Meeting 5. Acid-Base Titration
advertisement

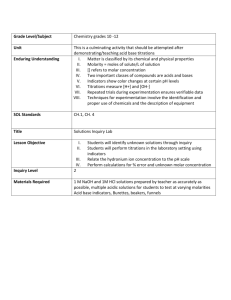
MEETING 5. ACID-BASE TITRATION - WHAT IS TITRATION? - WHY ACID-BASE TITRATION IS CALLED NEUTRALIZATION TITRATION? - WHAT IS ACIDIMETRIC TITRATION? - WHAT IS ALKALIMETRIC TITRATION? - WHAT IS THE PRINCIPLE OF REACTION? Titration Strong acid Weak acid Strong base Weak base Principle reaction: Involve the change of [H+] and [OH-] Involve the change of pH. What is pH? What is pH of 100 mL HCl 0,1 M? What is pH of 100 mL NaOH 0,1 M? Is pH of 100 mL HCl 0,1 M similar with pH of 100 mL CH3COOH 0,1 M Curve of Acid-base Titration HCl + NaOH NaCl + H2O Reaksi ionnya : H+ + OH- H2O Strong acid + Strong Base Example: HCl + NaOH NaCl + H2O Ionic reaction: H+ + OH- H2O What is the pH of titration? 10 mL HCl 0,1 M + 0 mL NaOH ? 10 mL HCl 0,1 M + 5 mL NaOH 0,1 M? 10 mL HCl 0,1 M + 10 mL NaOH 0,1 M? 10 mL HCl 0,1 M + 20 mL NaOH 0,1 M? HOW ABOUT THIS? 10 mL HCl 0,1 M + 0 mL NH4OH ? 10 mL HCl 0,1 M + 5 mL NH4OH 0,1 M? 10 mL HCl 0,1 M + 10 mL NH4OH 0,1 M? 10 mL HCl 0,1 M + 20 mL NH4OH 0,1 M? Indicator in Acid-base titration The color change should be as near as possible to equivalent point In Fact: it is very difficult end point of titration End Point ~ Equivalent Point How can we choose the indicator? pH range of some indicators Indikator Daerah pH Warna asam Warna basa Pelarut Thimol biru 1,2 - 2,8 Merah Kuning Air Metil kuning 2,9 – 4,0 Merah Kuning Etanol 95% Metil jingga 3,1 – 4,4 Merah Kuning jingga Air Metil merah 4,2 – 6,2 Merah Kuning Air Bromtimol biru 6,0 – 7,6 Kuning Biru Air Phenolphtalein 8,0 – 9,8 Tak berwarna Merahungu Etanol 70% Example: Contoh Perubahan Warna The color change of Acid-Base Indicator Indikator Asam-basa Universal Indicator