File
advertisement

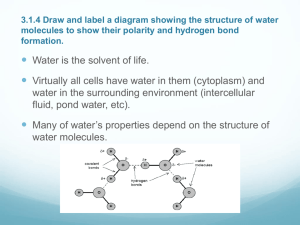
Water: The Chemistry of Living Organisms Water: Not a Universal Solvent, but a Versatile Solvent A. Water is a polar molecule. All of the properties of water stem from this fact. B. This allows water molecules to interact with one another and form up to 4 hydrogen bonds with oxygen atoms of neighboring molecules in liquid water. Water has more structure than most liquids. C) H bonds form, break, and reform in a few trillionths of a second. D) Property that gives water more structure than most liquids = cohesion. E) Water molecules can interact with other polar substances e.g. Ammonia (NH3), ionic compounds. Polar vs. Non-Polar Substances Polar substances are water-loving: hydrophilic Polar substances are attracted to 1 end or the other of water molecule and may form weak hydrogen bonds. Non-polar substances are water-dreading: hydrophobic e.g. increased amounts of covalent bonds between C and H such as lipids. Lipids are fats, oils, steroid hormones, and phospholipids which are the major component of biological membranes. Important Point F. Hydrogen bonds and hydrophobic interactions underlie the 3 properties of water that are biologically important. B. Properties of Water 1. Internal Cohesion: H bonds give cohesion to water. These bonds resist rupturing when placed under tension. This is important at the water-air interface where H bonds exert an inward pull on the uppermost molecules. The result: a) Surface Tension Surface of water – leaves float, insects lay eggs. Surface of alveoli in vertebrate lung is also wet so that gases dissolve in the film; this requires surfactant, a wetting agent. b) Water Transport: movement of water and minerals – soil to leaves Movement is against gravity and relies on 1) cohesion of water molecules 2) adhesion of water to sides of transport vessels (xylem) (think of the meniscus) and 3) evaporation of water into air which provides the necessary pull. 2. Solvent Properties Definition of a solvent: any fluid in which 1 or more substances can be dissolved. Water is a good solvent for ions and polar molecules (solutes). What does dissolve mean? A sphere of hydration forms around individual ions or molecules. E.g. NaCl dissolved in water. Importance: both seawater and living cells contain many different dissolved ions. NaCl dissociates: separation into ions Partial negative charge of oxygen is attracted to the positive sodium ion. Partial positive charge of hydrogen is attracted to the negative chloride ion. Spheres of hydration are formed around these dissociated ions Water Soluble Protein Temperature Stabilizing Effects Think about what it’s like living on the ocean’s coast. The ocean acts as a heat sink=it warms up slowly and cools down slowly. Moderates weather of coastal areas. Prevents severe temperature fluctuations. Temperature Measure of how fast molecules are moving – kinetic energy. As average speed increases, we record as a temperature increase. When water is heated, its molecules cannot move faster until hydrogen bonds among them are broken. This takes considerable heat to break H-bonds and prevent them from reforming. Think about boiling point of alcohol vs. water. High Specific Heat A) Because it takes a relatively large amount of energy to increase or decrease water temperature we say that water has high specific heat. Definition of specific heat: amount of heat that must be absorbed or lost for 1 gram of that substance to change temperature by 1oC. High Specific Heat High specific heat: a measure of the extent to which a substance resists changing temperature. Specific heat of water at 25oC is 1 calorie/gram/degree Specific heat of most carbon compounds = ½ that of water (think melting sugar) Specific heat of iron = 1/9 that of water (think of heating a cast iron skillet) Only ammonia has higher specific heat Aid to biological systems because heat generated by chemical reactions inside cells would destroy cells if not for high specific heat of water within it…coolant b) High heat of vaporization Heat of vaporization = the energy to change 1 gram of liquid water to a gas (586 calories). Evaporation of water from a surface causes cooling of that surface because the fastest moving molecules (i.e. those with the greatest temperature) evaporate and leave the surface cooler. Many organisms get rid of excess body heat by sweating, panting, transpiration = called evaporative cooling. High Specific Heat: Evaporative Cooling c) Behavior of water at low temperatures Cooling water below 4oC (which is waters densest point) causes water to become less dense, turn to ice and float. Water molecules are locked into a crystal-like lattice of hydrogen bonds = ice. Molecules no longer move fast enough to break H bonds. Ice is less dense than water because hydrogen bonds in ice space water molecules far apart. 10% less dense than liquid water. Water at Low Temperatures: Ice Water as Insulation that Provides Habitat Ice insulates large bodies of water and provides habitat for living organisms at low air temperatures. Water: Ice, Water, Steam Water As Habitat C) Wet Chemistry: 1 Mole of Each Most biochemical reactions involve solutes dissolved in water e.g. Digestion, reproduction, circulation, excretion To calculate concentrations of solute – measure substances in moles. Moles 1 mole (mol; M) = molecular weight of a substance in grams H+ = atomic mass = 1 1 mole of hydrogen ions weighs 1 gram 1 mole of any substance is Avocadro’s # or 6.02 x 1023 molecules of that substance Water forms ions spontaneously: ionizes At 25oC/ 1/554 million water molecules ionizes Water Dissociation Pure water and pH scale Pure water has 1/10,000,000 or 10-7 moles of hydrogen ions. Can express hydrogen ion concentration using pH scale 1-14. High hydrogen ion concentration = acid Strong acid: HCl hydrochloric acid Weak acid: H2CO3 Carbonic acid Bases Low hydrogen ion concentration = base Strong base: NaOH sodium hydroxide Weak base: NH3 + H+ yields NH4+ ammonium ion Convenient way to express hydrogen ion concentration is to use pH scale Define pH = -log (H+) where ( ) = concentration. Log of exponent is a whole number pH = exponent x (-1); low number < 7 acidic; > 7 basic pH of water with hydrogen ion concentration of 1 x 10-7 = 7 neutral Logarithmic scale: difference of 1 number = 10 x (H)+ Simple calculations If hydrogen ion concentration is 1 x 10-1 than pH = 1 Or if the hydroxide ion concentration is 1 x 10-13, pH = 1 If hydrogen ion concentration is 1 x 10-2 than pH = 2 Or if the hydroxide ion concentration is 1 x 10-12. pH Scale: Hydrogen ion concentration pH range of living organisms Staying in pH range necessary for enzyme activity. Otherwise the enzyme loses its 3D shape (denaturation). Main buffer in human blood is an acid base pair: carbonic acid and bicarbonate Acid Rain Effects of Acid Rain on Statuary Transparency Last characteristic of water that makes it important to living organisms is transparency. 1) Light can penetrate bodies of water and allows life below water. 2) Light can penetrate cells = photosynthesis.