Solid State Physics (1) , Phys3710 Dr Mazen Alshaaer

Solid State Physics (1) , Phys3710
Dr Mazen Alshaaer
Q1: Determine whether each of the following statements is false (F) or correct (C)?
1.
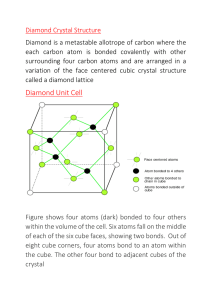
Real Crystals always have foreign atoms (impurities), missing atoms (vacancies), & atoms in between lattice sites (interstitials) ( )
2.
Liquids require more energy to break the interatomic bonds than solids. ( )
3.
Solids, liquids, and gasses are called condensed matter. ( )
4.
Primitive unit cell should include only one atom. ( )
5.
Simple cubic (SC) structure is considered as primitive unit cell. ( )
Q2: In the following multiple-choice questions, please circle the correct answer:
1.
The following Figure shows the 3D arrangement of atoms in a material, this material is: a.
Amorphous b.
Crystalline c.
Polycrystalline
2.
The ionic bond is a dominant bond in the following class of Materials: a.
Polymers b.
Metals c.
Semiconductors d.
Ceramics
3.
Which of the following is not representing a unit cell: a.
a b.
b c.
c d.
d
4.
The following Bravais lattice in 2D is called: a.
Centered rectangular b.
Rectangular c.
General or Oblique d.
Hexagonal
5.
The number of atoms per unit cell of BCC (Body Centered Cube) lattice is?
1
a.
1 b.
2 c.
4 d.
8
6.
Which of the following figures represents S orbital?
7.
The following diagram represents the potential energy curve of two materials. Based on this diagram which of the following is false sentence? a.
Material 2 exhibits lower E Modulus than Material 1. b.
Material 2 exhibits lower melting temperature than Material 1. c.
Material 2 exhibits lower thermal expansion coefficient than
Material 1.
8.
The following figure represents the unit cell in 3D of: a.
Simple cube structure b.
Body centered cube structure c.
Face centered cube structure
9.
Which is the most ionic bond of the following compounds (See the periodic table below): a.
NaCl b.
KCl c.
CsCl d.
H
2
10.
Which is the most covalent bond of the following compounds (See the periodic table
below): a.
HCl b.
CaCl
2 c.
SrCl
2 d.
LiCl
2
Q3: If the following figure represents a 2 dimensional (2D) lattice crystal, answer the following questions :
1.
Draw 2 possible primitive unit cells.
2.
Draw other 2 possible conventional unit cells.
3.
Show 2 possible primitive lattice vectors.
Q4: Here is the energy diagram against the interatomic separation between two atoms.
Where on the x-axis, the Cl atom have the most stable position regarding to the positive
Na atom on point zero?
3