Unit 5 Review - RHSChemistry
advertisement

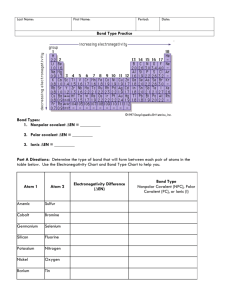
Unit 5 Review Multiple Choice: Circle the correct answer. 1.) In a covalent bond, electrons are _____________________ a.Transferred b. lost c. gained d. shared 2.) Two electrons are shared in a ____________________. a. single covalent bond b. double covalent bond c. triple covalent bond 3.) Another name for covalent compounds is __________________ a. Molecular b. ionic c. metallic d. acidic Ionic 4. Has a high melting point? ___ 5. Balance charges in formula?___ 6. Always solid at room temp? ___ 7. Names use prefixes? ___ Covalent ___ ___ ___ ___ 8. Does not conduct – ever! 9. Conducts in solution? 10. Hard and brittle? 11. Made of ions? Ionic Covalent ___ ___ ___ ___ ___ ___ ___ ___ 12. Has a crystal lattice? 13. Found as molecules? 14. Contains a metal? 15. Contains only nonmetals? ___ ___ ___ ___ ___ ___ ___ ___ Complete the table of covalent compounds. Give names or formulas. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. Disulfur trioxide Carbon monoxide Tetraphosphorus decaoxide silicon pentafluoride phosphourous tetrabromide S203 CO P4O10 SiF5 PBr4 ammonia NH3 Dinitrogen monoxide N2O CuCl2 P308 Copper(II) chloride Triphosphorus octaoxide Disilicon tetrabromide Si2Br4 Cesium oxide Cs2O Calcium phosphate Ca3(PO4)2 Draw the Lewis Structures and name the shape of the following molecules 28. NCl3 29. SF2 30. SO32- 31. SiO2 32. SO3 or or 33. which of the following(28-32) demonstrate(s) resonance? 32 (SO3) #_______________ 34. Define polarity. What are the 2 requirements for a molecule to be polar? The unequal sharing of electrons. The bond must be polar and the molecule. Must be asymmetrical (have unshared e-). 35. Identify the type of bond (ionic, Polar covalent, or nonpolar covalent). Polar covalent H and O: __________________ ionic Na and Cl: _________________ Nonpolar covalent H and C: __________________ 36. Go back to the Lewis Structures you completed in #28-32 of the review. Look at the molecule and the bond type. State whether the following molecules are polar or nonpolar: • • • • • Molecule 28 is ( polar Molecule 29 is ( polar Molecule 30 is ( polar Molecule 31 is ( polar Molecule 32 is ( polar / / / / / nonpolar nonpolar nonpolar nonpolar nonpolar ) ) ) ) )