PowerPoint *********
advertisement

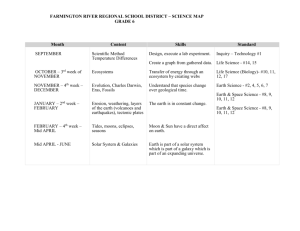
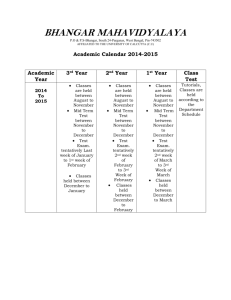
Development of Albumin-bound closoDodecaborate and its Promising Boron Delivery Efficacy to Tumor Daisuke Kanoh,1 Shoji Tachikawa,1 Shinichi Sato,2 Hiroyuki Nakamura*,2 1) Department of Chemistry, Faculty of Science, Gakushuin University, Japan 2) Synthetic Organic Division, Chemical Resources Laboratory Tokyo Institute of Technology, Japan Requirements for Efficient BNCT >25 ppm 10B in Tumor, T/B and T/N >2.5 In the clinical study: L-BPA (500 mg/kg) 1 hr before irradiation ⇒ Total dose: 30 g /person (60 kg body weight) Large amount administration is required! ⇒ Significantly low toxic Different approach is needed in comparison with conventional antitumor drug design How can we deliver a large amount of 10B to tumor? Use of Nanocarriers (Micelles, Liposomes…) Nano Carrier Drug Delivery System (DDS) -Enhanced permeability and retention (EPR) effect → particle size of 100 nm - Escape from Reticulo-endothelial-system (RES) → PEG-conjugation Liposome Tumor tissue Abnormal architectures without tight junction Tumor cells Tumor blood vessel Liposomal Boron Agents for BNCT Boron lipids Na H O H O O 10B Total lipid dose 743 mg/kg Total lipid dose 147 mg/kg Ligand 2013 H. Nakamura, et al Ligand 10 2009Ligand High Boron Content Liposome PEG B K, Tomizawa, H. Matsui et al 2004Anti-EGFR-antibody-Liposome H. Nakamura, et al 2004Boron Lipid Liposome O O Ligand K. Maruyama, et al S 10B O O 2003TF-PEG-Liposome O 2 E. B. Kullberg, et al O 2002EGF-PEG-Liposome R. J. Lee, et al DSBL 1992FA-PEG-Liposome M. F. Hawthorne, et al Boronated DSPC Liposome 1991H. Yanagie, et al Boron conc. in tumor 100 nm Boronated anti-CEA immunoliposome 143 ppm (50 mgB/kg) 82 ppm (30 mgB/kg) 30 ppm (15 mgB/kg) H. Koganei, et. al., Bioconjugate Chem. 2013, 24, 124. Biopolymer-Based Boron Delivery Antibody-conjugates BSD MoAb IB16-6 (B16-BL6 melanoma) 4.8 x 104 ~ 2.5 x 105 / cell ~1100 boron atoms/BSD BSD Cetuximab F98 glioma-bearing rats R. F. Barth, et al, Bioconjugate Chem. 1994, 5, 58-66 Growth factor-conjugates EGF (53 aminoacid residues) BSD BNCT effect BSD-EGF R. F. Barth, et al, Bioconjugate Chem. 2004, 15, 185-194 5 Rolf. F. Barth, et al, Cancer Res. 2002, 62, 3159. Biopolymer-Based Boron Delivery 12,000 17,000 68,000 (8 nm) 29,000 150,000 (15 nm) EPR Effect was observed in the proteins with the MW over 68,000 blood tumor Accumulation of Evans blue-stained albumin complex in tumor skin 6 Albumin as a Biopolymer DDS Vehicle Albumin, a major plasma protein constituent, is composed approximately 55% of the human plasma protein, and has been extensively investigated as a versatile carrier for therapeutic and diagnostic agents. Abraxane® (an albumin-paclitaxel nanoparticle) - FDA approval in 2005 Treatment of Breast cancer, non-small cell lung cancer, stomach cancer, and adenocarcinoma of the pancreas Structure of Abraxane A 130 nm albumin-bound particle form of paclitaxel Uptake of abraxane is presumably mediated by the EPR effect and the gp60 transcytosis. F. Kratz, B. Elsadek, J. Control. Release 161 (2012) 429–445 Maleimide Doxorubicin Conjugate in situ Binding to Albumin + Chemical structure of INNO-206, the (6-maleimidocaproyl)hydrazone derivative of doxorubicin Tumor targeting based on in situ binding to the circulating albumin after intravenous injection. C. Unger, B. Haring, M. Medinger, J. Drevs, S. Steinbild, F. Kratz, K. Mross, Clin. Cancer Res. 13 (16) (2007) 4858. 8 Design of Maleimide Boron Cluster Conjugates 2 - O H O N O N O O 2 N a BSA Maleimide-closo-dodecaborate conjugate(B12H12, MID) (bovine serum albumin) Easy to synthesize and low toxic (COCl)2 68% F(ab’)2 O 2- O N H O O O O N H N O 2 Et3NH antibody Maleimide-decaborate conjugate(B10H10) D.S. Wilbur, M. F. Hawthorne, Bioconjugate Chem. 2007, 18, 1226−1240 9 Synthesis of Maleimide-closo-Dodecaborate Conjugate (MID) - 2- O 2- O N a B F , H C l 4 O N3 O T B A N P h P , H O 3 3 2 1 , 4 d i o x a n e C H C l T H F 2 2 2 TBA TBA 1 2 TBA 87% 2 steps 2 3 quant V. I. Bregadze, Appl.Organometal. Chem., 2007, 21, 98-100 2O O O H O N O N 4 M a l e i m i d o b u t y r i c A c i d , B O P , E t N 3 2- NH2 O T H F 2 TBA 4 2 TBA 77% O 5 (MID-TBA) O H O N O N 1 ) T M A C l / E t O H O 2 ) N a A m b e r l i t e / H O 2 O 2- 2 Na 89% 2 steps MID 10 Cytotoxicity of closo-Dodecaborates MTT assay (CT26 and B16 cell、72h) IC50 (mM) 2SH >1 BSH 2 Na 2O 2 Na >1 4 2O NH2 O O H N O N O 2 Na O >1 MID MID is low toxic! 11 Reaction of MID with Cysteines 2O O O H N N O + HS OH RHN O 2 TMA O 7a: R = H 7b: R = Ac 6 (MID-TMA) HO 2O N S NHR O PBS (pH 7.4) O 2 TMA MID-2TMA ESI-negative MS m/z 205.1 O O O H N 8a: R = H 8b: R = Ac The reaction process was monitored by ESI-TOF MS. The reaction proceeded quantitatively in an hour at rt. 8a m/z 265.6 8b m/z 286.4 12 Conjugation of MID to BSA in vitro antibody* BSH was detected by the anti-BSH antibody in Western blot analysis. The intensity of BSH-conjugated BSA bands increased until the mixture of an 1:1 ratio and then became plateau. * anti-BSH antibody was kindly donated by Prof. Kirihata (Osaka Prefecture University) 13 Structure of BSA Cys34 Total number of Cys: 33 14 Analysis MID-BSA Conjugation by MALDI-TOF-MS BSA was digested with trypsin and the peptide fragments were analyzed by MALDI-TOF-MS. 2- The peptide including Cys34 O O O H N N O 2 TMA O MID: MW 410 +410 MID : BSA = 100 : 1 MID : BSA = 10 : 1 MID : BSA = 1 : 1 BSA only MW 2,835(+48) MW 2,845 We could detect the conjugation of MID and the peptide fragment including Cys34 in BSA. 15 Biodistribution of MID-BSA Conjugates in tumor-bearing mice • Tumor boron conc. reached 60 ppm in the mice treated with MID-BSA conjugates (10:1, 30 mgB/kg) 12 hours after injection from tail vein. • Boron concentration in other organs was low compared to boron liposomes. Tumor 60 ppm 38 ppm CT26-bearing mice (Balb/c/♀; 5 weeks) i.v. 16 Biodistribution of BSH-Boron Lipid Liposomes BSH-Liposome 180 BSH 3000ppm Tumor 10%DSBL 3000ppm 160 10%DSBL 5000ppm 140 Boron (ppm) 120 100 B/P = 2.7 80 10% DSBL 60 30 and 50 mg B/kg 40 20 24 500 Boron (ppm) Blood 400 36 80 BSH 3000ppm 10%DSBL 3000ppm 10%DSBL 5000ppm 60 time (h) 48 Kidney 72 BSH 3000ppm 10%DSBL 3000ppm 10%DSBL 5000ppm 250 Spleen 200 BSH 3000ppm 10%DSBL 3000ppm 10%DSBL 5000ppm 150 300 40 100 200 20 100 0 50 0 24 36time (h)48 72 0 24 36 time (h) 48 72 24 36 time (h) 48 H. Koganei, M. Ueno, S. Tachikawa, L. Tasaki, H. S. Ban, M. Suzuki, K. Shiraishi, K. Kawano, M. Yokoyama, Y. Maitani, K. Ono, H. Nakamura, Bioconjugate Chem. 2013, 24, 124. 72 Biodistribution of MID-BSA Conjugates in tumor-bearing mice • Tumor boron conc. reached 60 ppm in the mice treated with MID-BSA conjugates (10:1, 30 mgB/kg) 12 hours after injection from tail vein. • Boron concentration in other organs was low compared to boron liposomes. • EPR effect was also observed even in the case of the injection of MID itself, suggesting that the conjugation of MID to serum albumin in circulating blood. Tumor 60 ppm 38 ppm CT26-bearing mice (Balb/c/♀; 5 weeks) i.v. 18 MID + blood Control (blood only) MID + blood Control (blood only) Conjugation of MID to Serum Albumin in Blood ~60 kDa (serum albumin) MID was treated with the blood of mice for 1 h at 37oC and the MID-conjugated protein was determined by Western blotting analysis using anti-BSH antibody. The higher accumulation of the MID-BSA (10:1) conjugate is probably due to in situ binding of MID to circulating albumin after intravenous injection. 19 BNCT Effect of Mice Injected with MID-BSA (10:1) Conjugates 1400 MID(10:1) 3000 ppm 30 mg[B]/kg Tumor volume (mm3) 1200 MID(10:1) 1500 ppm 15 mg[B]/kg 1000 MID(10:1) 750 7.5ppm mg[B]/kg Hot Control 800 600 thermal neutron 400 200 0 0 3 6 10 14 17 Neutron flux 1.5-2.2 x 1012 n/cm2 Kyoto University Reactor (KUR) Days after Irradiation Tumor growth of mice injected with MID-BSA conjugates at a dose range of 7.5-30 mg[B]/kg was suppressed after BNCT, and the tumor of some mice completely disappeared 14 days after neutron irradiation. Collaborated with Prof. M. Suzuki’s group at KUR 20 Conclusion 1. We succeeded in the synthesis of maleimide closo-dodecaborate (MID) for conjugation of boron sources to biopolymers. 2. MID is low toxic. 3. MID readily reacted with cysteines including Cys34 in BSA. 4. MID also reacted with serum albumin in blood of mice. 5. Tumor boron conc. reached 60 ppm in the mice treated with MID-BSA (30 mg[B]/kg) conjugates 12 hours after injection. 6. Tumor growth of mice injected with MID-BSA conjugates was suppressed after BNCT. Acknowledgment We thank Professor Mitsunori Kirihata (Osaka Prefecture University) for kindly donating anti-BSH antibody and Stella Pharma Co. Ltd. for the supply of 10B12H12. We also thank Professors Suzuki, Kondo, Sakurai, and Tanaka for their kind supports to proceed thermal neutron irradiation experiments at KUR. 21 Thank you very much for your kind attention! 7F 9F