Organic Chemistry1
advertisement

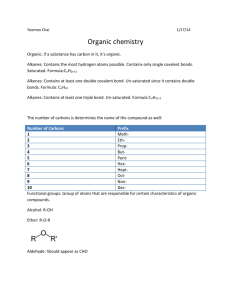
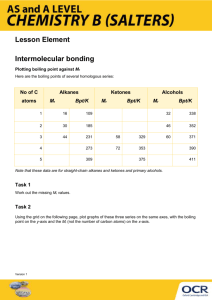
Organic Chemistry There are more than a million organic compounds HYDROCARBONS • The simplest organic compounds containing only carbon and hydrogens – Carbon has 4 valence electrons and therefore always forms 4 covalent bonds HYDROCARBON PREFIXES Prefix Meth- # of Carbons 1 EthPropBut- 2 3 4 PentHexHept- 5 6 7 OctNonDec- 8 9 10 Methods of Illustrating Hydrocarbons Formula C4H10 Description Molecular Formula Complete Structural Formula (Lewis Structure) CH3-CH2-CH2-CH3 Condensed Structural Formula: C-H bonds understood CH3CH2CH2CH3 Condensed Structural Formula: C-C and C-H bonds understood CH3(CH2)2CH3 Condensed Structural Formula: All bonds understood, parentheses indicate CH2 are linked in a continuous chain C-C-C-C Carbon skeleton: all hydrogens and C-H bonds understood Line-angle formula: All carbons and hydrogens understood; carbon atoms are located at each intersection and at the ends of lines. ALKANES • A hydrocarbon in which there are only single covalent bonds – In an alkane, all the carbon-carbon bonds are single covalent bonds • All other bonds are carbon-hydrogen bonds – The carbon atoms in an alkane can be arranged in a straight chain or in a chain that branches • Alkanes are called saturated compounds because they contain only single bonds Straight Chain Alkanes • Carbon atoms are one after another in a chain – Homologous Series: There is a constant increment of change from one compound in the series to the next • In an alkane a CH2 is the increment of change Naming Alkanes • • For all alkanes the name ends in –ane Count the carbon atoms and add the appropriate prefix Example: C6H14 To draw the structural formula: 1) Write the symbol for carbon as many times as necessary 2) Complete each carbon’s 4 bonds with hydrogen atoms Example: Octane Branched Chain Alkanes • An alkane with one or more alkyl groups – Substituent: An atom or group of atoms that takes the place of a hydrogen atom on a parent alkane – Parent Alkane: The longest continuous chain of a hydrocarbon – Alkyl Group: A hydrocarbon substituent Naming the alkyl groups • Name end in –yl • Add the appropriate prefix based on # of carbons in the alkyl group Practice Problems Drawing Structural Formulas for Alkanes 1) 2,2,4-trimethylpentane 2) 2,3-dimethylhexane 3) 4-ethyl-2,3,4-trimethyloctane 4) 3,3-dimethyl-4-ethyloctane 5) hexane 6) 2-methylbutane ISOMERS • Compounds that have the same molecular formula but different molecular structures – Structural Isomers: compounds that have the same molecular formula, but the atoms are joined together in a different order Differ in physical properties like boiling and melting points Also have different chemical reactivities The more highly branched the hydrocarbon structure, the lower the boiling point Unsaturated Compounds Compounds that contain double or triple carboncarbon bonds Alkenes: hydrocarbons containing one or more carboncarbon double covalent bonds Plastics (more reactive than alkane) Alkynes: hydrocarbons containing one or more carboncarbon triple covalent bonds Preparing organic solvents, starting material for polymers, oxy-acetylene flame for welding NAMING ALKENES/ALKYNES • The parent chain must contain the double/triple bond. Number from the side closest to that bond. • If it contains a double bond, it ends in –ene • If it contains a triple bond, it ends in –yne – # the double-triple bond – Name substituents as with alkanes Naming Examples Structural Formula Examples 1) Propyne 2) Propane 3) Propene HALOCARBONS • Alkane + Halogen Halocarbon + Hydrogen Halide CH4 + Cl2 CH3Cl + HCl • One of the branches is a halogen – Named chloro, fluoro, bromo, iodo • Building blocks for things like Teflon & PVC • Organic compounds can be classified according to their functional group – Functional Group: A specific arrangement of atoms in an organic compound that is capable of characteristic chemical reactions Functional Groups (R = any carbon chain) Compound Type Alcohol Compound Structure —OH Name ends in Functional Group -ol Hydroxyl Aldehyde -al Carbonyl Carboxylic Acid -oic acid Carboxyl Ester -oate Ester Ketone -one Ketone Naming: •drop the –e ending of the parent alkane name and add the ending to the proper functional group ending •Parent alkane is the longest continuous chain that includes the carbon attached to the functional group •If the functional group can occur at more than one position, its position is designated with the lowest possible number. •Alcohols containing 2, 3, or 4 –OH substituents are named diols, triols, and tetrols Compound Type Alcohol Compound Structure —OH Name ends in Functional Group -ol Hydroxyl Aldehyde -al Carbonyl Carboxylic Acid -oic acid Carboxyl Ester -oate Ester Ketone -one Ketone CHALLENGE NAME THIS! Organic Type Properties Uses Alkane Nonpolar Gases/Liquids Low boiling points Insoluble in water Gas/Oil/Fuels Methane: Natural Gas Propane: Homes/Grills Butane: Lighters Octane: Gasoline Alcohol Hydrogen Bonds Liquids Higher boiling points than the other groups Soluble in water (up to 4 carbons) Perfumes, Mouthwash, Hairspray, Antifreeze, Rubbing Alcohol, Antiseptic, Alcoholic Beverages Aldehyde (Ketones) Weak Hydrogen bonds Liquids or solids at room temperature Boiling points lower than alcohols higher than alkanes Soluble in water up to 5 carbons Flavoring agents Benzaldehyde: Almond oil Cinnamaldehyde: Cinnamon oil Vanilla flavoring Carboxylic Acid Hydrogen bonds Volatile liquids/Waxy solids Higher boiling points than other compounds Soluble in water up to 5 carbons Acetic acid: Vinegar Stearic acid: Wax candles Fatty acids Ester Polar but no hydrogen bonds (H not attached to O) Liquids Lower boiling points than other compounds Soluble in water up to 5 carbons Pleasant, fruity odors (blueberries, pineapples, pears, apples, bananas); perfumes & fragrances Boiling Points: Carboxylic Acid > Alcohol > Aldehyde > Alkane Solubility in Water Alkanes: Insoluble Alcohol/Aldehyde/Ester/Carboxylic Acid: Soluble up to 4-6 carbons SYNTHESIS OF ESTERS Esters may be prepared from a carboxylic acid and an alcohol NAMING ESTERS