Program Editors
Ralph Anthony DeFronzo, MD
Jaime A. Davidson, MD
Professor of Medicine and
Chief of the Diabetes Division
University of Texas Health
Science Center
Audie L. Murphy Memorial Veterans Hospital
San Antonio, Texas, USA
President, Worldwide Initiative
for Diabetes Education
Clinical Professor of Internal Medicine
Division of Endocrinology
University of Texas Southwestern
Medical School
Dallas, Texas, USA
Faculty
Professor Stefano Del Prato
Professor Rury Holman
Professor of Endocrinology and Metabolism
School of Medicine
University of Pisa
Pisa, Italy
Professor of Diabetic Medicine
Honorary Consultant Physician
Diabetes Trials Unit
University of Oxford
Oxford, United Kingdom
Professor Allan Vaag
Chief Physician
Steno Diabetes Center
Gentofte, Denmark
Educational Objectives
Upon completion of this activity, participants will be able to
• Name 5 current challenges for glycemic control in individuals
with type 2 diabetes
• List the key physiologic, biochemical, and molecular events
involved in the renal regulation of glucose metabolism
• Understand the effects of inhibiting glucose reuptake by the
kidney in individuals with type 2 diabetes
Magnitude of the Diabetes Epidemic
Global Projections for the
Diabetes Epidemic: 2007-2025
EUR
NA
53.2 M
64.1 M
20%
28.3 M
40.5 M
43.0%
EMME
SEA
24.5 M
44.5 M
82%
46.5 M
80.3 M
73%
AFR
World
2007=246 M
2025=380 M
54%
SACA
16.2 M
32.7 M
102%
WP
67.0 M
99.4 M
48%
10.4 M
18.7 M
80%
2007
2025
M=million; AFR=Africa; EMME=Eastern Mediterranean and Middle East; EUR=Europe; NA=North America;
SACA=South and Central America; SEA=South-East Asia; WP=Western Pacific.
International Diabetes Federation. Diabetes Atlas. 3rd ed. Available at: http://www.eatlas.idf.org/index.asp.
Global Increase in Obesity
2002
2007
2015
Obese
356 million
523 million
704 million
Overweight
1.4 billion
1.5 billion
2.3 billion
Prevalence of Obesity (%)
35
USA
30
25
UK
20
Australia
Finland
15
Sweden
Norway
Brazil
Cuba
10
5
0
1970
Japan
1975
1980
1985
1990
1995
Overweight, BMI ≥25 kg/m2; obese, BMI >28 kg/m2 (Asian) or >30 kg/m2.
James WP. J Intern Med. 2008;263:336-352.
2000
2005
Increasing Problem of Obesity
and Diabetes: United States
Obesity*
Diabetes
92% increase
40
34.3
20
17.9
0
1998
2006
20% increase
10
40
US
Population
(%)
7.8
6.5
20
5
0
0
1998
2007
*BMI ≥30 kg/m2.
Centers for Disease Control and Prevention. National diabetes fact sheet. Atlanta, GA: U.S.
Department of Health and Human Services, Centers for Disease Control and Prevention, 2008;
Mokdad AH, et al. JAMA. 1999;282:1519-1522; Mokdad AH, et al. Diabetes Care. 2000;23:1278-1283;
Ogden CL, et al. NCHS data brief no 1. Hyattsville, MD: National Center for Health Statistics, 2007.
Increasing Problem of Obesity
and Diabetes: Mexico
Obesity
Diabetes
21% increase
17% increase
40
40
25.1
19.4
14.9
0
Men Women
1993
Men
Women
8.2
Mexican
Population
(%)
29.0
20
10
7
20
5
0
0
1993
2000
Aguilar-Salinas CA, et al. Am J Med. 2002;113:569-574; Aguilar-Salinas CA, et al.
Diabetes Care. 2003;26:2021-2026; Sánchez-Castillo CP, et al. Public Health Nutr.
2005;8:53-60.
2000
Increasing Problem of Obesity
and Diabetes: China
Obesity*
Diabetes
169% increase
120% increase
10
9.0
5.5
Chinese
Population
(%)
6.9
5
6
10
4.1
5
3
0
0
2.5
1.8
0
Men Women
1991
Men
Women
1999-2000
*Asian-specific obesity cut-point: BMI ≥28 kg/m2.
Gu D, et al. Diabetologia. 2003;46:1190-1198;
Wildman RP, et al. Obesity (Silver Spring). 2008;16:1448-1453.
1994
2000-2001
Increasing Problem of Weight
Gain and Diabetes: India
Overweight*
Diabetes
750% increase
20
17.1
191% increase
8
20
6.4
Rural Indian
Population
(%)
10
10
4
2.2
2.0
0
0
1989
2003
*BMI ≥25 kg/m2.
Ramachandran A, et al. Diabetologia. 2004;47:860-865.
0
1989
2003
Hyperglycemia
• Biochemical marker by which the diagnosis of diabetes is made
– Assessed with HbA1c, daily SMBG, and eAG
• Major and treatable risk factor for microvascular disease (DCCT,
UKPDS 33 and 35)
• Independent and treatable risk factor for macrovascular disease
(DCCT-EDIC, UKPDS 35 and 80)
• Self-perpetuating cause of diabetes
– Glucotoxicity → insulin resistance and impaired insulin secretion
eAG=estimated average glucose.
SMBG=self-monitoring of blood glucose.
HbA1c Is Correlated With
Average Glucose
450
400
AG (mg/dL)
350
300
250
200
150
100
50
0
3
5
9
7
HbA1c (%)
AG=average glucose.
Nathan DM, et al. Diabetes Care. 2008;31:1473-1478.
11
13
15
Diabetes Report Card: HbA1c Levels
in the United States
100
80
6%
6%
11%
>10.0
9.0-9.9
20%
7.0-7.9
8.0-8.9
60
Patients
(%)
HbA1c
(%)
40
35%
6.0-6.9
22%
<6.0
20
0
Hoerger TJ, et al. Diabetes Care. 2008;31:81-86.
Advances in Therapy,
but Falling Short of Goals
10
ACCORD,
1997: 1998:
ADA lowered
2005:
eliminated
ADA2008:
added
HbA
UKPDS results 2003: ADA
1c added
2009:
ADA
VADT,
T2DM diagnosis
from
HbA1c “action point”
goal ADVANCE,
ofof
<6%
for
published
“less stringent”
and
UKPDSgoal
80 for
FPG ≥7.8 mmol/L
<8% from“individual
guidelines
patients”
to
HbA
1c
published
to ≥7.0 mmol/L
guidelines
patients with
significant
NHANES
comorbidities or risk
1999of hypoglycemia, or
NHANES short life expectancy
2000
7.8
Pre-DCCT
9.0%
HbA1c (%)
9
2003-2004
8
7
General ADA
Target: <7%
7.2
7.7
NHANES
1988-1994
6
SU / Insulin
Metformin (1995) TZDs (1998)
7.5
NHANES
2001-2002
Incretins (2004)
5
1980s
1990s
2000s
SU=sulfonylurea; TZDs=thiazolidinediones; T2DM=type 2 diabetes.
Koro CE, et al. Diabetes Care. 2004;27:17-20; Hoerger TJ, et al. Diabetes Care. 2008;31:81-86.
Future
6.0% ?
Unmet Needs in Diabetes Care
Multiple
Defects in Type 2
Diabetes
Weight
Management
Adverse Effects
of Therapy
Type 2
Diabetes
Hyperglycemia
CVD Risk
(Lipid and
Hypertension
Control)
CVD=cardiovascular disease.
Adapted from © 2005 International Diabetes Center, Minneapolis, MN. All rights reserved.
Relationship Between Hyperglycemia and
Microvascular and Macrovascular Complications
Incidence of Microvascular
Complications in IGT
Diabetic Retinopathy (%)
IGT (HbA1c=5.9%)
IGT………..…7.9%
IGT (HbA1c=6.1%)
T2DM………12.6%
Neuropathy (%)
IGT………..…13%*
*Prevalence.
Diabetes Prevention Program Research Group. Diabet Med. 2007;24:137-144; Singleton JR,
et al. Diabetes Care. 2001;24:1448-1453; Ziegler D, et al. Diabetes Care. 2008;31:464-469.
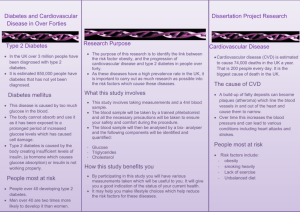
Diabetes Is a Cardiovascular Disease
Risk Equivalent
Nondiabetic
Diabetic
n=1373
n=1059
P<0.001
50
45.0
40
7-Year
Incidence
Rate of MI
(%)
30
P<0.001
20
18.8
20.2
No DM
MI
DM
No MI
10
3.5
0
No DM
No MI
DM=diabetes mellitus; MI=myocardial infarction.
Haffner SM, et al. N Engl J Med. 1998;339:229-234.
DM
MI
Historic Rationale for Improving Glycemia:
Microvascular Risk Reduction
80
Microvascular Disease
70
Estimated 37%
decrease in
microvascular
risk for each 1%
decrement in
HbA1c (P<0.0001)
60
Incidence per
1000 PersonYears
(%)
50
40
30
20
10
0
5
6
7
8
9
Mean HbA1c (%)
Stratton IM, et al. BMJ. 2000;321:405-412.
10
11
Less Strong Association Between
Hyperglycemia and Macrovascular
Risk in Type 2 Diabetes
80
Microvascular Disease
70
Macrovascular Disease
Estimated 37%
decrease in
microvascular
risk for each 1%
decrement in
HbA1c (P<0.0001)
60
Incidence per
1000 PersonYears
(%)
50
40
Estimated 14%
decrease in
myocardial
infarction risk
for each 1%
decrement in
HbA1c (P<0.0001)
30
20
10
0
5
6
7
8
9
Mean HbA1c (%)
Stratton IM, et al. BMJ. 2000;321:405-412.
10
11
Optimizing Glycemia in Advanced Type 2
Diabetes Exerts Unclear Macrovascular Benefit
ADVANCE
N=11,140
9
ACCOR
D
N=10,251
VADT
N=1791
Conventional
therapy
8
Endpoint
HbA1c (%)
7
Intensive therapy
6
Macro
Macro
Macro
↓6%
P=0.37
↓10%
P=0.16
↓13%
P=0.12
Primary Endpoint
ACCORD Study Group. N Engl J Med. 2008;358:2545-2559; ADVANCE Collaborative Group.
N Engl J Med. 2008;358:2560-2572; Duckworth W, et al. N Engl J Med. 2009;360:129-139.
Lasting Benefits of Early, Intensive Intervention:
UKPDS “Legacy” Effect
Intervention
Microvascular
Disease
Myocardial
Infarction
All-cause
Mortality
-6
-12
-16
-20
P=0.029
-25
P=0.44
P=0.052
P=0.0099
-40
Post-trial Monitoring
Relative Risk Reduction (%)
0
Any Diabetes
Endpoint
0
-9
-20
-15
P=0.040
-24
P=0.014
-13
P=0.007
P=0.001
-40
Holman RR, et al. N Engl J Med. 2008;359:1577-1589; UKPDS Study Group. Lancet. 1998;352:837-853.
Early vs Late Intervention in Type 2
Diabetes
Trial
Intensive Arm
HbA1c Reduction
Goal: <6.0%
ACCORD
Endpoint: 6.4%
↓1.4% from BL in 4 months
Goal: <6.5%
ADVANCE
Endpoint: 6.5%
↓0.6% from BL in 12 months
Goal: ↓1.5% vs standard
VADT
Endpoint: 6.9%
↓2.5% from BL in 3 months
UKPDS 80
No Patients /
Trial Duration
N=10,251
3.4 yr
N=11,140
Disease Severity
CVD or 2 risk
factors
10 yr from T2DM
diagnosis
Vascular disease or
1 risk factor
5.0 yr
8 yr from T2DM
diagnosis
N=1791
12 yr from T2DM
diagnosis
5.6 yr
Goal: FPG <6.0 mmol/L
(108 mg/dL)
N=4209
Intervention endpoint: 7.0%
17 yr
Macrovascular
Benefit
Newly diagnosed
with T2DM
No
Yes
Follow-up: 7.7%
ACCORD Study Group. N Engl J Med. 2008;358:2545-2559; ADVANCE Collaborative Group. N Engl J Med.
2008;358:2560-2572; Duckworth W, et al. N Engl J Med. 2009;360:129-139; Holman RR, et al. N Engl J Med.
2008;359:1577-1589.
Steno-2: Time to Cardiovascular Events
Conventional Treatment
80
60
Cumulative
Incidence of
Any CV Event 40
(%)
P<0.001
20
Intensive Treatment
Intervention
0
0
No. at Risk
Conventional
Intensive
2
4
Follow-up
6
8
10
12
38
56
29
50
25
47
Years
80
80
70
72
60
65
Gaede P, et al. N Engl J Med. 2008;358:580-591.
46
61
14
31
Steno-2: Goal Attainment
Intensive therapy
Conventional therapy
Intervention
P<0.001
80
60
40
20
P=0.21
P=0.005
P=0.001
P=0.06
0
100
Follow-up
Patients (%)
100
P=0.35
80
60
40
20
0
P=0.27
P=0.31
HbA1c
<6.5%
P=0.14
P=0.005
Cholesterol
<175 mg/dL
BP=blood pressure.
Gaede P, et al. N Engl J Med. 2008;358:580-591.
Triglycerides
<150 mg/dL
Systolic BP
<130 mm Hg
Diastolic BP
<80 mm Hg
Etiology of Type 2 Diabetes
Insulin Resistance and -Cell Dysfunction
Etiology of Type 2 Diabetes
Primary Predisposing
Factors
• Genes
• Adverse intrauterine
environment
Tertiary Accelerating
Factors
• Glucose and lipid toxicity
Secondary Precipitating
Factors
• Obesity
• Low physical activity
• Age
• Smoking
• Sleep disturbance
• Other
Type 2 Diabetes: A Heterogeneous
Disorder
Functional -cell
Failing -cell
Insulin resistance
Insulin resistance
Metabolic syndrome
Heine RJ, Spijkerman AM. 2006.
Hyperglycemia
Type 2 Diabetes: Insulin Resistance
Plus Impaired -Cell Function
Both insulin resistance and -cell dysfunction are present
at the time of diagnosis of type 2 diabetes
Insulin
resistance
Normal
-cell
function
Abnormal
-cell
function
Compensatory
hyperinsulinemia
Relative insulin deficiency
Normoglycemia
Type 2 diabetes
(Metabolic syndrome)
Hyperglycemia
Natural History of Type 2 Diabetes
300
140
Mean
Plasma Insulin
During OGTT
(µU/mL)
Mean
Plasma Glucose
During OGTT
(mg/dL)
250
100
200
60
150
20
400
100
300
200
100
Lean OB OB- OBNGT NGT IGT DM
Hi INS
OBDM
Lo INS
DM=diabetes mellitus; IGT=impaired glucose tolerance;
INS=insulin; NGT=normal glucose tolerance; OB=obesity.
DeFronzo RA. Diabetes. 1988;37:667-687; Jallut D, et al. Metabolism. 1990;39:1068-1075.
InsulinMediated
Glucose
Uptake
(mg/m2 • min)
Etiology of -Cell Dysfunction
in Type 2 Diabetes
↓ Incretin
Effect
Amyloid (Islet
Amyloid
Polypeptide)
Deposition
Age
-Cell
Dysfunction
Glucose
Toxicity
Genetics
(TCF 7L2)
Insulin
Resistance
Lipotoxicity
↑ Free Fatty Acids
Natural History of -Cell Dysfunction
in Type 2 Diabetes
β-Cell failure occurs much
earlier in the natural history
of type 2 diabetes and is more
severe than previously
appreciated
San Antonio Metabolism and
VAGES Studies
Subjects
Normal glucose tolerance
Impaired glucose tolerance
Type 2 diabetes
Number
318
259
201
Subjects were classified as
Nonobese
Obese
if
if
BMI <30 kg/m2
BMI ≥30 kg/m2
Methods: OGTT and insulin clamp
VAGES=Veterans Administration Genetic Epidemiology Study.
Abdul-Ghani MA, et al. Diabetes. 2006;55:1430-1435; Ferrannini E, et al.
J Endocrinol Metab. 2005;90:493-500; Gastaldelli A, et al. Diabetologia. 2004;47:31-39.
Plasma Glucose and Insulin AUC
NGT
IGT
T2DM
Gastaldelli A, et al. Diabetologia. 2004;47:31-39.
NGT
IGT
T2DM
Q4
Q3
Q2
0
Q1
4
<200
Q4
Q3
Q2
Q1
<160
0
<200
4
8
<180
8
<160
Insulin AUC
(pmol/L 120 min)
12
<180
Glucose AUC
(mmol/L 120 min)
12
Insulin Secretion / Insulin Resistance
(Disposition) Index During OGTT
40
Lean
30
∆I/∆G
÷IR
20
10
Obese
0
NGT
IGT
T2DM
2-Hour Plasma Glucose (mg/dL)
G=glucose; I=insulin; IR=insulin resistance.
Gastaldelli A, et al. Diabetologia. 2004;47:31-39.
Log Normalization of the Relationship Between
2-Hour Plasma Glucose and Insulin Secretion /
Insulin Resistance Index
6
NGT
IGT
4
Ln ∆I / ∆G
÷ IR
(mL/min • kgFFM)
T2DM
2
0
-2
r=0.91
P<0.00001
-4
4.0
4.5
5.0
5.5
Ln 2-Hour Plasma Glucose
(mg/dL)
Ln=log normalization.
Gastaldelli A, et al. Diabetologia. 2004;47:31-39.
6.0
6.5
GENFIEV: Insulin Secretion as a
Function of Insulin Sensitivity
0.04
0.03
Δ AUC
C-peptide /
Δ AUC
Glucose ÷
HOMA-R
Trend test
P<0.001
0.02
0.01
0
<100
120
140
160
180
200
240
2-Hour Plasma Glucose
(mg/dL)
HOMA-R=homeostasis model assessment index ratio.
Diabetes. 2006;55(suppl 2):A322.
280
>280
GENFIEV:
Stimulus-Response
CurveControl) of Insulin
GeNFIEV:
Stimulus-response
Curve (Proportional
Secretion
(Proportional Control) of Insulin Secretion
NFG/NGT
IFG/NGT
NFG/IGT
IFG/IGT
NFG/DGT
IFG/DGT
DFG/IGT
DFG/DGT
Insulin Secretion Rate
(pmol . min-1 . m-2)
1200
1000
*
800
600
§
400
#
200
0
3
6
9
Plasma Glucose (mmol/L)
*P<0.01 vs NFG/NGT; §P<0.05 vs NFG/IGT and IFG/NGT; #P<0.05 vs IFG/IGT and NFG/DGT.
Diabetes. 2006;55(suppl 2):A2472.
12
Insulin Secretion and Insulin Resistance
in Different Ethnic Populations With IGT
Decrease in AIR Necessary to Convert From NGT to IGT
Δ AIR (%)
0
-10
Pima Indian
Latino/Hispanic
White
-8
-20
-18
-30
-32
-40
Insulin
resistance
↑↑↑
AIR=acute insulin response to glucose.
Abdul-Ghani MA, et al. Diabetes Care. 2006;29:1130-1139.
↑↑
↑
Insulin Resistance and -Cell
Dysfunction: Summary
• Individuals with impaired glucose tolerance
– Are maximally or near-maximally insulin resistant
– Have lost ~80% of their -cell function
– Have an incidence of diabetic retinopathy of
~10%
Pathogenesis of Diabetes
Evolving Concepts
Pathogenesis of Type 2 Diabetes
Islet -cell
Impaired
Insulin Secretion
Increased
HGP
HGP=hepatic glucose production.
Decreased Glucose
Uptake
Pathogenesis of Type 2 Diabetes
Islet -cell
Diabetes
Diabetes
Normal glucose tolerance
Insulin Secretion
Normal glucose tolerance
Insulin Secretion
Impaired
Insulin Secretion
1
0
5
1st Phase
0
5
i.v. Glucose
1
0
1
5
2nd Phase
2
0
2
5
3
0
3
5
4
4
5
5
0
5
0
5
Time (minutes)
6
0
6
5
7
0
7
5
8
0
8
5
9
0
9
5
1st Phase
-10 -5 0
Increased
HGP
5
1
0
0
2nd Phase
10 15 20 25 30 35 40 45 50 55 60 65 70 75 80 85 90 95 100
i.v. Glucose
Time (minutes)
Decreased Glucose
Uptake
Adapted from Weyer
C,production.
et al. J Clin Invest. 1999;104:784-789; Ward WK, et al. Diabetes Care. 1984;7:491-502.
HGP=hepatic
glucose
Pathogenesis of Type 2 Diabetes
Islet -cell
Impaired
Insulin Secretion
Increased
HGP
Decreased Glucose
Uptake
Pathogenesis of Type 2 Diabetes
Islet -cell
4.0
Control
T2DM
Basal HGP
(mg/kg• min)
Control
4.0
3.5
r = 0.85
P<0.001
T2DM
3.0
2.0
100
Basal HGP
(mg/kg • min)
2.5
200
FPG (mg/dL)
3.5
Impaired
Insulin Secretion
r=0.85
P<0.001
3.0
300
2.5
2.0
Increased
HGP
100
200
FPG (mg/dL)
DeFronzo RA, et al. Metabolism. 1989;38:387-395.
300
Decreased Glucose
Uptake
Pathogenesis of Type 2 Diabetes
Islet -cell
Impaired
Insulin Secretion
Increased
HGP
Decreased Glucose
Uptake
Pathogenesis of Type 2 Diabetes
Islet -cell
7
12
7
1
2
Impaired
Insulin Secretion
5
5
8
4
3
4
2
P<0.
01
P<0.
05
8
1
4
Leg Glucose Uptake
(mg/kg leg wt per min)
Total Body Glucose Uptake (mg/kg•min)
6
Leg Glucose Uptake
(mg/kg leg wt per min)
Total Body Glucose Uptake
(mg/kg • min)
6
0
0
CON
T2DM
0
4
0
6
0
10
0
14
0
Time (minutes)
3
4
2
P<0.01
180
1
0
Increased
HGP
CON
T2DM
0
40
60
100
0
Decreased Glucose
140Uptake180
Time (minutes)
DeFronzo RA, et al. J Clin Invest. 1979;63:939-946; DeFronzo RA, et al. J Clin Invest. 1985;76:149-155.
The Disharmonious Quartet
Islet -cell
↑ FFA
Increased
Lipolysis
Impaired
Insulin Secretion
Increased
HGP
FFA=free fatty acids.
Decreased Glucose
Uptake
Role of Free Fatty Acids
Hyperglycemia
Increased
Lipolysis
Lipolysis
Plasma FFA
Muscle
FACoA
Glucose Utilization
Liver
FACoA
Gluconeogenesis
HGP
FACoA=FFA-derived long-chain acyl-CoA esters.
Boden G. Proc Assoc Am Physicians. 1999;111:241-248.
Free Fatty Acids Impair -Cell Function
Hyperglycemic Clamp Procedure in NGT
Individuals With Positive Family History of T2DM
100
80
75
60
Δ C-peptide
Concentration
(%)*
40
25
20
P<0.001
P<0.04
0
-20
-40
-35
-60
-60
-80
First Phase
*Percent difference between lipid infusion and saline infusion
in subjects with family history of T2DM.
Kashyap S, et al. Diabetes. 2003;52:2461-2474.
Second Phase
The Quintessential Quintet
Islet -cell
Decreased
Incretin Effect
Impaired
Insulin Secretion
Increased
HGP
Increased
Lipolysis
Decreased Glucose
Uptake
GLP-1 and GIP Responses in
Type 2 Diabetes
Postprandial GLP-1 Levels Are
Decreased in Patients with
IGT and T2DM
NGT
*** **
15
IGT
T2DM
*
100
P<0.01
GIP (pmol/L)
GLP-1 (pmol/L)
20 Meal
GIP Levels Are
Increased in T2DM
**
10
*
5
*
*
80
60
40
20
0
0
0
60
120
Time (min)
180
240
-30 0
60
120
Time (min)
*P<0.05.
GLP-1=glucagon-like peptide-1; GIP=glucose-dependent insulinotropic polypeptide.
Jones IR, et al. Diabetologia. 1989;32:668-677;
Toft-Nielsen MB, et al. J Clin Endocrinol Metab. 2001;86:3717-3723.
180
210
GLP-1, GIP, and Insulin AUC Across the
Spectrum of Glucose Tolerance
8
6
4
2
0
16
3
2
1
0
-1
-2
Controls NGT
IGT
T2DM
· min)
10
P<0.05
4
14
AUC1 GIP (nmol/L
12
· min)
P<0.005
AUC1 GLP-1 (nmol/L
AUC1 Insulin (mU/mL · min)
P<0.00005
10
12
8
6
4
2
0
Controls NGT
Vaag AA, et al. Eur J Endocrinol. 1996;135:425-432.
IGT
T2DM
Controls NGT
IGT
T2DM
The Setaceous Sextet
Islet -cell
Impaired
Insulin Secretion
Decreased
Incretin Effect
Islet a-cell
Increased
Lipolysis
Increased
Glucagon Secretion
Increased
HGP
Decreased Glucose
Uptake
Pancreatic a-Cells and -Cells in
Normal Individuals
Endocrine mass
Role
Mechanism of
action
Metabolic effect
-Cells
a-Cells
~50%
~35%
Produce insulin and amylin
Produce glucagon
Secrete insulin in response to
blood glucose elevations
Secrete glucagon in
response to blood
glucose decreases
Permit glucose uptake by
peripheral tissues
Suppress glucagon and HGP
Stimulate HGP to meet
energy needs between
meals
Cabrera O, et al. PNAS. 2006;103:2334-2339; Cleaver O, et al. In: Joslin’s Diabetes Mellitus.
Lippincott Williams & Wilkins; 2005:21-39.
Area of a-Cells Is Increased in
Type 2 Diabetes
P<0.05
18
15.8
16
14
a-Cell
Islet Area
(%)
12
10
10
8
6
4
2
0
Control
Type 2 Diabetes
(n=10)
(n=15)
Clark A, et al. Diabetes Res. 1988;9:151-159.
Basal Glucagon Levels and Basal Hepatic
Glucose Production in Type 2 Diabetes
250
200
120
P<0.001
P<0.001
150
80
100
40
0
NGT
T2DM
SRIF=somatostatin infusion.
Baron A, et al. Diabetes. 1987;36:274-283.
58%
44%
T2DM
+ SRIF
T2DM
+ SRIF
NGT
T2DM
50
0
Plasma Glucagon (pg/mL)
Basal HGP (mg/m2 • min)
160
Plasma Insulin (mU/L)
120
100
80
60
40
20
0
600
16
14
12
10
8
6
4
2
0
700
Plasma FFA (mol/l)
Plasma Glucagon (mU/L) Plasma Glucose (mmol/L)
Hyperglucagonemia and InsulinMediated Glucose Metabolism
500
400
300
200
100
0
0
24
48 hr
Del Prato S, et al. J Clin Invest. 1987;79:547-556.
600
500
400
300
200
100
0
0
24
48 hr
Inverse Relationship Between Insulin:Glucagon
Ratio and Plasma Glucose in IGT
Glucose Appearance (mmol/5 hr)
100
r=0.72
P<0.0001
90
r=-0.62
P<0.001
80
70
60
50
40
6
8
10
12
Peak Postprandial Plasma
Glucose Level (mmol/L)
14
0
5
10
15
20
Plasma Insulin:Glucagon Ratio
Yellow symbols=NGT; green symbols=IGT; circles=nonobese; squares=obese.
Mitrakou A, et al. N Engl J Med. 1992;326:22-29.
Abnormal Meal-Related Insulin and
Glucagon Dynamics in Type 2 Diabetes
Meal
Glucose
(mg %)
Insulin
(µU/mL)
Glucagon
(pg/mL)
360
330
300
270
240
110
80
Type 2 diabetes (n=12)
Normal subjects (n-=11)
120
90
60
30
0
140
130
120
110
100
90
-60
Delayed/depressed
insulin response
Nonsuppressed glucagon
0
60
120
Time (min)
Müller WA, et al. N Engl J Med. 1970;283:109-115.
180
240
The Septicidal Septet
Islet -cell
Impaired
Insulin Secretion
Decreased
Incretin Effect
Increased
Lipolysis
Islet a-cell
Increased
Glucagon Secretion
Increased
HGP
Increased Glucose
Reabsorption
Decreased Glucose
Uptake
Renal Glucose Reabsorption
in Type 2 Diabetes
• Sodium-glucose cotransporter 2 (SGLT2) plays a role in
renal glucose reabsorption in proximal tubule
• Renal glucose reabsorption is increased in type 2
diabetes
• Selective inhibition of SGLT2 increases urinary glucose
excretion, reducing blood glucose
Wright EM, et al. J Intern Med. 2007;261:32-43.
Renal Handling of Glucose
(180 L/day) (900 mg/L)=162 g/day
Glucose
SGLT2
S1
SGLT1
S3
90%
10%
No Glucose
Increased Glucose Transporter Proteins
and Activity in Type 2 Diabetes
SGLT2
GLUT2
AMG Uptake
8
P<0.05
2000
6
1500
4
1000
P<0.05
2
500
0
0
NGT
T2DM
NGT
T2DM
AMG=methyl-a-D-[U14C]-glucopyranoside; CPM=counts per minute.
Rahmoune H, et al. Diabetes. 2005;54:3427-3434.
NGT
T2DM
CPM
Normalized Glucose
Transporter Levels
P<0.05
The Ominous Octet
Islet -cell
Decreased
Incretin Effect
Impaired
Insulin Secretion
Increased
Lipolysis
Islet a-cell
Increased Glucose
Reabsorption
Increased
Glucagon Secretion
Increased
HGP
Neurotransmitter
Dysfunction
Decreased Glucose
Uptake
Altered Hypothalamic Function in
Response to Glucose Ingestion in
Obese Humans
Time to Max Inhibitory
Response (min)
Magnitude of Inhibitory
Response (%)
Lower Posterior Hypothalamus
Matsuda M, et al. Diabetes. 1999;48:1801-1806.
8
P<0.01
4
0
12
Obese
Lean
P<0.01
8
4
0
Obese
Lean
Treatment of Type 2 Diabetes
1.
Should be based upon known pathogenic
abnormalities, and NOT simply on the reduction in
HbA1c
2.
Will require multiple drugs in combination to correct
multiple pathophysiologic defects
3.
Must be started early in the natural history of T2DM,
if progressive -cell dysfunction is to be prevented
Treatment of Type 2 Diabetes: A Sound
Approach Based Upon Its Pathophysiology
Islet -cell
Impaired
Insulin Secretion
TZDs
GLP-1 analogues
DPP-4 Inhibitors
Sulfonylureas/
Meglitinides
Increased
Lipolysis
TZDs
Metformin
TZDs
TZDs
Metformin
Increased
HGP
DPP-4=dipeptidyl peptidase-4.
Decreased Glucose
Uptake
UKPDS: Effect of Glibenclamide and
Metformin Therapy on HbA1c
Conventional
Glibenclamide
Metformin
9
8
Median
HbA1c
(%)
7
IDF
Treatment
Goal:
<6.5%
6
0
0
3
9
6
Years
UKPDS Group. Lancet. 1998;352:854-865.
12
15
ADOPT: Effect of Glyburide, Metformin,
and Rosiglitazone on HbA1c
Glyburide
Metformin
Rosiglitazone
7.6
-0.42% (P<0.001)
HbA1c
(%)
-0.13% (P=0.002)
7.2
IDF
Treatment
Goal:
<6.5%
6.8
6.4
0
0
1
2
3
Years
Kahn SE, et al. N Engl J Med. 2006;355:2427-2443.
4
5
Unmet Needs in Diabetes Care
Multiple
Defects in Type 2
Diabetes
Weight
Management
Adverse Effects
of Therapy
Type 2
Diabetes
Hyperglycemia
CVD Risk
(Lipid and
Hypertension
Control)
Adapted from © 2005 International Diabetes Center, Minneapolis, MN. All rights reserved.
SGLT2 Inhibition
A Novel Treatment Strategy for
Type 2 Diabetes
Normal Glucose Homeostasis
Pancreas
Fat
Liver
Muscle
5 mmol/L
Fasting
Plasma Glucose
Pathophysiology of Type 2 Diabetes
Islet -cell
Impaired Insulin
Secretion
Insulin
Resistance
10 mmol/L
5 mmol/L
Fasting
Plasma Glucose
Increased
HGP
Rationale for SGLT2 Inhibitors
• Inhibit glucose reabsorption in the renal proximal tubule
• Resultant glucosuria leads to a decline in plasma
glucose and reversal of glucotoxicity
• This therapy is simple and nonspecific
• Even patients with refractory type 2 diabetes are likely to
respond
Pathophysiology of Type 2 Diabetes
Islet -cell
Impaired Insulin
Secretion
Insulin
Resistance
10 mmol/L
Fasting
Plasma Glucose
Increased
HGP
Glucosuria
Pathophysiology of Type 2 Diabetes
Islet -cell
Impaired Insulin
Secretion
Insulin
Resistance
10 mmol/L
Increased
HGP
5 mmol/L
Fasting
Plasma Glucose
Glucosuria
Renal Handling of Glucose
(180 L/day) (900 mg/L)=162 g/day
Glucose
SGLT2
S1
SGLT1
S3
90%
10%
No Glucose
Sodium-Glucose Cotransporters
Site
Sugar specificity
Glucose affinity
Glucose transport
capacity
Role
SGLT1
SGLT2
Intestine, kidney
Kidney
Glucose or galactose
Glucose
High
Low
Km=0.4 mM
Km=2 mM
Low
High
Dietary absorption of
glucose and galactose
Renal glucose
reabsorption
Renal glucose
reabsorption
SGLT2 Mediates Glucose
Reabsorption in the Kidney
Lumen
Blood
K+
Na+
Glucose
S1 Proximal
Tubule
Na+
ATPase
SGLT2
GLUT2
Glucose
Major transporter of glucose in the kidney
• Low affinity, high capacity for glucose
• Nearly exclusively expressed in the kidney
• Responsible for ~90% of renal glucose reabsorption
in the proximal tubule
Hediger MA, Rhoads DB. Physiol. Rev. 1994;74:993-1026.
Renal Glucose Handling
TmG
Splay
Glucose
Reabsorption
and Excretion
Actual Threshold
5
10
Theoretical threshold
15
Plasma Glucose Concentration (mmol/L)
Effect of Phlorizin on Insulin Sensitivity
in Diabetic Rats: Study Design
Rat
Group
I (n=14)
II (n=19)
III
(n=10)
IV (n=4)
•
•
Pancreatectomy /
Diabetic Status
Sham
Control
90%
Diabetes
90%
Diabetes
90%
Diabetes
Phlorizin
Meal Tolerance Test
–
+
–
+
+
+
+/–
10-12 days after
discontinuation of
phlorizin
Phlorizin treatment period: 4-5 weeks
Diet was same for all groups; body weight was similar across groups at end
of study
Rossetti L, et al. J Clin Invest. 1987;79:1510-1515.
Effect of Phlorizin on Fed and Fasting
Plasma Glucose in Diabetic Rats
†
*
20
†
6
15
4
10
2
5
0
0
*P<0.05 vs control and phlorizin. †P<0.001 vs control and phlorizin.
Rossetti L, et al. J Clin Invest. 1987;79:1510-1515.
Fed Glucose (mmol/L)
Fasting Glucose (mmol/L)
8
Insulin-Mediated Glucose Uptake in Diabetic
Rats Following Phlorizin Treatment
40
35
Glucose
Uptake
30
(mg/kg ∙ min)
*
25
20
Control
*P<0.001 vs control and phlorizin.
Rossetti L, et al. J Clin Invest. 1987;79:1510-1515.
Diabetes
*
Diabetes
Diabetes
+ Phlorizin +/- Phlorizin
Mechanism of Action of
SGLT2 Inhibitors
Inhibition of SGLT2
Reversal of glucotoxicity
Insulin sensitivity in muscle
• ↑ GLUT4 translocation
• ↑ Insulin signaling
• Other
Insulin sensitivity in liver
• ↓ Glucose- 6-phosphatase
Gluconeogenesis
• Decreased Cori cycle
• ↓ PEP carboxykinase
-Cell function
Effect of Phlorizin on -Cell Function
in Diabetic Rats: Study Design
Rat Group
I
II
III
•
•
•
Pancreactomy / Diabetic Status
Sham
Control
90%
Diabetes
90%
Diabetes
Phlorizin
–
–
0.4 g/kg/day
Sprague-Dawley male rats weighing 80-100 g
Phlorizin treatment period: 3 weeks
Arginine clamp (2 mM); hyperglycemic clamp (≥5.5 mmol/L)
Rossetti L, et al. J Clin Invest. 1987;80:1037-1044.
Plasma Insulin Response to Glucose
First Phase
Second Phase
6
4
Plasma Insulin
(ng/mL ∙ min / g
Pancreas)
*
2
*
0
Control Diabetes Diabetes
+ Phlorizin
*P<0.001 vs control.
Rossetti L, et al. J Clin Invest. 1987;80:1037-1044.
Control Diabetes
Diabetes
+ Phlorizin
Plasma Glucagon Concentration in Diabetic
Dogs Before and After Phlorizin
Glucose Infusion Rate (mg/kg • min)
2
6
8
12
16
24
0
Glucagon
(pg/mL) -200
-400
Starke A, et al. Proc Natl Acad Sci. 1985;82:1544-1546.
Diabetic
Diabetic +
Phlorizin
Familial Renal Glucosuria: A Genetic Model of
SGLT2 Inhibition
Familial Renal Glucosuria
Presentation
• Glucosuria: 1-170 g/day
• Asymptomatic
Blood
• Normal glucose concentration
• No hypoglycemia or hypovolemia
Kidney / bladder
• No tubular dysfunction
• Normal histology and function
Complications
• No increased incidence of
– Chronic kidney disease
– Diabetes
– Urinary tract infection
Santer R, et al. J Am Soc Nephrol. 2003;14:2873-2882;
Wright EM, et al. J Intern Med. 2007;261:32-43.
Familial Renal Glucosuria
Theoretical
Normal
Observe
d
Type B
Glucose
Reabsorption
Type A
5
10
15
Plasma Glucose Concentration (mmol/L)
Santer R, et al. J Am Soc Nephrol. 2003;14:2873-2882.
Analysis of SGLT2 Gene in Patients
With Renal Glucosuria
•
•
•
23 families analyzed for mutations
In 23 families, 21 different mutations were detected
in SGLT2
Cause of glucosuria in other 2 families remains
unknown
Santer R, et al. J Am Soc Nephrol. 2003;14:2873-2882.
Increased Glucose Transporter Proteins
and Activity in Type 2 Diabetes
SGLT2
GLUT2
AMG Uptake
8
P<0.05
2000
6
1500
4
1000
P<0.05
2
500
0
0
NGT
T2DM
NGT
Rahmoune H, et al. Diabetes. 2005;54:3427-3434.
T2DM
NGT
T2DM
CPM
Normalized Glucose
Transporter Levels
P<0.05
Implications
•
An adaptive response to conserve glucose (ie, for
energy needs) becomes maladaptive in diabetes
•
Moreover, the ability of the diabetic kidney to
conserve glucose may be augmented in absolute
terms by an increase in the renal reabsorption of
glucose
SGLT2 Inhibitors for the Treatment
of Type 2 Diabetes
Effect of SGLT2 Inhibition on
Renal Glucose Handling
TmG
Splay
Glucose
Reabsorption
and Excretion
Actual Threshold
5
10
Theoretical threshold
15
Plasma Glucose Concentration (mmol/L)
Effects of Dapagliflozin on Fasting
Plasma Glucose in ZDF Rats
Vehicle (n=6)
0.01 mg/kg (n=6)
0.1 mg/kg (n=6)
1 mg/kg (n=6)
10 mg/kg (n=6)
400
300
*
*
FPG 200
(mg/dL)
†
*
*
100
† †
0
Baseline
Day 8
Day 15
*P<0.05; †P<0.0001 vs vehicle. ZDF=Zucker diabetic fatty.
Han S, et al. Diabetes. 2008;57:1723-1729; Whaley J, et al. Diabetes. 2007;56(suppl 2). Abstract 0559-P.
Effect of Dapagliflozin on Insulin-Stimulated
Glucose Disposal and Hepatic Glucose
Production in ZDF Rats
4.0
8.0
Glucose Infusion Rate
(mg/kg • min)
6.0
3.0
4.0
2.0
P<0.01
2.0
1.0
0
0
CON
DAPA
CON=controls; DAPA=dapagliflozin.
Han S, et al. Diabetes. 2008;57:1723-1729.
CON
DAPA
Hepatic Glucose Production
(mg/kg • min)
P<0.01
Dapagliflozin-Induced Glucosuria
Reduces HbA1c: A Dose-Ranging Trial
Study design
• 12 week, double-blind, placebo-controlled
– Dapagliflozin: 2.5, 5, 10, 50 mg/day
– Metformin XR: 1500 mg/day
– Placebo
Patients
• 389 drug-naive T2DM patients
• HbA1c >7.0%
Measurements
• FPG, PPG, HbA1c
List JF, et al. Diabetes Care. 2009;32:650-657.
Effect of Dapagliflozin on HbA1c
Baseline HbA1c (%) 7.7
8.0
8.0
7.8
7.9
7.7
DAPA
5
DAPA
10
DAPA
50
PBO
MET
XR
1500
P<0.01
P<0.01
0
DAPA
2.5
-0.2
-0.4
Δ HbA1c
(%) -0.6
-0.8
-1
P<0.01
P<0.01
All comparisons vs placebo; no statistical comparisons with metformin were made.
List JF, et al. Diabetes Care. 2008;2009;32:650-657.
Dapagliflozin: Glucosuric and
Metabolic Effects
Glucosuria
↑ 52-85 g/day
FPG
↓ 16-30 mg/dL
PPG
↓ 23-29 mg/dL
Body weight
↓ 2.2-3.2 kg (↓ 2.5%-3.4%)
Urine volume
↑ 107-470 mL/day
List JF, et al. Diabetes Care. 2009;32:650-657.
Adverse Events With Dapagliflozin
PBO
(n=54)
Met 1500
mg QD
(n=56)
Dapa 2.5
mg QD
(n=59)
Dapa 5
mg QD
(n=58)
Dapa 10
mg QD
(n=47)
Dapa 20
mg QD
(n=59)
Dapa 50
mg QD
(n=56)
Hypoglycemia,
n (%)
2 (4)
5 (9)
4 (7)
6 (10)
3 (6)
4 (7)
4 (7)
UTIs, n (%)
3 (6)
5 (9)
3 (5)
5 (9)
5 (11)
7 (12)
5 (9)
Genital infection,
n (%)
0 (0)
1 (2)
2 (3)
1 (2)
1 (2)
4 (7)
4 (7)
Hypotensive
event, n (%)
1 (2)
2 (4)
0 (0)
0 (0)
0 (0)
0 (0)
1 (2)
UTI=urinary tract infection.
List JF, et al. Diabetes Care. 2009;32:650-657.
Investigational SGLT2 Inhibitors
Agent
Manufacturer
Phase III
Dapagliflozin
AstraZeneca/Bristol-Myers Squibb
Phase II
AVE-2268
sanofi-aventis
BI 10773
Boehringer Ingelheim
JNJ-28431754
Remogliflozin
Sergliflozin
Johnson & Johnson
TS-033
Taisho
YM-543
Astellas/Kotobuki Pharmaceuticals
CSG-452A
Chugai/Roche
SAR-7226
sanofi-aventis
TA-7284
Mitsubishi Tanabe/Johnson & Johnson
Phase I
GSK/Kissei
ISIS 388626 – A Specific SGLT2
Antisense Oligonucleotide
•
Highly specific for the kidney and SGLT2 transporter
•
~80% reduction in SGLT2 mRNA/protein in SpragueDawley rats, ZDF rats, and dogs without any effect on
SGLT1
•
Marked reduction in FPG, PPG, and HbA1c in all three
species
•
No changes in plasma or urine electrolytes
Wancewicz EV, et al. Diabetes. 2008;57(suppl 2). Abstract 334-OR.
Unanswered Questions About
SGLT2 Inhibition
Durability
Safety and
tolerability
Renal
impairment
The efficacy of SGLT2 inhibition may wane once
blood glucose falls into the normal range
The long-term safety of this class remains to be
proven
Risk of nocturia and genitourinary infections may limit
use in some patients
SGLT2 inhibition may not be effective in patients with
renal impairment
SGLT2 Inhibition: Meeting Unmet
Needs in Diabetes Care
Multiple
Corrects
a Novel
Pathophysiologic
Defects
in Type 2
Defect
Diabetes
Weight
Promotes
Management
Weight
Loss
Adverse
Effects
No
Hypoglycemia
of Therapy
Improves
Type 2
Glycemic
Diabetes
Control
CVD Risk in
Improvements
(Lipid
and
Glucose
and
Weight
Support Other
Hypertension
CVDControl)
Interventions
Complements
Action of Other
Hyperglycemia
Antidiabetic
Agents
Conclusions
• SGLT2 inhibition represents a novel approach to the
treatment of type 2 diabetes
• Studies in experimental models of diabetes have
demonstrated that induction of glucosuria reverses
glucotoxicity
– Restores normoglycemia
– Improves -cell function and insulin sensitivity
Conclusions
• Genetic mutations leading to renal glucosuria support the
long-term safety of SGLT2 inhibition in humans
• Early results with dapagliflozin provide proof of concept
of the efficacy of SGLT2 inhibition in reducing both
fasting and postprandial plasma glucose concentrations
in type 2 diabetes
Overall Conclusions
• Understanding of the pathophysiology of type 2 diabetes
is an evolving process
• As new concepts emerge, there is potential for new
treatment modalities
• Optimal management of type 2 diabetes requires a
multifaceted approach that targets multiple defects in
glucose homeostasis