1. Chromatography ppt
advertisement

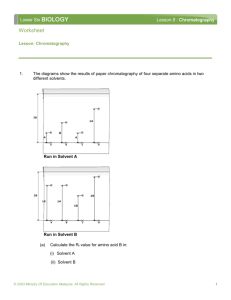
Using chromatography to identify amino acids 12 March 2016 Learning Outcome • explain the principle of chromatography • identify amino acids in a mixture • interpret chromatograms and calculate Rf values from chromatograms. https://youtu.be/J8r8hN05xXk Key words: SOLVENT – a liquid in which chemicals dissolve to make a solution (e.g. water, ethanol) SOLUTE – a substance that is dissolved in a solvent. SOLUTION – a solute dissolved in a solvent AQUEOUS SOLUTIONS – solutions where a solute is dissolved in water NON-AQUEOUS SOLUTIONS – solutions where the solute is dissolved in a solvent that isn’t water (e.g ethanol) Chromatography • A technique used to separate and identify chemicals in a mixture. • Relies on the movement of a gas or liquid through a medium. • The liquid or gas that moves is the mobile phase. • The medium that does not move is the stationary phase Paper Chromatography • The stationary phase is water trapped between the fibres in the paper. • The mobile phase is a solvent • The choice of solvent depends on what is being separated. Thin Layer Chromatography • The stationary phase is a thin coating of absorbent solid on a sheet of plastic or glass. • The mobile phase is a solvent. • TLC is quicker than paper chromatography and can be used for more substances. • Dyes in food products and clothing, drugs, medicines etc. Paper Chromatography clip Filter paper cylinder Solvent Direction of solvent movement Concentrated spot of chemicals to be separated and identified Paper chromatography is a technique used for the separation & identification of relatively small chemical substances by a moving solvent on sheets or strips of filter paper Substances to be identified are “spotted” near one end of the filter paper As the solvent moves up the paper, different molecules move at different rates with the smallest molecules moving the fastest The technique is used for small molecules such as amino acids, small peptides and sugars Developing the Chromatogram • The chromatogram is the paper or thin layer plate. • The solvent front is marked with a pencil line. • Coloured spots are marked in case the colour fades. • Invisible spots can be viewed using a UV lamp and then marked. • Some chemicals need to be developed – sprayed with a chemical that causes a colour change. SIDE VIEW Stationary phase When doing the thin layer chromatography with painkillers, the stationary phase was solid coating on the card. Test sample FRONT VIEW When doing the paper chromatography with food colours, the stationary phase was the water in the paper. SIDE VIEW Sample moves from stationary phase to mobile phase. Mobile phase (e.g water, ethanol) Solvent front – how far the solvent has moved FRONT VIEW SIDE VIEW This substance has moved further because it is more attracted to the mobile phase So, chromatography separates substances in a mixture because they are more or less attracted to the mobile phase. FRONT VIEW Identify Amino Acids In A Mixture Mark the solvent front & allow paper to dry Spray the dry paper with locating agent (ninhydrin) to make the spots visible Solvent front Dry the paper with gentle heat to develop the amino acid spots Purple spots develop located at different distances from the origin line Amino acid spots origin solvent Reference materials • Testing a sample alongside a set of reference samples can be used to identify the chemicals present. • The spots in the sample can be compared with the spots from the known reference samples. Reference samples What does this show about Unknown? -Contains 2 substances (2 spots) -They are E120 and E124 because the spots match the reference samples Retardation Factors (Rf) • A chemical can also be identified by its retardation factor (Rf) • The formula is: Rf = distance moved by sample distance moved by solvent The answer is never greater than 1. Calculate Rf for the three spots. Substance E120 E133 E124 Distance moved by sample (cm) 7.2 4.1 9.5 Distance moved by solvent front (cm) 10 10 10 Rf 0.72 0.41 0.95 The Rf Value Rf distance from origin to solute (spot) distance from origin to solvent front X Rf Y The Rf value is always a value less than one as the solvent front always moves further than the solute molecules Solvent front X3 Y X1 X4 X2 X 5 origin Rf values • Any substance has a particular Rf value when used with a particular mobile and stationary phase. • This can be used to identify substances if you know the Rf values of the reference samples Rf • E.g. A sample moves 3cm, whilst the solvent front moves 12cm. Which chemical is present? Chemical 1 Rf = 0.44 Chemical 2 Rf = 0.12 Chemical 3 Rf = 0.25 Chemical 4 Rf = 0.60 Identifying Unknown Amino Acids Solvent front The mixture of unknown amino acids is seen to contain four different amino acids Of these four amino acids, two can be positively identified The mixture contains four amino acids; two unknown together with arginine & leucine Mixture of unknown amino acids origin met arg leu ala Qualitative Analysis • Paper and TLC are both examples of qualitative analysis. • They don’t show how much chemical is present – instead they just show what is or isn’t present. • TLC gives better results and can be used for a wider range of chemicals. • TLC is quicker, more sensitive and produces a clearer separation. Quantitative Analysis • Paper and TLC can give a rough idea of how much based on how intense the coloured spot is. • Sometimes the spots can be removed and processed to find an amount – giving quantitative data. TWO-WAY PAPER CHROMATOGRAPHY Solvent front Paper dried and rotated clockwise through 90o Solvent front Mixture of amino acids on origin line Second solvent First solvent Two-way chromatography provides better separation of substances that behave in a similar fashion in the first solvent. A second run in a different solvent resolves two very close spots more clearly