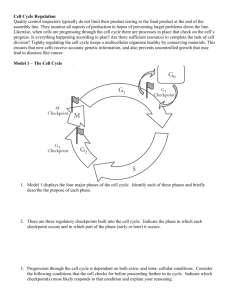
The Cell Cycle
advertisement

The Cell Cycle Jonathon Pines Gurdon Institute j.pines@gurdon.cam.ac.uk http://www.gurdon.cam.ac.uk/~pineslab/New_We b_Site/Site/Lectures.html Outline How do we know there is a cell cycle? How is the cell cycle controlled? What goes wrong in cancer? What does a cell need to do to proliferate? Chromosome Separation M Product - Substrate? Oscillator? S DNA replication How do we know there is a cell cycle? G1 or S or G2 M = + G1 S + + M S S = G2 G2 delay = Rao & Johnson, 1970, Nature 225, 159-164; Johnson & Rao, 1970, Nature 226, 717-722 After Murray and Hunt, 1993 M phase and S phase are different cellular states Problems to be solved: Alternation and Completion Chromosome Separation M Murray and Hunt, 1993 The Cell Cycle: an introduction. Morgan, D.O., 2007 The Cell Cycle: Principles of Control S DNA replication Cyclin-CDK complexes drive the cell cycle • Three converging lines of evidence: Yeast genetics Xenopus meiosis Translational control in sea urchin eggs M ‘M phase’ Cyclin-CDK ‘S phase’ Cyclin-CDK S How can you isolate cell cycle regulators? Schizosaccharomyces pombe (fission yeast): Position in cell cycle related to length of cell Screen for genes that accelerate or slow down the cell cycle cdc2 is a conserved protein kinase required at 2 points in cell cycle and with a wee allele Wee1, mik1 cdc25 cdc2 Interphase cdc13 (Cyclin B) Mitosis After Murray and Hunt, 1993. MPF: a potent trigger for mitosis Xenopus laevis: arrested in G2 of meiosis I, egg arrested in metaphase of meiosis II Factor in egg cytoplasm forces oocyte to enter M phase - M phase promoting factor (MPF) Self amplifies and does not require protein synthesis: pre-MPF in oocyte Universal property of M phase cells Progesterone Inject cytoplasm Masui and Markert, 1971, J. Exp. Zool 177, 129. After Murray and Hunt, 1993. Cyclins: Coincidence or cause? Sea urchins: large eggs arrested in G1 of first mitotic cell cycle Fertilisation causes large increase in translation One protein only translated after fertilisation and destroyed at each mitosis: Cyclin Cyclin MPF S M S M Evans et al. 1993. Cell 33, 389 MPF eventually shown to consist of cyclin and Cdc2 (Dorée and Hunt, 2002, J Cell Sci, 115, 2461-4) Cyclin-CDK complexes define the state of the cell cycle Mitosis Initiation cdc13-cdc2 M Degrade cdc13 or cdc2 G2 G1 S DNA Synthesis START Re-initiate DNA synthesis DNA Synthesis Initiation cig2-cdc2 Hayles et al., 1994, Cell 78, 813 and Broek et al., 1991, Nature 349 , 388. The cell cycle as alternation of CDK activity Low CDK activity: Assemble and fire origins of replication High CDK activity: Repress origins of replication Build mitotic apparatus S. pombe paradigm - the Threshold hypothesis Stern and Nurse, 1996, Trends Genet. 12, p345-350 Norton and Diffley, 2000, Molecular Cell, 5, 85-95 Testing the Cdk threshold hypothesis Cyclin B (Cdc13) Analogue Sensitive Cdc2 Kinase Cyclin-cdc2 fusion drives cell cycle in absence other cyclins Control amount of kinase activity through analogue sensitive Cdc2 Low concentrations of inhibitor block mitosis High concentrations block DNA replication Add different inhibitor concentrations to drive cell cycle with non-degradable Cdc13 Block G2 cells and release into low concentrations of inhibitor - Cells re-replicate Coudreuse & Nurse, 2010 Nature 468, 1074-1079 The Cell Cycle: Alternation Metaphas e Initiation of Mitosis Cyclin B1-CDK1 Cyclin A-CDK1/2 G0 M G2 G1 START or R point Cyclin D-CDK4/6 Completion of DNA Synthesis S Initiation of DNA Synthesis DNA Synthesis Cyclin A-CDK2 Cyclin E-CDK2 Waves of cyclin-CDK kinase activity during the human cell cycle G1 S G2 M G1 Kinase activity Cyclin B-CDK1 Cyclin A-CDK2 Cyclin E-CDK2 Cyclin D-CDK4/6 time M Growth Factors Cdc25 A But Cdk2 is Not Essential for Mitosis Ortega et al., 2003, Nature Genetics, 35, p25-31 Cyclins E and A are partially redundant Cyclin E is only essential for endo-replication Geng et al., 2003 Cell 114, 431-443 Cyclin A is only essential in early embryos & stem cells Kalaszczynska et al., 2009 Cell 138, 352-365 Most mitotic cycles only require one Cdk Santamaria et al., 2007, Nature 448, 811-815 • • Looks like dividing animal cells are just like fission yeast But for cells to differentiate and form particular tissues they need specialised Cdks • • • Cdk2 - spermatogenesis, oogenesis Cdk4 - pancreatic beta cells, pituitary, mammary epithelium Cdk4 or Cdk6 - haematopoesis, cell size Why are there multiple Cyclin-Cdks? • Partial Redundancy? S. cerevisiae paradigm • Tissue specific roles The Cell Cycle: Alternation Metaphas e Initiation of Mitosis Cyclin B1-CDK1 Cyclin A-CDK1/2 G0 M Polo Aurora G2 G1 Cyclin D-CDK4/6 Completion of DNA Synthesis S Initiation of DNA Synthesis DNA Synthesis Cyclin A-CDK1 & 2 Cyclin E-CDK2 S Coordination between mitotic kinases Kinase Domain • PBD PBD Polo box binds to S-pT or S-pS - Elia et al., 2003, Science 299, p1228 • • Often generated by Cdk (S-T-P) Thus Cdk phosphorylation generates Plk substrate Eg: Cdk1 phosphorylates INCENP to recruit Plk1 to kinetochores - Goto et al., 2006, Nature Cell Biology 8, p180 Regulating a CDK Cyclin Proteolysis (Ubiquitin) Cyclin Binding Thr14 & Tyr15 Phosphorylation (wee1/mik1) Thr14 & Tyr15 dephosphorylation (Cdc25) T-loop Thr dephosphorylation (p24 KAP ) T-loop Thr phosphorylation (CAK) Inhibitor binding Inhibitor removal T14 Y15 Deactivate CDK Activate T160 Morgan 1995, Nature 374, 131. Cyclin-CDK inactivation: the paradigm N-term P P ATP P Cyclin Box KINASE Cyclin Fold 2 C-term Cks OFF ON Wee1 - Generating a Robust Switch • Fission yeast wee1 and mik1 prevent premature mitosis • mik1 stabilised by unreplicated DNA •Wee1 in animal cells blocks mitosis in interphase Ultrasensitive response to inhibition by Cdk1 - bistable state SP SP SP SP SP Kinase Domain Sites not well conserved Not inhibitory Conserved inhibitory sites Kim & Ferrel, 2007, Cell 128, 1133-1145 Cyclin-CDK inactivation: the Paradigm 2 Cki N-term ATP P Cyclin Box KINASE Cyclin Fold 2 C-term Cks OFF ON CKIs inhibit both the CDK and the cyclin Russo et al, 1996 Nature 382, 325. Yeast CKI Paradigm: Cell Cycle Co-ordination Sic1 Cln2-Cdc28 Clb5-Cdc28 Cdc34 - SCF p40 Sic1 Cdc28-Clb2 M Sic1 p40 Degradation Cdc28-Clb5/6 G1 S Phase Cdc4 Sic1 Inactivation has Parallels with Wee1 Inactivation Sic1 is processively phosphorylated by Cln2 and Clb5 Clb5 phosphorylation generates a positive feedback loop Kõivomägi et al., 2011, Nature 480, 128–131 Generating a Robust Switch also requires Regulating Antagonistic Phosphatases How to turn off a specific complex? Generating a Robust Switch - Target a Specific Phosphatase Complex with an Inhibitor Mochida et al., 2010 Science 330, p1670 States of the Cell Cycle are generated by Proteolysis Different complement of proteins present in different cell cycle states The Cell Cycle is Co-ordinated by Ubiquitindependent Proteolysis Effectively an interplay between the SCF and the APC/C SCF = Skp1 + Cullin + F-box protein APC/C = Anaphase Promoting Complex/Cyclosome Ubiquitination E1 E1 ATP ADP + Pi Ubq UBC Ubiquitin Ligase E2 E3 E2 E3 Ubq Su b st r at e Ubq DESTRUCTION 2 6 S Prot eoso me Ubq Ubq Schematic of RING E3 ligases COP9/Signalosome UBQ F-Box/ BTB/ SOCS Box Substrat e E2 RING DOC1 Nedd8 Cullin Mammalian cells: F-box proteins 69 F box proteins - bind to Cul 1 BTB domain: proteins Fbw1- bind to CulFbw2 3 Fbw>200 = WD40 = bTrcp1, = bTrcp2, Fbw7 = Cdc4 SOCSrich boxdomain: proteinsFbl1 - bind to Cul2/5 Fbl >50 = leucine = Skp2 DDB1 proteins - bind to Cul 4 Fbx>15 = other domains Nedd8 closes the gap between E2 and substrate Nedd8 Duda et al., 2008 Cell 134, 995-1006 Interplay between the SCF and the APC/C • • SCFSkp2 is degraded by the APC/C Allows p27 (Cdk inhibitor) to accumulate in G1 phase Bashir et al., 2004, Nature 428, 190-193 Ubiquitination: Mitosis Destruction box proteins UBC (UBC10/UBC5 + Ube2S) E2 Cdc20 E2 E3 E3 Cyclin B Securins P APC/C Ubq APC7 APC3 APC10 APC6 APC11 Apc6 APC4 APC2 Su b st rat e Apc3 Ubq APC1 Ubq Ubq Pines, J. 2011. Nature Reviews Molecular and Cell Biology 12, 427-438 APC8 Apc8 APC5 Co-ordinating Mitosis by Proteolysis Antephase Pro-metaphase NEBD Metaphase Chromosome Chromatid Attachment Separation Anaphase Telophase Cytokinesis Spindle Disassembly Spindle checkpoint Cyclin A Securin Cyclin B1 Cdc20 Plk1 UbcH10 Aurora A Pines, J. 2006, Trends Cell Biol., 16, 55-63 APC10 and Cdc20 form the Destruction Box receptor EM reconstructions Da Fonseca et al., 2011, Nature 470, 274-278 APC/C specificity: two co-activators Cdc20 Cdh1 Essential for Mitosis Required for correct G1 phase & to degrade Aurora kinases Regulated by Regulated by spindle checkpoint proteolysis by Cdh1 Proliferating cells phosphorylation (by CDKs) Rca1/Emi1 Somatic & Differentiated cells (brain & trophectoderm) Yu, H. 2007, Mol Cell., 27, 3-16; Garci-Higuera et al., 2008, Nature Cell Biology, 10, 802-811 Floyd et al., 2009 Curr Biol., 18, 1649-1658 Completion: Timing and Checkpoints Chromosome Separation M S DNA replication Completion: Timing Budding yeast mitosis (at least under laboratory conditions) Embryonic cell cycles Xenopus (all you need is cyclin B) Drosophila Problem: High error rate Inflexible Completion: Checkpoints First defined by Weinert and Hartwell, 1989, Science 246, 629. “The events of the cell cycle .. are ordered into dependent pathways in which the initiation of late events depends on the completion of early events. Some dependencies can be relieved by mutation ….suggesting that the dependency is due to a control mechanism and not an intrinsic feature of the events themselves. Control mechanisms enforcing dependency in the cell cycle are here called checkpoints.” Checkpoints: S phase checkpoint M G2 Checkpoints Ds breaks UV Morphogenesis Checkpoint Prophase Checkpoint Spindle Checkpoint S Checkpoints Think about the biology – DNA damage: budding yeast arrest in mitosis fission yeast and animal cells in G2 What should be the phosphorylation state of Cdk1? What should be the targets of the checkpoint? Checkpoints • Budding yeast do use Cdk1 Y18 phosphorylation • Part of the morphogenesis checkpoint to prevent budding in inappropriate conditions • Major regulation is on the stability of Swe1p • Degradation of Swe1 accompanied by relocalisation to bud neck via binding to Hsl7p McMillan et al., 2002, Mol. Biol. Cell., 13, p3560-75 Summary The cell cycle is driven by alternation of high and low Cdk activity Robust switches are driven by ultrasensitivity and phosphatase feedback loops Alternation in Cyclin-Cdk activity is underpinned by proteolysis In somatic cells checkpoints ensure that the switch is not thrown until previous stage is complete (embryos often rely on timing) The Cell Cycle & Development Cells must proliferate only in response to the correct cues Proliferation, growth and differentiation must be coordinated to generate tissues and organs of the correct size and structure To Cycle or Not? The decision to proliferate or differentiate or become quiescent is made between mitosis and DNA replication The Restriction point: the time when cells are committed to replicate their DNA and divide The Restriction Point Committment D-type Cyclins are important signal transducers D-type Cyclins are particularly important in specialised cell types E.g.: Retina, Cyclin D1 binds to notch promoter - Bienvenu et al., 2010 Nature 463, 374 D-type Cyclins are basically there to turn on E-types Rescue Cyclin D1 KO with a knock-in of Cyclin E - Geng et al., 1999 Cell 97. 767-777 Committment Turning on genes required for S phase and mitosis requires de-repressing the E2F-family CKIs are important Regulators of G1 Phase Cyclin-Cdks INK4 family inhibit only D-Cdk4/6 CIP/Kip (p21/p27/p57) inhibit E & A Ubiquitin-mediated proteolysis is important to regulate Ckis and to maintain quiescence Wirth et al., 2004 Genes Dev 18, 88-98 SCFSkp2 is degraded by the APC/C - allows p27 to accumulate in G1 phase Balancing proliferation & differentiation Number of progenitor divisions will determine tissue or organ size E.g.: Neural cortex in Cdh1 heterozygotes Microcephaly (centrosome proteins) - Spindle orientation or cilia signalling? Dysregulation Hit the gas Cut the brakes Loosen the steering Hit the gas The problem of ‘overexpression in cancer’ Some pathways are very frequently perturbed in cancer, e.g.: PI3 Kinase pathway, activates AKT & promotes survival and growth Activated/amplified RTKs & AKT PTEN is a commonly deleted tumour suppressor PI3K PATHWAY Engelman 2009 Nature Reviews Cancer Hit the gas Chromosome rearrangements drive proliferation signals Frequent for the D-type Cyclins (D1 & D2) Parathyroid adenoma Hit the gas •Mantle cell lymphoma: Cyclin D1 next to IgH or Cyclin D2 next to IgK IgK fused to Cyclin D2 Cut the Brakes Rb & the pocket proteins p16 p21 & p27 - Skp2 Senescence and telomerase Loosen the Steering Genomic Instability Cancer Genomes can undergo massive rearrangements - mechanism unclear but could be due to double stranded breaks in mitosis (IR or telomeres) - Stephens et al., 2011 Cell 144, 27-40 Loosen the Steering Genomic Instability DNA replication - Beginning replication too early causes problems E.g.: Too much Cyclin E, eg: Cdc4 mutation Strhmaier et al., 2001, Nature 413, 316-322 Interferes with Pre-RC assembly- EckholmReed, 2004, J. Cell Biol 165, 789-800 Lack of Cdh1 - Garcia-Higuera et al., 2008, Nat Cell Biol 10, 802-811 Loosen the Steering Genomic Instability DNA damage repair pathways ATM p53 or Chk2 - Li-Fraumeni syndrome Loosen the Steering Genomic Instability Chromosome segregation Very few genetic diseases because essential Mosaic variegated aneuploidy - BubR1 - Hanks et al., 2004 Nat Genet 36, 1159-1161 The Spindle Assembly Checkpoint Requires Mad1, Mad2, Bub1, Mad3/BubR1, Bub3, Mps1, kinases STOP GO CDC20 CDC20 + APC/C Checkpoint monitors kinetochore attachment Loosen the Steering The Spindle Assembly Checkpoint detects most errors in chromosome attachment but not merotely Loosen the Steering Genomic Instability: Tetraploidy as a permissive state - Fujiwara et al., 2005, Nature 437, 10431047 Tetraploidy could lead to aneuploidy because: i) Multipolar mitosis (Boveri’s centrosome theory) ii) Increase in improper kinetochore attachments because too many chromsomes in the spindle Loosen the Steering Extra centrosomes increase lagging chromosomes and mis-segregation