File
advertisement

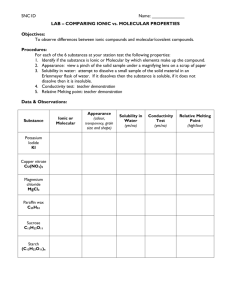
Solutions Trivia Game!!! Gen Chem. Pd. 4 Molecular or Ionic? Making a Solution Hot Hands Bonds & Forces Random! 100 points 100 points 100 points 100 points 100 points 200 points 200 points 200 points 200 points 200 points 300 points 300 points 300 points 300 points 300 points 400 points 400 points 400 points 400 points 400 points 500 points 500 points 500 points 500 points 500 points Molecular or Ionic? - 100 Which of the two categories of elements are present in molecular compounds. Answer Answer: Molecular or Ionic? – 100 Only Non-Metals! Back to the Game! Molecular or Ionic? - 200 Which of the two categories of elements are present in Ionic Compounds? Answer Answer: Molecular or Ionic? – 200 Metals and Non-Metals are both present Back to the Game! Molecular or Ionic? - 300 Is AgNO3 a molecular or ionic compound? Answer Answer: Molecular or Ionic? – 300 Ionic! It contains a metal (Ag) It also contains non-metals (N, O) Back to the Game! Molecular or Ionic? - 400 Is C2H5OH a molecular or ionic compound? Answer Answer: Molecular or Ionic? – 400 Molecular Compound Contains only non-metals (C, H, O Back to the Game! Molecular or Ionic? - 500 Using the Periodic Table, “create” an Ionic Compound Answer Answer: Molecular or Ionic? – 500 Does the compound have both metals and non-metals? Back to the Game! Making a Solution - 100 What vocabulary word do we use to describe the substance that does the dissolving? Answer Answer: Making a Solution – 100 Solvent Back to the Game! Making a Solution - 200 Some chemicals can be dissolved in ethanol (C2H5OH). If they were, what part of the solution would ethanol be? Answer Answer: Making a Solution – 200 Solvent Back to the Game! Making a Solution - 300 What are the 3 steps to making a solution? Answer Answer: Making a Solution – 300 Break apart the Solute Break apart the Solvent Make new bonds between the Solute and Solvent Back to the Game! Making a Solution - 400 Is “breaking apart” an endothermic or exothermic step? Why Answer Answer: Making a Solution – 400 Endothermic. Energy must be added to the chemical to force the particles apart. Back to the Game! Making a Solution - 500 Is making new bonds endothermic or exothermic? Why? Answer Answer: Making a Solution – 500 Exothermic; The energy that was required to hold the particles apart is no longer needed, so it is released. Back to the Game! Using Hot Hands - 100 the data table below, identify which substances were exothermic when they dissolved. How can you tell? Answer Answer: Hot Hands – 100 LiBr, RbF; they caused the water’s temperature to rise. Back to the Game! Hot Hands - 200 Identify which substances are endothermic when dissolving, using the data table. How can you tell? Answer Answer: Hot Hands – 200 NaNO3, KBr; both caused the water’s temperature to drop. Back to the Game! Hot Hands - 300 Which of these 4 substances has particles that are hardest to separate? How can you tell? Answer Answer: Hot Hands – 300 KBr; Most Endothermic per gram Back to the Game! Which Hot Hands - 400 substance releases the most energy, per gram, when dissolving? How can you tell? Answer Answer: Hot Hands – 400 LiBr; Caused the temperature to rise the same amount, but using less chemical. Back to the Game! Hot Hands - 500 Predict, what would happen if we added 5g of NaNO3 AND 5g of RbF to 100.0g of water. Answer Answer: Hot Hands – 500 The final temperature would be around 22.5oC. They would cancel out. Back to the Game! Bonds & Forces - 100 Describe the strength of the bonds in a molecular compound. Do they break in when dissolving? Answer Answer: Bonds & Forces – 100 Strong bonds hold the elements together. They do not break when dissolving, they are too strong. Back to the Game! Bonds & Forces - 200 Describe the strength of the bonds in an ionic compound. Do they break when dissolving? Why/Why Not? Answer Answer: Bonds & Forces – 200 Weak attractive force; They only break if the solute is more attracted to the solvent. Back to the Game! Bonds & Forces - 300 NaCl dissolves in water, but AgCl does not. Why Not? Answer Answer: Bonds & Forces – 300 The attraction between Ag and Cl is stronger than its attraction to water. Back to the Game! Bonds & Forces - 400 Which two elements are connect by a stronger bond/force? How can you tell? Cobalt Flouride (CoF2) or Xenon Tetrafluoride (XeF4) Answer Answer: Bonds & Forces – 400 Xenon Tetrafluoride (XeF4) is a molecular compound. Cobalt Fluoride (CoF2) is an ionic compound Since molecular compounds have strong bonds and ionic compounds do not, the bonds in XeF4 are stronger Back to the Game! Bonds & Forces - 500 Ammonium Nitrate (NH4NO3) is an example of an ionic compound that that is made by combining the two molecular compounds Ammonium (NH4+) and Nitrate (NO3-). Draw a line “cutting” the bonds that would break in water. Answer Answer: Bonds & Forces – 500 Only the Weak Attractive Force between the two ions breaks. Back to the Game! Random! - 100 Where on the periodic table to you find “Metals”? Answer Answer: Random! – 100 All elements to the left of the “Zig-Zag”, except for Hydrogen Back to the Game! Random! - 200 This bank to the hand warmer project. We used 40g of CaCl2 and 100g of H2O to make the hand warmers Predict what would happen is we used only 10 g. Answer Answer: Random! – 200 The hand warmer would have not have gotten as hot (¼ as hot to be exact) Back to the Game! Random! - 300 Make Sasse a detailed observation about Mr. and Make an inference from this observation Answer Answer: Random! – 300 Is your observation a fact, measurement, or something you directly observe with your senses? Is your inference an opinion, explanation, or an interpretation? Back to the Game! Random! - 400 This bank to the hand warmer project. We used 40g of CaCl2 and 100g of H2O to make the hand warmers Predict what would happen is we used 300 g of water instead. Answer Answer: Random! – 400 The hand warmer would have not have gotten as hot (1/3 as hot to be exact) Back to the Game! Random! - 500 If we put too much Kool-Aid powder into water it sinks to the bottom and doesn’t all dissolve. What can we do to the water to make it all dissolve? Why? Answer Answer: Random! – 500 Heat it up! We need energy to break apart the solute. Give it more energy, and more will break up. Back to the Game!