Chapter 4 PPT
advertisement

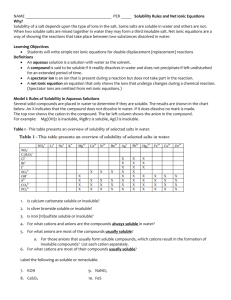
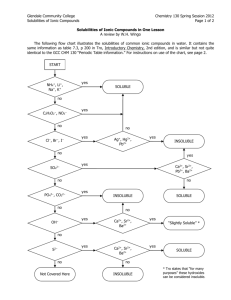
Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 4 Aqueous Reactions and Solution Stoichiometry 4.1 Properties of Aqueous Solutions 1) Solution • Homogeneous mixtures of two or more pure substances (solute(s) dissolved in solvent) – Solvent - substance present in the greatest quantity – Solute(s) - all other substances present in lesser quantities (recall that solutions in which water is the solvent are called aqueous solutions) 2) Water: the Universal Solvent • How is a substance dissolved in water? – Water is known as the “universal solvent” due to the fact that more substances dissolve in water than any other chemical (it has to do with the high polarity of each H2O molecule) • Polarity – physical property of a substance which results from an uneven distribution of positive and negative charge in a molecule or chemical bond • However, not all substances can dissolve in water and those that do dissolve in water dissolve to varying degrees: – Soluble • a substance which dissolves easily and completely in water, forming an aqueous solution – Slightly (or partially) Soluble • a substance which only dissolves a tiny bit in water leaving the majority of the substance behind in its original state of matter – Insoluble • a substance that does not dissolve in water and remains in its original state of matter separate from the H2O molecules (no interaction) Solubility: a visual • The process of solvation: – During the solvation process, the cations and anions that make up a soluble ionic solute become dissociated (or separated) from one another; these ions are then attracted to and surrounded by the water (solvent) molecules (see visual of this process on next slide) NaCl + H2O 1 2 3 4 • These dissociated ions are free to move throughout the solution resulting in electrical conductivity (movement of charged particles through a medium) 3) Determining Solubility of Compounds in Water • To know if an ionic compound is soluble or not, you must consult the solubility rules (see next slide) Solubility Rules for Common Ionic Compounds in Water Soluble Ionic Compounds Nitrates, NO3Acetates, C2H3O2Chlorides, ClBromides, BrIodides, ISulfates, SO42Alkali metal cations Ammonium, NH4+ Exceptions: these are insoluble w/ respective anions none none Ag+, Hg22+ (2 Hg atoms), Pb2+ Ag+, Hg22+, Pb2+ Ag+, Hg22+, Pb2+ Ag+, Hg22+, Pb2+, Sr2+, Ba2+ none none Insoluble Ionic Compounds Sulfides, S2Carbonates, CO32Phosphates, PO43Hydroxides, OH- Exceptions: these are soluble w/ respective anions alkali metal cations, NH4+, Ca2+, Sr2+, Ba2+ alkali metal cations, NH4+ alkali metal cations, NH4+ alkali metal cations, NH4+, Ca2+, Sr2+, Ba2+ Solubility Rules - Practice • Are the following compounds soluble or insoluble in water? – Be(C2H3O2)2 – MgS soluble insoluble – BaSO4 insoluble – K3PO4 soluble • In order to determine if covalent compounds are soluble in water, you must determine the molecular polarity of the compound (this is something you have not yet learned how to do)* • Acids are not explicitly listed in the rules because all the acids that you will deal with are aqueous solutions (and thus soluble) – Substances that produce H+ ions when dissolved in water (Arrhenius) – Ex) HNO3 H+ + NO3– • The only types of bases that you will see at this point will be handled like ionic compounds – Substances that produce OH− ions when dissolved in water (Arrhenius) – Ex) NaOH Na+ + OH– 4) Strong vs. Weak Acids & Bases • In the picture below, which acid is weak and which is strong? • What is different about the contents in each of these beakers? STRONG Acids & Bases • These dissociate completely and exist as 100% ions in solution Beaker (a) contains HCl which dissociates 100% into H+ and Cl– ions Seven Strong Acids MUST BE MEMORIZED! • Hydrochloric (HCl) • Hydrobromic (HBr) • Hydroiodic (HI) • Nitric (HNO3) • Sulfuric (H2SO4) • Chloric (HClO3) • Perchloric (HClO4) When dissolved in water, they dissociate 100% into H+ ions and their respective anion. Strong Bases MUST BE MEMORIZED! Soluble hydroxide compounds • Alkali metal hydroxides • LiOH, NaOH, KOH, etc… • Calcium hydroxide, Ca(OH)2 • Strontium hydroxide, Sr(OH)2 • Barium hydroxide, Ba(OH)2 When dissolved in water, they dissociate 100% into their respective metal cation and OH– WEAK Acids & Bases • These only partially dissociate in solution (only a few ions exist) Beaker (b) contains HNO2 and it appears that only 1/5 of the HNO2 molecules have dissociated into H+ and NO2– 5) Predicting Ions in Solution • If a substance is soluble, then you can predict what types of and how many ions present are in the solution when each compound dissociates – Ex) Ammonium sulfate (NH4)2SO4 soluble Dissociates into: 2 NH4+ ions 1 SO42- ion – Ex) Phosphoric Acid H3PO4 soluble Dissociates into: 3 H+ ions 1 PO43- ion 6) Electrolytes • An electrolyte is a substance which possesses all of the following three qualities: – is soluble water – dissociates into ions when dissolved – AND is a good conductor of electricity • Aqueous solutions can be classified into three categories based on how electrolytic they are: Strong Electrolytes • dissolves readily in water (soluble) • dissociates almost completely into ions (~100%) • good conductor of electricity • Includes: – all soluble ionic compounds – all strong acids & bases Weak Electrolytes • Dissolves in water to a degree (slightly soluble) • dissociate only partially into ions (solution exists mostly as molecules surrounded by water = very, very few ions present in solution) • poor conductor of electricity • Includes: – slightly soluble ionic compounds – weak acids & bases* Nonelectrolytes • does NOT dissociate into any ions (dissolved substance consists of intact molecules surrounded by H2O) • does not conduct electricity • Includes: – all insoluble ionic compounds – most covalent compounds (w/the exception of acids & bases which are either strong or weak) Electrolytes: a visual How to Classify Electrolytic Solutions First determine if the substance is ionic, covalent, or acid/base • If IONIC, determine if it is soluble… – If soluble = strong electrolyte – If insoluble = nonelectrolyte • If COVALENT and not an acid/base… – These do not contain any ions and are therefore considered nonelectrolytes • If ACID/BASE, determine if it is a strong acid/base… – If strong acid or base = strong electrolyte – if it is any other acid or base = weak electrolyte Classify the following as: strong, weak, or nonelectrolyte compound ionic, covalent, acid, or base soluble or insoluble strong, weak, or nonelectrolyte 1 CaCl2 ionic soluble strong 2 HNO3 acid soluble strong 3 Ag3PO4 ionic insoluble nonelectrolyte 4 HCHO2 acid soluble weak 5 KOH ionic & base soluble strong 6 C2H5OH covalent (not a base) unknown* nonelectrolyte 4.2 Precipitation Rxns Precipitation Reactions • Precipitate (ppt.) – an insoluble solid formed from the double replacement reaction of two aqueous solutions – In order to know if a precipitate will form… • Predict the products of the double replacement reaction (write the correct chemical formulas!) • Use the solubility rules to determine if the products are soluble (aq) or insoluble (s) – If there is an insoluble (s) product then a precipitate has formed and you must balance the chemical equation – If all products are aqueous, then no chemical reaction (NR) has occurred DEMO!!! Will precipitates form? • Solutions Used: 0.1 M KI 0.1 M CaCl2 0.1 M Pb(NO3)2 Net Ionic Equations & Spectator Ions • Balanced chemical equations show chemical formulas without considering the ionic character of any of the substances • However… recall that soluble ionic compounds are strong electrolytes • strong acids/bases & soluble ionic compounds • This means they dissociate and are truly present as ions in a sol’n (which leads us to…) • Complete Ionic Equation (CIE) – rewrite a balanced chemical equation as the CIE to show which compounds dissociate into ions (this is true only of soluble, strong electrolytes in the aq state of matter) • Spectator Ions – ions present on both the reactant and product side of a CIE which play no direct role in the chemical reaction • Net Ionic Equation (NIE) – after the CIE is written, cancel out all the spectator ions so that the NIE includes only ions and compounds directly involved in the chemical reaction *When writing CIE and NIE, do NOT separate solid, liquid, or gaseous reactants/products* **When ALL reactants and products are AQUEOUS (no reaction), every ion is a spectator ions (a CIE can be written but the NIE would be NR)** 4.4 OxidationReduction Reactions Single Replacement Rxns • General Rxn Formula: A + BC AC + B • The reactants: – One reactant is a single element by itself – The other reactant is an ionic compound • The products: – If the single element is a metal, it will replace the cation in the ionic compound – If the single element is a halogen gas, it will replace the anion in the ionic compound Oxidation-Reduction Rxns (aka: Redox) • All single replacement rxns can be classified as Redox rxns (but not all redox rxns are single replacement) • Redox rxns occur when electrons are transferred between reactants • Oxidation occurs when an atom, ion, or molecule loses electrons. (LEO) • Reduction occurs when an atom, ion, or molecule gains electrons. (GER) * Both processes happen simultaneously in a rxn Activity Series of Metals - Ease of Oxidation • When a single replacement rxn occurs, the metal that is alone will be oxidized. • However, some metals are easier to oxidize than others (they lose electrons more easily than others, thus forming cations) – Active Metals: • metals at the top of the activity series are more reactive and more easily oxidized – Noble Metals: • metals at the bottom of the activity series which have very low reactivity Activity Series of Metals In a single replacement rxn: ---------------------------------------------------------------------- (1) a solid metal element can be OXIDIZED by any elements listed below it in the activity series (2) Look to see if the metal solid is ABOVE the metal cation (in the 2nd reactant) If yes, the rxn does happen predict products If not, NR (b/c the solid metal cannot be oxidized) Using the Activity Series Examples 1) magnesium sulfate+ zinc metal NR 2) iron metal + copper (II) nitrate Fe (s) + Cu(NO3)2 (aq) Cu (s) + Fe(NO3)2 We can also write CIE & NIE!!! (only aqueous dissociate) CIE: Fe (s) + Cu2+ (aq) + 2 NO3– (aq) Cu (s) + Fe2+ (aq) + 2 NO3– (aq) NIE: Fe (s) + Cu2+ (aq) Cu (s) + Fe2+ (aq) Metals Reacting w/Acids • Only metals listed ABOVE hydrogen in the series are able to react with acids • If the reaction does occur, H2 gas is produced. • Example #1: 2 ___Ni (s) + ___HCl (aq) NiCl2(aq) + H2 (g) Ni is above H2, so the rxn occurs! • Example #2: ___Cu (s) + ___HCl (aq) NR Cu is below H2, so no rxn occurs! Activity Series of Halogens: ACTIVITY SERIES • The idea is the same here… OF HALOGENS: – Fluorine is the most reactive Fluorine – Iodine is the least reactive Chlorine • Only a MORE reactive halogen can Bromine replace a LESS reactive one Iodine • In these single replacement rxns, the single element (halogen gas) must be listed ABOVE the halogen anion in the 2nd reactant for a rxn to occur 2 2 (aq) + ___Br2 (g) • ___Cl2 + ___NaBr (aq) ___NaCl