Net ionic equation
advertisement

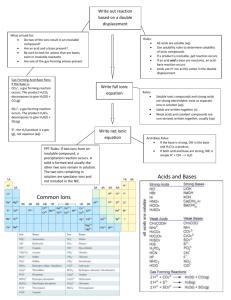
Vocabulary In SOLUTION we need to define the SOLVENT the component whose physical state is preserved when solution forms SOLUTE the other solution component The CONCENTRATION is most often expressed by chemists as MOLARITY. Other common expressions of concentration are w/w, vol/vol, and normality. number of moles of solute molarity number of liters of solution moles M L mmol M mL 1.0 L of water was used to make 1.0 L of solution. Notice the water left over. CCR, page 206 PROBLEM: Dissolve 5.00 g of NiCl2•6 H2O in enough water to make 250 mL of solution. Calculate molarity of the solution and the concentration of each of the ions. Problem: What mass of oxalic acid, H2C2O4, is required to make 250. mL of a 0.0500 M solution? PROBLEM: You have 50.0 mL of 3.0 M NaOH and you want 0.50 M NaOH. What do you do? SOLUTION STOICHIOMETRY Section 5.10 • Zinc reacts with acids to produce H2 gas. • Have 10.0 g of Zn • What volume of 2.50 M HCl is needed to convert the Zn completely? Zinc reacts with acids to produce H2 gas. If you have 10.0 g of Zn, what volume of 2.50 M HCl is needed to convert the Zn completely? Aqueous Solutions Non-Electrolytes – they dissolve, but won’t conduct electricity. These are generally polar solids capable of forming hydrogen bonds – Sugar, ethanol, ethylene glycol Strong Electrolytes – Completely dissociates, conducts electricity well – most ionic solids and strong acids Weak Electrolytes – Dissolve, ionize to a small extent, conducts electricity weakly. Weak acids, such as Acetic acid, ionize only to a small extent, so it are a weak electrolyte. CH3CO2H(aq) → CH3CO2-(aq) +H+(aq) Water Solubility of Ionic Compounds If one ion from the “Soluble Compound” list is present in a compound, the compound is water soluble. There are three ways to write reactions in aqueous solutions. Molecular equation: Show all reactants & products in molecular or ionic form Zn (s) + CuSO 4(aq) ZnSO 4(aq) + Cu(s) Total ionic equation: Show the ions and molecules as they exist in solution Zn (s) + Cu 2 aq + SO 24 aq Zn 2 aq + SO 24 aq + Cu (s) Net ionic equation: Shows ions that participate in reaction and removes spectator ions. Spectator ions do not participate in the reaction. Zn (s) + Cu 2 aq Zn 2 aq + Cu (s) Net Ionic Equations Mg(s)+ 2HCl(aq)→ H2(g) + MgCl2(aq) The molecular formula above can be written as the total ionic formula Mg(s)+ 2H+(aq)+ 2Cl-(aq)→ H2(g)+ Mg2+(aq)+ 2Cl-(aq) The two Cl- ions are SPECTATOR IONS — they do not participate. Could have used NO3- for the spectator ion as salts of nitrates are all soluble. By leaving out the spectator ions out you get the net ionic reaction Mg(s) + 2 H+(aq) ---> H2(g) + Mg2+(aq) ACIDS Table 5.2 A Brönsted-Lowry Acid → H+ in water Strong Brönsted-Lowry acids are strong electrolytes HCl H2SO4 HClO4 HNO3 hydrochloric sulfuric perchloric nitric HNO3 Weak Brönsted-Lowry acids are weak electrolytes CH3CO2H H2CO3 H3PO4 HF acetic acid (CH3COOH) carbonic acid phosphoric acid hydrofluoric acid Carbonic Acid Acetic acid BASES Table 5.2 Brönsted-Lowry Base → OH- in water NaOH(aq) → Na+(aq) + OH-(aq) NaOH is a strong base Ammonia, NH3 an Important weak Base ACIDS Nonmetal oxides can be acids CO2(aq) + H2O(l) → H2CO3(aq) SO3(aq) + H2O(l) → H2SO4(aq) NO2(aq) + H2O(l) → HNO3(aq) Acid Rain is an example of nonmetal oxides behaving as acids. This process can result from burning coal and oil. BASES Metal oxides can be bases CaO(s)+H2O(l) → Ca(OH)2(aq) CaO in water. Phenolphthalein indicator shows a of calcium oxide solution is basic. You should know the strong acids & bases A brief history of Acid-Base Identification Systems System Arrhenius BrönstedLowry Lewis Acid (HCl) Base (NaOH) pH, a Concentration Scale pH: a way to express acidity -- the concentration of H+ in solution. Low pH: high [H+] Acidic solution Neutral Basic solution High pH: low [H+] pH < 7 pH = 7 pH > 7 The pH Scale pH = log (1/ [H+]) = - log [H+] In a neutral solution, [OH-] = 1.00 x 10-7 M at 25 oC pH = - log [H+] = If the [H+] of soda is 1.6 x 10-3 M, the pH is ____. If the pH of Coke is 3.12, it is _____. [H+] = ACID-BASE REACTIONS Titrations H2C2O4(aq) + 2 NaOH(aq) → Na2C2O4(aq) + 2 H2O(l) acid base Carry out this reaction using a TITRATION. Oxalic acid, H2C2O4 Titration 1. Add solution from the buret. 2. Reagent (base) reacts with compound (acid) in solution in the flask. 3. Indicator shows when exact stoichiometric reaction has occurred. 4. Net ionic equation H+ + OH- --> H2O 5. At equivalence point moles H+ = moles OH- PROBLEM: Standardize a solution of NaOH — i.e., accurately determine its concentration. 1.065 g of H2C2O4 (oxalic acid) requires 35.62 mL of NaOH for titration to an equivalence point. What is the concentration of the NaOH? PROBLEM : Use standardized NaOH to determine the amount of an acid in an unknown. Apples contain malic acid, C4H6O5. 76.80 g of apple requires 34.56 mL of 0.663 M NaOH for titration. What is weight % of malic acid? C4H6O5(aq) + 2NaOH(aq) → Na2C4H4O5(aq) + 2 H2O(l) Types of Reactions: 1. Combination Reactions • More than one reactant, one product 2. Decomposition Reactions • Single reactant, more than one product 3. Displacement Reactions • One element displaces another from a compound 4. Redox – Oxidation Reduction Reactions • Oxidation numbers of some elements change; at least one element must increase and one must decrease in oxidation number. 5. Metathesis Reactions - Exchange Reactions • Precipitation: products include an insoluble substance which precipitates from solution as a solid • Acid-base neutralization: product is a salt and water • Gas formation – primarily the reaction of metal carbonates OXIDATION NUMBERS NH3 ClOH3PO4 MnO4Cr2O72- Recognizing a Redox Reaction 2 Al(s) + 3 Cu2+(aq) → 2 Al3+(aq) + 3 Cu(s) Oxidation-Reduction Reactions Thermite reaction Fe2O3(s) + 2Al(s) → 2 Fe(s) + Al2O3(s) 2Al(s) + 3Cu2+(aq) → 2Al3+(aq)+ 3Cu(s) 2 H2(g) + O2(g) → 2H2O(l) In all reactions if something has been oxidized then something has also been reduced. Redox reactions are characterized by ELECTRON TRANSFER between an electron donor and electron acceptor. Transfer leads to 1. increase in oxidation number of some element = OXIDATION 2.decrease in oxidation number of some element = REDUCTION Chemical Reactions in Water Metathesis • EXCHANGE REACTIONS The anions exchange places between cations. AX + BY AY + BX Precipitation Pb(NO3)2(aq) + 2 KI(aq) → PbI2(s) + 2KNO3(aq) Pb2+(aq) + 2 I-(aq) → PbI2(s) Neutralization: NaOH(aq) + HCl(aq)→ NaCl(aq)+ H2O(l) OH-(aq) + H+(aq) → H2O(lq) Gas Formation MgCO3(s)+ 2HCl(aq) → 2Mg(Cl)2(aq)+ H2O(l) + CO2(g)