chemiosmosis
advertisement

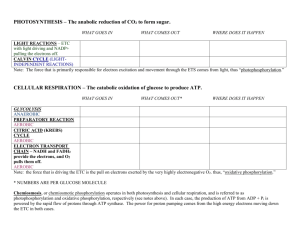
Cellular Respiration Part IV: Oxidative Phosphorylation Curriculum Framework • 2A2 Organisms capture and store free energy for use in biological processes. g. The electron transport chain captures free energy from electrons in a series of coupled reactions that establish an electrochemical gradient across membranes. 2 Figure 9.6-3 Electrons carried via NADH Glycolysis Glucose Pyruvate Electrons carried via NADH and FADH2 Citric acid cycle Pyruvate oxidation Acetyl CoA CYTOSOL Oxidative phosphorylation: electron transport and chemiosmosis MITOCHONDRION ATP ATP ATP Substrate-level phosphorylation Substrate-level phosphorylation Oxidative phosphorylation Curriculum Framework 2A2g2. In cellular respiration, electrons delivered by Oxidative NADH and FADH2 Phosphorylation: are passed to a series of electron acceptors as they move toward the terminal Electron Transport and electron acceptor, oxygen. Chemiosmosis Curriculum Framework 2A2g3. The passage of electrons is accompanied by the formation of a proton gradient across the inner mitochondrial membrane or the thylakoid t and Chemiosmosis membrane of chloroplasts, with the membrane separating a region of high proton concentration from a region of low proton concentration. In prokaryotes, the passage of electrons is accompanied by the outward movement of protons across the plasma membrane. Electron transport chain Outer mitochondrial membrane Inner mitochondrial membrane Electron carrier (NADH) Electrons Oxygen Hydrogen ions Electrons Water Hydrogen ions Area of high hydrogen ion concentration Inner mitochondrial membrane ATP ATP synthase Outer mitochondrial membrane Inner mitochondrial membrane Chemiosmosis: The Energy-Coupling Mechanism • Electron transfer in the electron transport chain causes proteins to pump H+ from the mitochondrial matrix to the intermembrane space • H+ then moves back across the membrane, passing through the enzyme, ATP synthase • ATP synthase uses the exergonic flow of H+ to drive phosphorylation of ATP • This is an example of chemiosmosis, the use of energy in a H+ gradient to drive cellular work Figure 9.14 INTERMEMBRANE SPACE H Stator Rotor Internal rod Catalytic knob ADP + Pi ATP MITOCHONDRIAL MATRIX Figure 9.15 H H H Protein complex of electron carriers Cyt c Q I IV III II FADH2 FAD NADH H 2 H + 1/2O2 ATP synthase H2O NAD ADP P i (carrying electrons from food) ATP H 1 Electron transport chain Oxidative phosphorylation 2 Chemiosmosis • The energy stored in a H+ gradient across a membrane couples the redox reactions of the electron transport chain to ATP synthesis • The H+ gradient is responsible for establishing a proton-motive force, emphasizing its capacity to do work Mitochondrial Membrane • Name and describe three structural features that make the mitochondrial membrane effective at the process of energy transfer. 15 ATP Production by Cellular Respiration Arrange these in order of energy transfer through chemiosmosis: • • • • • electron transport chain Glucose proton-motive force NADH ATP Figure 9.16 Electron shuttles span membrane 2 NADH Glycolysis 2 Pyruvate Glucose MITOCHONDRION 2 NADH or 2 FADH2 2 NADH Pyruvate oxidation 2 Acetyl CoA 2 ATP Maximum per glucose: CYTOSOL 6 NADH 2 FADH2 Citric acid cycle Oxidative phosphorylation: electron transport and chemiosmosis 2 ATP about 26 or 28 ATP About 30 or 32 ATP