Acids and Bases (JLO)
advertisement

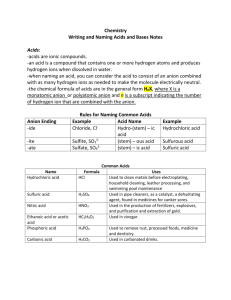
Acids and Bases Names and Formulas Binary Acids • Binary acids are made of two ions. The first ion (cation) is always H+. Change hydrogen to hydroReplace the ending, –ide, on the second ion (anion) with –ic. Naming Binary Acids Example: Formula: HCl H+ hydrogen = hydro- Cl- chloride = chloric hydro-chlor-ic hydrochloric acid Writing Binary Inorganic Acid Formulas • The first ion (cation) is always hydrogen, H+. • The second ion (anion) is usually from Group VII A (halogen). • Exception: Group VIA - S (sulfur). Binary Acid Formula Example: hydroiodic acid cation H+ Formula: HI anion I- Oxyacids Oxyacids contain hydrogen, oxygen and a third element (usually a nonmetal). The first ion (cation) is always H+ The second ion (anion) is a polyatomic ion. Naming Oxyacids I • If the anion name ends in –ate, the suffix –ic is added to the root name. Example: HNO3 H+ NO3NO3- , is the anion. nitrate, Replace –ate with –ic nitric Add “acid” = nitric acid HNO3 Naming Oxyacid Acids II • If the anion name ends in –ite, the suffix –ous is added to the root name. One less oxygen? HNO2 NO2- , is the anion. Replace –ite with –ous Add “acid” nitrite, = nitrous = nitrous acid HNO2 Try These 1. H2SO4 2. H2SO3 Example: H2SO4 • SO42- = sulfate ion • Replace –ate with –ic Add “acid” S = sulfur = sulfuric = sulfuric acid H2SO4 One less oxygen? H2SO3 SO32- = sulfite ion Replace –ite with –ous Add “acid” S = sulfur = sulfurous = sulfurous acid H2SO3 Writing Oxyacid Formulas • The first ion (cation) is always hydrogen, H+. • The second ion (anion) is a polyatomic ion. Writing an Oxyacid Formula Example: phosphoric acid cation anion H+ PO43Formula = H3PO4 Naming Inorganic Bases Don’t change the name of the lst ion. The last name of a base will always be hydroxide. Examples: NaOH (NH4)OH = sodium hydroxide = ammonium hydroxide Writing Formula for an Inorganic Base Example: zinc hydroxide cation anion Zn2+ OH- Formula: Zn(OH)2 Name the Acids Put in Classwork/Homework Notebook 1. HF 5. HClO3 2. HBr 6. HIO4 3. HC2H3O2 7. HIO3 4. H2S 8. HIO2 Write formulas for the acids Put in your Classwork/Homework Notebook. 1. 2. 3. 4. 5. 6. Hydrosulfuric acid Carbonic acid Hypochlorous acid Hypoiodous acidd Perchloric Chlorous acid Naming Bases Put in Classwork/Homework Notebook 1. 2. 3. 4. 5. LiOH KOH RbOH Ca(OH)2 Sr(OH)2 6. Ba(OH)2 7. Al(OH)3 8. Fe(OH)3 9. CsOH Write Formulas for Bases Put in your Classwork/Homework Notebook. 1. Chromium (III) hydroxide 2. Mercury(II) hydroxide 3. Nickel (II) hydroxide 4. Tin (II) hydroxide 5. Cobalt (III) hydroxide Answer Check Be prepared to share answers in class tomorrow.