bond is
advertisement

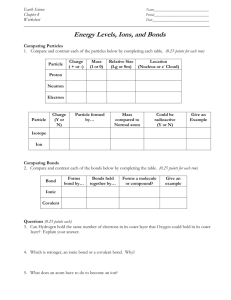
Covalent Compounds and Intermolecular Forces Bonds • Chemical bonds are __________ forces • They act between atoms ________ a molecule Why does bonding occur? • Bonding occurs to maximize stability of the atoms involved. • More stable = LOWER potential energy Bond types • Dependent on the difference between the electronegativities of the elements involved in bond ▫ Electronegativity ______________________________ Highest found in small non-metals ______________ are not ranked Bond Types (cont) • To Determine Type ▫ Subtract the electronegativities ▫ If difference is Zero – bond is ________________ 0.1-1.67- bond is ________________ Greater than 1.67- __________ Bonding is all about the electrons! • Bond type tells us what will happen to the electron(s) • Octet rule will give us an idea of how many electrons will be involved ▫ Have to look at valance electrons ▫ Remember most atoms are stable with 8 ▫ Common exceptions H, He, Li, and Be can be stable with _____ B is stable with ______ Elements with d orbitals (can have more than 8) Valence electrons Covalent bonding • Covalent bonding involves the ___________ of valence electrons. • Atoms can share electrons in order to have full valence shell around them. (usually 8 electrons) Two Types of Covalent Bonds • Non-polar ▫ _________ sharing of electrons ▫ Occurs between the same element bonded together ▫ For example: ______ • Polar ▫ __________ sharing of electrons ▫ Occurs between two different elements bonded together ▫ For example: _______ Polarity of Bonds • Polarity indicates how much electrons are pulled to one end of the molecule or the other. • Creates two ends (or poles) ▫ One end is slightly negative and the other is slightly positive ▫ Creates a ______________ This atom has greater electronegativity. + δ δ Labeling a Polar Bond • Need to draw a vector to show polarity • Vectors ▫ A vector is an arrow that represents the strength of something (like a charge), and the direction in which it is acting. Vectors for Dipoles • The vector points in the direction of the partial ______________ • It has a + on the partial positive end. Vector of dipole + δ δ Polarity • Ionic compounds are very polar. • They have one atom that’s so strong it can pull one electron (or more) away from the other atom. So the electrons are pulled to one end of the molecule. • This is a matter of degrees and there really isn’t a distinct line between polar covalent and ionic. Covalent Compounds • _________ Covalent ▫ Form molecules ▫ Example: ________ • ________ Covalent ▫ Form large interconnected networks ▫ Example: _________ Naming Binary Covalent Compounds • Compound made from two nonmetals (covalent bond) ▫ For example: H2O, CO, NH3, CH4 • No _________ Involved!! • Form ___________ Creating Binary Covalent Compound Formulas • Can only be created from the name or a description of how many atoms of each element there are Creating Binary Covalent Compound Formulas (cont) • For example, ▫ What is the formula for a compound made of 2 boron atoms and three oxygen atoms? _____________ Naming Binary Covalent Compounds • • • • Base name with _____________ First element has element name Second element’s ending is changed to –ide Prefix is put before each element to designate how many atoms there are ▫ Mono- is never put in front of the first element Binary Covalent Compound Prefixes • • • • • • • • • • Mono Di Tri Tetra Penta Hexa Hepta Octa Nona Deca 1 2 3 4 5 6 7 8 9 10 Naming Binary Covalent Compounds (cont) • For example: ▫ ▫ ▫ ▫ CO2 is _______________ BF3 is _______________ B2O3 is _______________ P2O5 is _________________ Representing bonds • There are several ways we represent bonding so that we can have a visual picture of how the molecules look. • The most common is called ▫ ▫ ▫ ▫ Lewis structure Lewis dot diagram Electron dot diagram (they all mean the same thing.) Drawing Lewis Dots • Write the symbol. • Find out how many valence electrons. • Draw that many dots ▫ 2 Dots max / side ▫ Max of 8 dots! Examples of Lewis Dots • Carbon – ______________ C • Nitrogen – ____________ N Lewis Structure for Compounds • Ionic Compounds ▫ Show the Lewis Structure of each ion (including charge) ▫ Sit them next to each other Lewis Structure for Compounds • Covalent Compounds ▫ The central atom is usually the one with the lowest electronegativity (but never ______) ▫ Determine total valence electrons ▫ Move electrons so that each terminal atom has an octet (but not H who gets 2) ▫ Any extra electrons go on the _____________ Covalent bonding •Let’s make carbon tetrachloride, CCl4 •Start with the atoms C Cl Cl Cl Cl Covalent bonding Double and Triple Bonds • Atoms can form single, double or triple bonds with other atoms. • One example – carbon dioxide Structural formula • Electron dot diagrams show all valence electrons as “dots”. • We frequently represent BONDED electrons (shared electrons) as lines. One line is a single bond, two lines is a double bond, three lines is a triple bond. • This is called the structural formula. • _______________ are still dots. Examples Lewis dot diagram Structural formula Cl Cl Cl C Cl Cl Shared pairs are dots Cl - C - Cl Cl Shared pairs are lines, unshared electrons are still dots. Limitations of Lewis diagrams • Work great on paper, but they are drawn in two dimensions. • Molecules exist in ________ dimensions. Electrons repel each other • In order to understand how molecules behave in three dimensions, we need to realize that electrons (or groups of electrons) repel each other. • When do we have groups of electrons on a Lewis diagram? ▫ _______________ ▫ ________________ VSEPR • VSEPR (Valence Shell Electron Pair Repulsion) Theory states that pairs of electrons repel each other. • This allows us to predict the shapes of molecules in three dimensions. Rules for VSEPR • Used for covalently bonded molecules only (all non-metals) • “Electron groups” can be bonds or lone pairs. • Double and triple bonds behave like single bonds (so they’re really 2 or 3 pairs of electrons, but they act like 1 group and we count them as 1 group). Steps for using VSEPR to predict molecular shape (geometry) 1. Draw the Lewis structure. 2. Count the number of electron groups (on the central atom). 3. Look at the chart to determine electron group geometry (first row). 4. Count the number of lone pairs on the central atom (if none, you’re done) and move down the chart to name the molecular geometry. VSEPR Chart No lone pairs 1 lone pair 2 lone pairs 3 lone pairs 4 lone pairs 2 E groups 3 E groups 4 E groups 5 E groups 6 E groups LINEAR TRIGONAL PLANAR TETRAHEDRAL TRIGONAL OCTAHEDRAL BIPYRAMIDAL BENT TRIGONAL PYRAMIDAL SEE-SAW SQUARE PYRAMIDAL BENT T-SHAPED PLANAR SQUARE PLANAR LINEAR T-SHAPED PLANAR LINEAR Linear Trigonal Planer Tetrahedral Let’s try some examples: Draw the Lewis structure for methane, CH4. How many electron groups are on the central atom? (electron groups = bonds + lone pairs) So the electron group geometry is TETRAHEDRAL. Since there are no lone pairs on the central atom, the molecular geometry is the same as the electron group geometry: TETRAHEDRAL. Now make a model of methane. In the Lewis structure, the terminal atoms are 90 degrees apart. In the 3-D model, they’re more like 109.5°. Another example: Draw the Lewis structure for water. How many electron groups on the central atom? Electron group geometry is _______. How many lone pairs on the central atom? Find the 3D model that matches the electron group geometry and pull of an atom for each lone pair. Molecular geometry is ____________. One more example: Draw the Lewis structure of COH2 How many electron groups are on the central atom? Electron group geometry is _______. How many lone pairs are on the central atom? Molecular geometry is ________. Choose the best model to represent this. How does it compare to MY model of this? Does the double bonded atom repel the other terminal atoms differently than a singly bonded atom does? Polar Bond vs Polar Molecule • Polar Bond ▫ Between two different non-metals ▫ Electronegativity difference creates a pull in the electrons to one side of bond ▫ Slightly negative and positive end of bond • Polar Molecule ▫ Must contain at least one polar bond ▫ Polar bonds can cancel each other out ▫ Creates slightly negative and slightly positive end of a molecule Deciding if a Molecule is Polar • Need to draw a vector to show polarity ▫ Vectors of equal strength and in opposite directions cancel out! Molecules with 2 Atoms • Any 2-atom molecule with a polar bond has a dipole moment. • The molecule will be polar Molecules with 3+ atoms. • Vectors are very important for helping us determine the polarity of a molecule that has more than one bond. • In some molecules, the polar bonds will cancel each other out making the molecule non-polar (even though it contains polar bonds) • This is bases on the molecules geometry Bond Energy and Length • The energy required to break a bond is called bond energy. • The distance between the nuclei in a molecule is known as the bond length. • Higher bond energy = lower bond length, in general ▫ Short bonds are harder to break. ▫ Triple bonds are really short ▫ Double bonds are medium length ▫ Single bonds are longer What is IMF? • Force between different molecules BONDS IMF IMF’s • Also referred to as Van Der Waals forces • ________ than bonds • Two main types ▫ London dispersion forces ▫ Dipole-dipole London Dispersion Forces Very Weak Occur in all molecule types Electrons can become temporarily unbalanced Can create a temporary polarity in a non-polar molecule Opposite poles attract London Dispersion Forces (cont) Forces increase ◦ With increasing molar mass of compound For example: C2H6 has a greater force than CH4 ◦ With elongated geometries offering more surface area For example: n-Butane has a greater force than isobutane Dipole-Dipole • Weak • Attraction between opposite poles of polar molecules Hydrogen Bonding • IMF not a bond • Strong (but not as strong as a bond) • Attraction between a H of one molecule and a N, O, or F of another Phases of Matter • IMF’s explain the phases of matter • Solid ▫ Particles move relatively slow (basically vibrating in fixed position) ▫ IMF’s hold them together • Liquid ▫ Particles move more (able to flow past each other) ▫ IMF’s still hold together but not as tightly • Gas ▫ Particle move quickly (each in rapid, random motion) ▫ Little to no IMF’s (we usually assume none) Intermolecular forces are Intermolecular forces are very important. of little significance Intermolecular forces must be considered. Phase Changes • Solid Liquid ▫ Solid Liquid = Melting ▫ Liquid Solid = Freezing • Liquid Gas ▫ Liquid Gas = Evaporation, Vaporization, or Boiling ▫ Gas Liquid = Condensation . • Solid Gas ▫ Solid Gas = Sublimation ▫ Gas Solid = Deposition Evaporation vs Vaporization • Evaporation ▫ Occurs at temperatures below boiling point ▫ Some molecules have enough energy to escape surface of liquid • Vaporization ▫ Occurs at boiling point ▫ Change to gaseous phase occurs throughout liquid Affect of Forces Between Particles • Stronger forces require more energy to break • Substances with stronger forces will phase change at higher temperatures Network Covalent • Very Strong Bonds • Sublime or melt at very high temperatures • Other properties ▫ Very Hard ▫ Do not conduct electricity Another Bond Type • Bond between metals • Called alloys • Have a special kind of bonding, called metallic bonding. Metallic bonding • Electron sea • The atoms in a metal share their valence electrons in a “sea” of electrons. • These electrons can flow from one atom to another freely. • This is why metals are good conductors of electricity. Metallic Bonds • Depend on what metal(s) bond is between • Range from weak to very strong • Melting points range from low to very high • Other properties ▫ Range from soft to hard ▫ Malleable and Ductile ▫ Good conductor of heat and electricity Ionic Bonds • Strong (created by attraction between cations and anions) • Moderate to high melting points • Other Properties ▫ Conduct electricity in liquid or aqueous form ▫ Many soluble in polar substances like water Hydrogen Bonding • Strongest IMF’s • Low to moderate melting points • Other Properties ▫ Soluble in some hydrogen-bonded and some polar solvents Dipole-Dipole • Strong IMF’s • Low to moderate melting points • Other properties ▫ Soluble in some polar and some non-polar solvents Dispersion Forces • Weakest IMF’s • Extremely low to moderate melting points • Other properties ▫ Soft ▫ Soluble in non-polar solvents Other Properties Affected by IMF’s • Surface Tension ▫ “Skin” on surface of liquid ▫ With increasing IMF’s it is harder for molecules to leave surface Strong IMF’s will have lower vapor pressures Low IMF’s lead to high vapor pressures (substance is considered to be volatile) • Viscosity ▫ Resistance of flow in a liquid ▫ Substances are more viscous the stronger their IMF’s