mass
advertisement

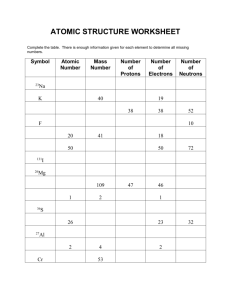
Mr. Shields Regents Chemistry U01 L05 1 Atomic Mass Up to now we discussed Atomic mass number in terms of The Number of Neutrons and Protons Ex. 8p + 9n = atomic mass number 17 But mass should tell us how much matter is present. What does “17” really tell us about how much matter (mass) is present? 2 Atomic Mass Remember - Neutrons and Protons DON’T really Have the same mass. Mass of a Proton = 1.6726 x 10-24 gram Mass of a Neutron = 1.6749 x 10-24 gram So a neutron is really a little heavier than a Proton. So what’s the implication? If I have 19 neutrons and 19 protons in a nucleus it DOES NOT have exactly the same mass as A nucleus with 21 neutrons and 17 protons even though The stated mass number is the same (38) 3 Atomic Mass Let’s look at the mass of one of the isotopes of Carbon. Carbon-12: 6 protons + 6 neutrons 6p x (1.6726 x 10-24) + 6 x (1.6749 x 10-24) grams 1 atom of carbon-12 = 2.00850 x 10-23 grams But this is an “awkward” number to work with 4 Atomic Mass Units Instead of actual weights in grams Scientist use a unit called an ATOMIC MASS UNIT - Abbreviated “AMU” Scientist decide NOT to base the AMU on either the proton or neutron Instead an arbitrary “Standard” was chosen 5 Atomic Mass Unit The “Standard” they chose was an isotope of Carbon - Carbon-12 (REMEMBER THIS!!) The mass of CARBON-12 was defined to be EXACTLY equal to 12 AMU’s In other words 1 AMU EQUALS 1/12 the mass of a Carbon-12 Atom 1 AMU is slightly less than the mass of either a neutron or a proton - 1 AMU = 1.661 x 10-24 g 6 AMU’s EXCEPT for Carbon-12 the MASS of an atom in AMU’s is NEVER EXACTLY the same as it’s MASS NUMBER For example here’s some examples of AMU’s vs Mass No. Atomic Mass in AMU’s Proton 1.007825 Carbon-12 12.00000 Oxygen-16 15.994915 Magnesium-25 24.985837 Nickel-60 59.930791 Uranium-235 235.043925 Mass # 1 12 16 25 60 235 7 8 Isotopic Abundance/Composition In nature an element may have several isotopes Isotopes have a specific percent composition no matter where the sample is collected on earth. liquid For example, oxygen in the air we breath has this composition: % AMU Oxygen-16 99.76% 15.994915 Oxygen-17 0.038% 16.999132 Oxygen-18 0.200% 17.999160 Total: 100.00% 9 Relative Abundance These %’s are known as The “Relative Abundances” of the isotope. In our example of Oxygen the Average AMU’s of a sample of Oxygen must be between 15.994915 and 17.999160. Why? The AVERAGE MASS of all the elements isotopes is called the ATOMIC MASS or the ATOMIC WEIGHT 10 THIS IS DIFFERENT THAN ATOMIC Mass Number Isotopic Composition So how do we calculate Atomic Mass? Well it’s simply a weighted average. Since we’re considering Oxygen …. Oxygen-16 Oxygen-17 Oxygen-18 total: Rel Abundance 99.762% 0.038% 0.200% 100% AMU 15.994915 16.999132 17.999160 So, What is the Atomic Mass for Oxygen? 11 Isotopic Composition Remember: the Atomic mass is the Avg. Atomic mass of all the elements isotopes & we need to use a weighted avg. to Calculate it. i.e. (Mass x abundance) + (mass x Abundance) etc. (.9976 x 15.994915) + (.00037 x 16.999132) + (.00204 x 17.999160) = (15.956527) + (0.006290) + (0.036718) = 15.9995 LOOK AT OXYGEN’S MASS ON THE PERIODIC TABLE. Is it pretty close to our answer ? 12 Estimating Atomic Mass Look at our Calculated average Atomic Mass of Oxygen (15.9995) and the relative abundance of the isotopes Of Oxygen. Abundance AMU Oxygen-16 99.762% 15.994915 Oxygen-17 0.038% 16.999132 Oxygen-18 0.200% 17.999160 Could you guess what the Calculated Atomic mass would be Close to? Which isotope is present in the greatest amount? Isn’t it’s mass pretty close to the calculated value? 13 Estimating Atomic Mass 1. In the following problem which isotope will have a mass Closest to the actual atomic mass? 2. Calculate the atomic mass of Chlorine (Assume these values are correct though they are not) Chlorine-35 Chlorine-37 Abundance 70% 30% AMU 35.0 37.0 1. Chlorine 35 2. (0.70 x 35) + (0.30 x 37) = 24.5 + 11.1 = 35.6 AMU 14