Periodic Table
advertisement

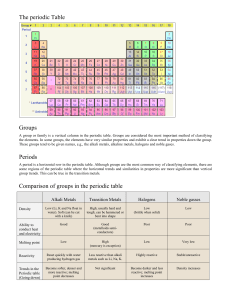
Periodic Table Alabama Course of Study • 3.) Determine the number of protons, neutrons, and electrons, and the mass of an element using the periodic table. • 3.1 Locating metals, nonmetals, metalloids, and noble gases on the periodic table • 3.2 Using data about the number of electrons in the outer shell of an atom to determine its reactivity Mendeleev • Russian chemist • Saw a pattern to elements • Arrange a table in order of increasing atomic mass Shiny Conduct Conduct Malleable Ductile Example / dull Electricity Heat /Brittle Metal Nonmetal Metalloid 3 main categories of Elements • Three main categories of elements are Metals, Nonmetals, and metalloids. – Metals- shiny, and they conduct heat energy and electric current – Nonmetals-do not conduct heat or electric current and are dull in color – Metalloids- have properties of both metals and nonmetals More details • Metals-shiny, good conductors, malleable, and ductile. • Nonmetals-dull, poor conductors, brittle, and unmalleable. • Metalloids- catch-all category or a category that has all the leftovers. General Terms • Periodic Law- repeating chemical and physical properties of elements change periodically with the atomic numbers of the elements • Period- a horizontal row of elements • Group- vertical row of elements Metal, Nonmetals, and Metalloids on a Periodic Table • Metals are to the left of the staircase • They are the elements in tan • Nonmetals are found to the right of the staircase except Hydrogen • They are in the light blue • Metalloids- are along the staircase except Aluminum • They are in the green, teal color Parts of a Periodic Table Hydrogen is in its own group on the chart with one electron Group 1 Alkali Metals are in the yellow color on this table Characteristics-Electrons in the outer level-1 Most Reactive so they must be stored in oil Group 2 Alkaline-Earth Metals are in the light green color Characteristics-Electrons in the outer level-2 Very Reactive but less than alkali Groups 3-12 Transition Metals are in the green color in the center Characteristics-Electrons in the outer level-1 or 2 3rd Most reactive group Group 13 Boron Group is in the teal color on this table Characteristics-Electrons in the outer level-3 4th most reactive Lanthanides- Purple on table/ shiny in color and reactive Actinides- Blue on table/ do not occur in nature and are man-made Group 14 Carbon Group is in the light blue color on this table Characteristics-Electrons in the outer level- 4 5th most reactive Group 15 Nitrogen Group is in the tan color on this table Characteristics-Electrons in the outer level- 5 6th most reactive Group 16 Oxygen Group is in the orange color on this table Characteristics-Electrons in the outer level- 6 7th most reactive Group 17 Halogens are in the Red color on this table Characteristics-Electrons in the outer level-7 2nd least reactive Group 18 Noble gases are in the Pink color on this table Characteristics-Electrons in the outer level- 8 Least reactive Using the info answer the Question • As the number of electrons increase so does the _____________. • As you move from left to right in the groups, the number of outside electrons ____________. (increase or decrease)