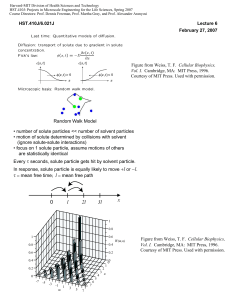
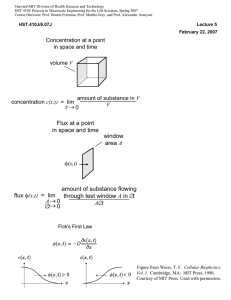
maissasscience
advertisement

The amount of matter in the object How much an object weighs. The space that an object takes up as expressed in cubic units. Anything that has mass and occupies space. Table of elements put in order A positively charged particle in the nucleus of an atom Combined with or containing all the solute that can be dissolved without changing the temperature Having a little solute in a solution in comparison with the amount of solvent. Having a large amount of solute in a solution in a comparison with the amount of solvent The change into a substance into a different substance with different properties Change in some properties of matter with out forming a different kind of matter A formula that describes a chemical reaction The smallest particle of a element that has all the properties of the elements A substance made up of a combination of two or more elements held together by chemical bonds that can not be separated by physical means. The basic building blocks of matter cannot be separated into parts by chemical means A negatively charged particle moving nucleus of am atom A particle with no electric charge in the nucleus of an atom YES I AM DONE WITH MY SCIENCE VOCABULARY!!!!!!!!!!!!!!!!!!!