chemistry honors quarter 2 review
advertisement

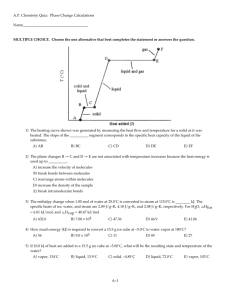
CHEMISTRY HONORS QUARTER 2 REVIEW (chapter numbers correspond with textbook chapters order) CHAPTER 8 Define the term ion Differentiate between a cation and anion Describe the composition of ionic compounds and list their characteristics Determine the probable charge of an element based on its periodic placement Construct the electron configuration for ions Define and give examples for ionic and metallic bonds Name compounds containing ions and polyatomic ions Construct chemical formulas based on chemical name CHAPTER 13 Define/ describe following terms: surface tension crystalline vs. amorphous capillary action sublimation volatility diffusion vapor pressure effusion boiling point hydrogen bond London-dispersion forces dipole-dipole forces viscosity phase diagram Kinetic molecular theory Graham’s law of effusion Dalton’s law of partial pressures Manometer Barometer CHAPTER 9 Name covalent compounds using prefixes Name acids (binary and oxyacid) Write the formula for a covalent compound from its name Write the formula for a binary and oxyacid from its name Construct Lewis structures for compounds Determine shape, bond angle, polarity & hybridization Define and explain covalent bonding Use electronegativity to predict bond polarity Compare the relative bond energy and length of single, double and triple bonds Define coordinate covalent bond and explain how one works VSEPR theory Define hybrid orbitals Describe the polarity of molecules and the formation of bonds between them List the characteristics of covalently bonded molecules Explain resonance. Draw resonance structures for molecules. CHAPTER 14 Convert the number of moles to grams, grams to moles, molecules to moles, moles to molecules, grams to molecules and molecules to grams. Boyle’s law Charles’s law Gay- Lussac's law Combined gas law CHAPTER 16 CHATPER 11 Determine the: o Molar mass of compounds o Percent composition o Molecular formula o Hydrates o Empirical formula Heating curves Specific heat capacity Heat of fusion Heat of vaporization Calorimetry endothermic / exothermic MULTIPLE CHOICE: Choose the best answer for each question. 1. Calculate the molecular weight of CrSO4? A. 100 g/mol B. 244 g/mol Cr = 52; S = 32; O =16 C. 400 g/mol D. 148 g/mol 2. What is the number of total atoms in a molecule of Fe3(PO4)2? A. 3 B. 5 C. 13 D. 12 3. The mass of 0.10 mol of H2O A. 18 g B. 180 g D. none of the above H = 1; O = 16 C. 1.8 g 4. How many mole(s) are in 300 g of CaCO3? A. 3 B. 300 Ca = 40; C = 12; O = 16. C. 9 D. 900 5. The mole is A. 1,000 grams. B. The number of C-atoms in 12.0 g of Carbon C. The SI unit for the amount of substance in Chemistry D. B and C 6. Which of the following element is diatomic? A. Iodine B. Copper C. Silicon D. Potassium 7. What is the electron configuration of Al3+? A. 1s2 2s2 2p6 3s2 3p6 B. 1s2 2s2 2p6 3s2 3p6 4d2 C. 1s2 2s2 2p6 3s2 D. 1s2 2s2 2p6 8. Which of the following has a linear shape? A. CH4 B. BCl4- C. CO2 D. All of these 9. If chlorine has an electronegativity of 3.0, what type of bond is formed in Cl2? A. ionic B. polar covalent C. nonpolar covalent D. Metallic 10. The concept used in writing the electron-dot structures of certain molecules that oscillate between two or more possible structures is called: A. hybridization B. electrostatic repulsion C. resonance D. electronegativity 11. Which of the following compounds violates the octet rule? A. NF3 B. CCl4 C. SbCl5 D. Cl2 12. Which of the following is the most polar bond? EB = 2.0; EC = 2.5; EO = 3.5; ES = 2.5 A. B-C B. S-O C. C-O D. B-O 13. Which substance below is composed of cations surrounded by a “sea of mobile electrons”? A. Mn B. Si C. NaCl D. CH4 14. The VSEPR Theory basically: A. explains why bonds form B. helps predict the shape of the molecules C. has to do with why hybrid orbitals form D. allows scientist to calculate the bond energies 15. A triple bond is made of sharing of _____ electrons between two atoms. A. 2 B. 4 C. 6 D. 3 FREE RESPONSE: 16. A compound is found to be 40.00% carbon, 6.67% hydrogen, and 53.33% oxygen. Its molar mass is 210.0 g/mol. What is the molecular formula of this compound? C7H14O7 17. Name the following: a. b. c. d. e. f. g. h. P2O5 Ca(ClO3)2 NH4F AlN Fe(NO2)3 SnO2 H2SO3 H3P Write formulas for the following: diphosphorus pentoxide calcium chlorate ammonium fluoride aluminum nitride iron(III) nitrite tin(IV) oxide or tin(II) peroxide sulfurous acid hydrophosphoric acid a. b. c. d. e. f. g. h. lead (IV) sulfide hydrosulfuric acid dinitrogen trioxide chromium (III) hydroxide chromic acid sodium carbonate zinc oxalate sulfur hexafluoride PbS2 H2 S N2O3 Cr(OH)3 H2CrO4 Na2CO3 ZnC2O4 SF6 18. Fill in the following chart Formula Lewis Structure Geometry Bond Angles 107° Hybridizati on sp3 Molecular Polarity polar pyramidal 104° sp3 polar H2S bent or angular 109.5° sp3 nonpolar CCl4 tetrahedra l linear 180° sp nonpolar trigonal planar 120° sp2 polar trigonal bipyramid al 90°, 120° sp3d nonpolar octahedral 90° sp3d2 nonpolar PH3 6 lone pr on each Cl CS2 CH2O AsI5 (substitute I for F) SeBr6 (substitute Br for F) 19. Find the mass of 2.25 x 1021 formula units of magnesium iodate. Answer: 1.40g 20. Find the mass percent of water in nickel(II) cyanide tetrahydrate. Answer: 39.43% H2O 21. A 5.0g sample of nickel(II) cyanide hydrate is heated until all water is removed. What mass of anhydrous nickel(II) cyanide will remain? (REFER BACK TO #20 ON PREVIOUS PAGE!) Answer: 3.0g Ni(CN)2 22. Identify all types of intermolecular forces in a-d: a. Attraction between any two polar molecules ___dipole- dipole__________________ b. Very weak force that increases with molar mass ___London dispersion________________ c. Attraction between two induced dipoles _____ London dispersion ____________________ d. Very strong attractive force between molecules with N-H, O-H, or F-H bonds___H- bond______ 23. Compare and contrast solids and liquids 24. Identify the type of solid in a-e: a. Every atom is covalently bonded to another atom ___network covalent_________________ b. Atoms are surrounded by a sea of electrons _metallic________________________ c. Particles are connected only by IMF ___Molecular___________________________ d. There is no geometric pattern in the structure ______amorphous_________________ e. Charged particles in a geometric pattern _____ionic______________________ 25. Explain the relationship between strong intermolecular forces and the following properties – volatility, vapor pressure and boiling point: __The stronger IMFs , the lower the vapor pressure, the higher the boiling point. Volatility is the tendency of a substance to vaporize. Volatility is directly related to a substance's vapor pressure. At a given temperature, a substance with higher vapor pressure vaporizes more readily than a substance with a lower vapor pressure. 26. Refer to the phase diagram below when answering the questions: a. What is the normal freezing point of this substance? __100°C______ b. What is the physical state of this substance at 300°C and 0.50 atm ? __gas __________ c. If I had a quantity of this substance at a pressure of 1.25 atm and a temperature of 3000 C and lowered the pressure to 0.25 atm, what phase transition(s) would occur? _vaporization________ 27. Indicate whether a heating curve would be flat or rising in a-e. a. Liquid is boiling _flat__________ b. Potential energy is increasing __flat__________ c. Solid is warming __rising_________ d. Kinetic energy is increasing ___rising___________ e. Solid is melting ____flat__________ 28. How much heat does a 23.0 g ice cube absorb as its temperature increases from -17.4°C to 0.0°C? (use constants from homework sheets). 836 J 29. A 50.6 g sample of silver at 97.4°C is placed into 104 g of water at 19.7°C. What is the final temperature of the system? (CAg= 0.235J/g°C) 21.8°C 30. How much heat is required to change 20.0 g of ice at -13.0°C to steam at 125.0°C? (use constants from homework sheets) 61.7 kJ 31. 50.0 L of gas has a temperature of 75°C. What is the Celsius temp when the volume changes to 110. L? 493°C 32. A balloon is filled with 3.0 L of helium at 1 atm. What is the volume when the balloon rises to an altitude where the pressure is only 0.25 atm? (assume temperature remains constant.) 10L 33. The gas left in a used aerosol can is at a pressure of 1 atm at 27°C. If this can is thrown into a fire, what is the internal pressure of the gas when its temperature reaches 927°C? 4 atm 34. 5.2 L of a gas is at STP. Find the new volume when the temperature rises to 38°C and the pressure drops to 600 mmHg. 8L 35. Pressure and volume of a gas at constant temperature are ____ inversely ___ proportional. Why? Particles collide more often in the smaller amount of space. 36. Volume and temperature of a gas at constant pressure are ___directly_______________ proportional. Why? Faster moving particles need more space in order to keep pressure constant. 37. Which travels faster CO2 or O2? How much faster? O2, it travels 1.17x faster 38. Gas A travels 4 times faster than Gas B. If the molar mass of Gas B is 80 g/mole, find the molar mass of Gas A. 5 g/mol 39. A 250. ml sample of oxygen is collected over water at 25 ° C and 760.0 mmHg pressure. What is the pressure of the dry oxygen alone? (Vapor pressure of water at 25°C = 23.8 mmHg). 736. 2 mmHg