO2 Phase Gas SCF Liquid Density 0.6–2.0 x 10-3 0.2 – 0.9 0.8 – 1.0 viscosity 0.5–3.5 x 10-4 2.0–9.9 x 10-4 0.3–2.4 x 10-2 Diffusivity 0.01 – 1.0 0.5–3.3 x 10-4 0.5–2.0 x 10-5 Advantages of Supercritical Fluids as solvents Solvating power related to density (at constant T) Gas-like mass transport properties Facile penetration into porous material Critical Parameters of Common Fluids Tc (°C) c (g mL ) -1 Pc (atm) CO2 31.3 72.9 0.47 N2 O 36.5 72.5 0.45 SF2 45.5 37.1 0.74 NH3 132.5 112.5 0.24 H2 O 374 227 0.34 n-C 4H10 152 37.5 0.23 n-C 6H12 197 33.3 0.23 Xe 16.6 58.4 1.10 CCl 2F2 112 40.7 0.56 CHF 3 25.9 46.9 0.52 Source: “SFE and Its use in Chromatographic Sample Preparation” Ed. S. Westwood. Chapter 1 Advantages of CO2 for SFE Low critical parameters Inert, Non-toxic, Nonflammable, Noncorrosive Easily purified (inexpensive) Nonpolar: dielectric constant similar to hexane Modifiers can be used to increase polarity Supercritical Fluid Extraction (SFE) Basic Theory SFE System Components CO2 Pump (high pressure) Modifier Pump sample cell in heated chamber CO2 Liquid Carbon Dioxide (requires a dip tube) Restrictor CO2 Trap Solid or Liquid Advantages of SFE over Solvent Extraction Faster Selectivity results in cleaner extracts Low Critical Parameters Results in minutes rather than hours Handling of thermally labile analytes Non-hazardous solvents Automated Cost per test is lower Limited or no solvent removal required No solvent disposal costs The Mechanism of SFE A Three Step Process 1. Dissolving/Resolving analyte(s) 2. Sweeping the analyte(s) from the matrix 3. Trapping the analyte(s) SFE Mechanism High Pressure Liquid CO2 SC-CO2 diffuses matrix, dissolves and resolves analyte from the matrix Heat SC-CO2 + dissolved analyte to the trap Trapping Gaseous CO2 CO2 is changing from a SF (2 ml/min) to an expanded gas (1 L/min) •Analyte no longer soluble •Mechanical movement of analyte due to the rapid expansion requires the use of trapping material Gaseous CO2 Trapping Solid •High Surface Area •Adequate Amount Trapping Liquid •High Surface Tension •Analyte is Soluble •Low Volatility •Pressurized •Cryogenically Cooled SFE - Method Development Pressure Temperature Flow Rate Fluid Composition Increase temperature may decreases density = decrease in solubilizing power (ie CO2 at 100 bar) Extraction Time Increase pressure increases density = increase in solubilizing power. Co-solvents/modifiers Reactant Additive Static vs. Dynamic Extraction Why Use Modifiers? Analytes that have polar components require the use of a cosolvent Triglycerides CH2OCOR1 R2COOCH CH2OCOR3 Phospholipids CH2OCOR1 R2COOCH O CH2OPOR3 OR 1, 2 & 3 groups are long chain hydrocarbons (nonpolar) R 1 & 2 groups are long chain hydrocarbons (nonpolar), while R 3 contains phosphorus and nitrogen and is polar Modifiers (Co-solvents) in SFE Role of Modifiers in SFE Changes in Solvent Polarity Interaction with Matrix Interaction with Analyte Methods of Addition Directly into extraction cell (spiking) On line modifier addition (uses a second pump) SFE vs. Traditional Sample Extraction Methods Conclusion SFE can be versatile, selective and faster SFE reduces hazardous solvent use and cost SFE can produce cleaner, more concentrated extracts for post extraction analysis Analyst’s Sample Process. 61% Time Allotment Collection 6% Analysis 6% Data Management 27% Routine and Novel Applications of Analytical SFE SFE for Research Environmental SFE Applications • Matrices Soil Tissue Clay Sandy Loam Sludge River Sediment Marine Sediment Fly Ash Incinerator Ash • Target Analytes TPH PAH PCBs Pesticides Dibenzofurans Dioxins Approved Methods Environmental • US EPA 3560 - TPH in Soil: Supercritical Fluid Extraction of Total Recoverable Petroleum Hydrocarbons • US EPA 3561 - PAH in Soil: Supercritical Fluid Extraction of Polyaromatic Hydrocarbons • US EPA 3562 - PCB and OCP: Supercritical Fluid Extraction of Polychlorinated Biphenyls (PCBs) and Organochlorine • US EPA 3545: Pressurized Fluid Extraction (PFE) • USDOE STD-3013-99: Determination of Residual Water in Impure Plutonium Oxides • AOAC draft: SFE-GC/MS determination of pesticide residues in non-fatty fruits and vegetables Pharmaceutical/Natural Product Applications SFE of Natural Products -- Roger M. Smith LC-GC International, Jan. 1996, 9-15 Catharanthus roseus German chamomile Magnolia grandiflora Tansy Chamomile flowerheads Ginger Peppermint Thyme Clove Oil Kola nuts Pimento Turmeric Dragon head Lavender Poppy seeds Wheat germ oil English yew Lemon grass Rosemary Feverfew Lemon peel Savory Microbial Natural Products R. M. Smith, op. cit. Organism Extract Agaricus species Carboxylic and fatty acids Beuveria nivea Cyclosporin Filamentous fungi Polyunsaturated fatty acids Flour, moldy bread, mushrooms Ergosterol Moldy bran Sterol Moldy grain Aflatoxin Extracts from Biomass R. M. Smith, op. cit. Microorganism Extract Actinomycete species Mycolutein and luteoreticulin Actinomycete species Oligomycin A Aspergillus fumigatus Sydowinin B and epoxide Bipolaris urochloae Ophiobolin A Penicillium expansum Chaetoglobosin A Penicillium sclerotium (+)-Sclerotiorin Streptomyces species Elaiophylin Summary R. M. Smith, op. cit. • Extracts typically cleaner than those obtained with organic solvents. • Mild conditions minimize degradation. • SFE methods are faster than organic solvent extractions. Extraction of Pharmaceuticals Using Pressurized Carbon Dioxide J. R. Dean, S. Khundker, J. Pharm. & Biomed. Anal, 15 (1997) 875-886 • Recoveries from 81% - 95+% • CO2 and CO2 with modifiers • Generally faster than other methods with better selectivity for target analytes. • Preconcentration steps could be eliminated in some cases. • Liquid matrices required immobilization on solid support or SPE cartridge. Animal Feeds J. R. Dean, S. Khundker, op. cit. Analyte Matrix Menadione Rat chow Tipradane Rodent diet Hypolipidermic drug Rat feed Halogenated aromatic phenoxy derivatives Dog feed/rodent feed Atovaquone Rat feed Fluconazole Animal feed Propanolol, Tamoxifen, ZM 95527, 169369 Rodent diet Formulations J. R. Dean, S. Khundker, op. cit. Analyte Matrix Megesterol Acetate Tablet Felodipine Tablet Benzodiazipines (7) Tablet/capsule Caffeine,vanillin Tablet Vitamin A, E Tablet Retinol palmitate, tocopherol acetate Ointment Polymyxin B sulphate Cream/Ointment Acylvoir Ointment Sulfamethazole, trimethoprim Septra infusion Triamincinolone Dermatological patches Misoprostol Hydroxypropyl methylcellulose Biological Matrices J. R. Dean, S. Khundker, op. cit. Analyte Matrix Veterinary drugs (4) Pig kidney Nitrobenzamide residue Liver Codeine, morphine, ethyl morphine Hair Ketorolac, flavone Plasma Mebervine alcohol Dog plasma Morphine Serum Beudesonide Plasma Caffeine Kola nuts Taxanes Yew tree needles Chinese herbal medicines Plants Diosgenin Tubers of Dioscorea nipponica Taxol and baccatin III Needles of Taxus cuspidata Zingiber zerumet rhizomes Plants Mevinolin and hydroxy acid form Fermentation broth Phylloquinone Soy protein and infant formula Miscellaneous J. R. Dean, S. Khundker, op. cit. Analyte Matrix Triprolidine, pseudoephedrine Aqueous Steroids (10) Aqueous Ibuprofen Aqueous Natural Materials Studied M. J. Noh, et. al., op. cit. Specific Name Part Used Lycium chinese Fruit Schizandre chinensis Fruit Citrus unshiu Fruit bark Angelica gigas Root Cornus officinalis Fruit Cnidium officinale Rhizome Ginko biloba Leaf Aralia cordata Root Evodia officinalis Fruit Crataegus pinnatifida Fruit Paeonia lactiflora Root Leonurus sibricus All Sophora japonica Flower Artemisia capillaris All Platago asiatica Seed Natural Materials Studied, contd. M. J. Noh, et. al., op. cit. Specific Name Part Used Ephedra sinica All Aconitum carmichaeli Tuber Scolopendra subspines All Paeonia suffruticosa Root Pueraria thunbergiana Root Polygala tenuifolia Root Coptis japonica Rhizome Astragalus membranaceus Root Eucommia ulmoides Stem bark Bupeuri falcatum Root Acanthopanax sessiliflorum Bark Epimedium koreaum All Morus alba Root bark Artium lappa Fruit Spirodela polyrhiza All Summary M. J. Noh, et. al., op. cit • For many materials, SFE yielded extracts with higher bioactivity than LSE. • SFE was found to be more selective than LSE for target compounds. • SFE conditions could be optimized to produce maximum levels of bioactivity. Drug Residues Analyte Matrix Reference Sulfamethazine Swine Muscle Tissue Cross, et.al. Anabolic Steriods Bovine Tissue (Muscle Houpalahti and Henion and Liver) Opiates Hair, blood and tissue Multiple Authors Temazepam Whole Blood Scott and Oliver Cocaine, benzoylecgonine, codeine and morphine Hair Brewer, et.al. Study Summary • Compared to a conventional SPE method, the SFE method was more efficient and gave cleaner extracts with recoveries above 80% • K.S. Scott, J.S. Oliver, J. Anal.Toxicol. 21 (1997) 297. Supercritical Fluid Chromatography • SFC is a separation technique similar to HPLC and GC where the mobile phase or carrier gas is replaced by a supercritical fluid Limitations of GC and HPLC GC Sample Limitations : Volatility Thermal stability Low molecular weight HPLC Analytical Limitations : No universal detector Low efficiency Low resolution SFC Overcomes Limitations of GC and HPLC • • • • • • Extends molecular weight range of GC Lower operating temperature than GC Faster separation time than HPLC Higher separation efficiency than HPLC Universal detector can be used, FID Both packed (HPLC-type) and GC-type columns can be used Carbon Dioxide, CO2 has desirable properties as a SFC solvent • • • • • • • Inexpensive Highly pure Very low UV absorbance NO FID background noise Low critical pressure and temperature Non-toxic Supercritical CO2 behaves as a nonpolar solvent such as heptane • Polar organic modifiers can be mixed with CO2 for more polar samples SFC Applications Industrial : • • • • • • • Synthetic oligomers, polymers / additives Surfactants (polyglycols) Oligo / polysaccharides, sucrose polyesters Pesticides Isocyanates Dyes Waxes SFC Applications Biochemical : • • • • • Steroids Prostaglandins Fatty acids / lipids Antibiotics Drugs of abuse SFC Applications Fossil Fuels : • Fractionation of petroleum and coal-derived fluids • Hydrocarbon group analysis • Simulated distillation Other Applications of Supercritical Fluid • • • • • • Supercritical Fluid Cleaning Supercritical Fluid Drying Supercritical Fluid Reactions Micro Particles Formation Supercritical Water Oxidation System Others Applications of Supercritical Fluid Technologies in Taiwan • IN THE PAST Applications of Supercritical Fluid Technologies in Taiwan • AT PRESENT Applications of Supercritical Fluid Technologies in Taiwan • IN THE FUTURE Thanks For Your Attention ! • 林華經 • • • 國立清華大學化工系學士 國立清華大學生科所碩士 國立清華大學化工系博士班 • • 友翔實業股份有限公司 儀器部經理 引光生物科技有限公司 研發部經理