Electron transport and chemiosmosis - MsBragg
advertisement

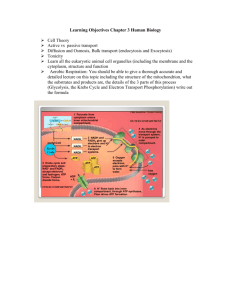
ELECTRON TRANSPORT AND CHEMIOSMOSIS Cellular Respiration: Stage 4 Review: Stages of Cellular Respiration (per 1 Glucose) Glycolysis: occurs in cytosol. Glucose 2 Pyruvate 2 ATP 2 NADH Pyruvate Oxidation: occurs in matrix 2 Pyruvate 2 acetyl-CoA 2 NADH Krebs Cycle: occurs in matrix 2 acetyl-CoA 2 oxaloacetate (cycle) 6 NADH 2 FADH2 2 ATP Stage 4: Electron Transport and Chemiosmosis NADH and FADH2 eventually transfer their hydrogen atom electrons to series of compounds in the ETC. Components of the ETC arranged in order of increasing electronegativity Weakest attractor of electrons at beginning of chain Strongest at the end. How it works... Each component is alternatively reduced and oxidized Reduced: gains two electrons from component before it in the chain Oxidized: by losing two electrons to component after it in the chain. Like a baton being handed from runner to runner in a relay race. As Electrons shuttle through the ETC... Going from less stable to more stable Therefore, energy is _____________. This energy is used to move H+ ions from the matrix into intermembrane space. Three proton pumps do this NADH dehydrogenase Cytochrome b-c1 complex Cytochrome oxidase complex At end of ETC... Electrons very stable, therefore, extremely electronegative substance needed _______________ strips two electrons from the final protein complex in the chain Two protons added from matrix to form water. Oxygen is the FINAL ELECTRON ACCEPTOR in the ETC. Oxygen is very ___________________. Components of the ETC 1) NADH dehydrogenase (Pump) 2) Ubiquinone (Q) (Mobile Electron Carrier) 3) Cytochrome b-cl complex (Pump) 4) Cytochrome c (Mobile Electron Carrier) 5) Cytochrome oxidase complex (Pump) NADH gives up its two electrons to NADH dehydrogenase. As electrons move through protein, they lose energy. This energy is used to pump H+ from matrix intermembrane space. Q shuttles electrons to Cytochrome b-c1 complex Electrons move through C b-c1 complex. Again, electrons _______ energy, powering the protein to pump H+ ions from the _________ to the ______________ space. C shuttles electrons from C b1-c complex to cytochrome oxidase. Electrons reach final protein complex in the chain Cytochrome Oxidase Complex Again, H+ is pumped into the intermembrane space. Contains the enzyme cytochrome oxidase: catalyzes the reaction between the electrons, protons, and molecular oxygen to form _________. Needed because Oxygen is electronegative enough to pull stabilized electrons through protein. Yeah...so? Electrons from NADH and FADH2 donated to electron transport. As electrons move through the chain of proteins, they lose energy. Energy from each 2e- used to pump one H+ ion from matrix to intermembrane space of mitochondrion. So, H+ is being moved OUT of the matrix. Results of this? ____________________________ Electrochemical Potential Energy Type of energy stored by a battery Caused by accumulation of charged objects The energy becomes stored in the electrochemical gradient and will be used to power ATP synthesis in the next part of the process... ____________________________________ EACH H+ in the intermembrane space eventually produces ONE ATP. NADH vs. FADH2 (Formed in the matrix) NADH passes electrons on to to NADH dehydrogenase Therefore, oxidation of each NADH molecule will help pump ________protons into the intermembrane space. EACH NADH WILL POWER THE FORMATION OF ____ ATP. FADH2 passes electrons on to Q. Therefore, oxidation of each FADH2 molecule will help pump _______ protons. EACH FADH2 WILL POWER THE FORMATION OF ____ ATP. Cytosolic NADH Cytosolic NADH is produced by ___________. May diffuse through outer membrane into intermembrane space. Intermembrane is impermeable to NADH Glycerol-phosphate shuttle: transfers electrons from cytosolic NADH to FAD (within matrix) to produce FADH2. FADH2 transfers electrons to Q ____ ATP. How many ATP will one cytosolic NADH eventually produce? _______________ Yazeed Essa Guilty… by Michael Small | March 5, 2010 at 02:29 pm Yazeed Essa, an Ohio Doctor, Has Been Found Guilty of Murdering His Wife with Cyanide Poison Yazeed Essa was convicted of murder on Friday, March 5th. The Ohio doctor was found guilty of lacing his wife's calcium pills with cyanide so that he could be with his mistress. Essa fled to Lebanon after his wife's death, but gave up an extradition fight and returned to Ohio to face trial. Yazeed Essa Guilty: Doctor Murdered Wife with Cyanide | NowPublic News Coverage http://www.nowpublic.com/world/yazeed-essa-guilty-doctormurdered-wife-cyanide-2587107.html#ixzz1FGGtO1QF Cyanide Cyanide inhibits cytochrome oxidase activity This prevents _____________ from acting as the final electron acceptor Shuts down the ETC, H+ pumps, and consequently, ATP production. Coma death Not poisonous to all organisms! MIT-13 (anaerobic bacteria) live on cyanide. Used in the same way as aerobes use oxygen. Importance of Cristae Allows multiple copies of the ETC to be located throughout inner membrane. Increased ______________________ for reactions to occur. ... and finally... CHEMIOSMOSIS and OXIDATIVE ATP SYNTHESIS(but some terms first) TERMINOLOGY Electrochemical Gradient: concentration gradient created by pumping ions into a space surrounded by a membrane that is impermeable to ions. Proton-motive force (PMF): a force that moves protons through an ATPase complex on account of the electrochemical gradient of proteins across a biological membrane. ETC forms an Electrochemical Gradient Electrochemical gradient formed by ETC Electrical component: higher positive charge in the _________________ space than the ________. Chemical component: higher concentration of protons in the ____________space than the _____________. Inner membrane impermeable to H+ ions. Intermembrane space becomes H+ reservoir. (H+ reservoir = proton reservoir) Potential difference (voltage) across inner mitochondrial membrane. Electrochemical Gradient drives Chemiosomosis Chemiosmosis: energy that drives synthesis of ATP comes from the “osmosis” of protons. ____ forced to diffuse through protein channels associated with ATP synthase (ATPase). Electrochemical gradient loses potential energy which is converted to chemical potential energy: ATP! This energy drives the synthesis of ADP + Pi ATP Electrochemical Gradient and the Formation of ATP Fate of ATP ATP molecules transported through both mitochondrial _______________ by ____________ diffusion into the ____________ where they are used to drive ___________ processes such as movement, active transport, and synthesis reactions. Relationship between ETC and Chemiosmosis? (review...) Electron transport chain obtains electrons from hydrogen atoms from _________ and ____________ molecules. At each sequential step in the ETC, electrons _________ energy by becoming more _____________. Energy is harnessed by pumping _________ into the _____ reservoir. ________________ gradient is formed, which forces _______ to diffuse back into the mitochondrial matrix via a __________ complex. Conditions Necessary for ETC and Chemiosmosis This is a continuous process H+ reservoir must be maintained requires ____________ movement of __________ through the ETC dependant on availability of ______________ to act as the final electron acceptor. Hence, why we have lungs and fish have gills: _______________________. Continuous source of electrons electrons are transferred via _______ and _______ coenzymes are formed during the first ____ stages of cellular respiration in the first three stages of cellular respiration, __________ is catabolized need of glucose means a need of _________. Hence, why heterotrophs must continually ____ and photoautotrophs must continually ___________________. Importance of Oxygen (in aerobes) No chemical is electronegative enough to oxidize the last protein in the chain, except for oxygen. If no oxygen no substance to act as final electon acceptor last protein can not be ‘freed up,’ ETC shuts down FADH2 and NADH can no longer be oxidized no NAD+ or FAD to recycle back into steps 1, 2 and 3. Homework: look this diagram over and study it until you understand the idea. The Exergonic Flow of Electrons in Aerobic Respiration What’s happening to the ‘lost’ energy? THE ENERGETICS OF OXIDATIVE PHOSPHORYLATION Overall – CELLULAR RESPIRATION! Seatwork/Homework Read page 109 – 110 Make notes and a diagram on the theoretical coenzyme and ATP yield. This is for YOU to study from! Answer the following questions: 1) The theoretical yield of ATP is 36. Give two reasons why the actual yield may differ from this. 2) What is the estimated number of ATP molecules formed for each glucose molecule? Read the section on “Efficiency of Energy Conversion...” Answer the following questions: 1) How is the efficiency of aerobic respiration calculated? 2) How does the efficiency of aerobic respiration differ from glycolysis? Seatwork/Homework Read the section “Metabolic Rate.” Make notes and answer the following questions: 1) What is metabolic rate? 2) What is BMR? For humans, how much energy does the BMR account for ? 3) What are some factors that effect the BMR? 4) What is a Benzinger calorimeter? Briefly, how does it work? (skip the calculation) Seatwork/Homework Read the section on “Controlling Aerobic Respiration.” Answer the following questions: 1) What is phosphofructokinase? How does it regulate aerobic respiration? (in terms of ADP, ATP, and citrate). 2) How do NADH levels regulate respiration?