Pressure and Volume 2
advertisement

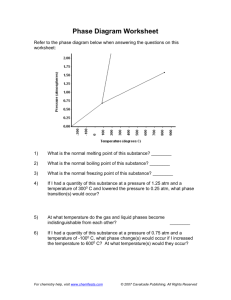
TODAY: FOLDERS: Opening and Closing Exercise New Assignment: Worksheet on Pressure/Volume CALCULATORS HEADPHONES AND ELECTRONICS AWAY UNTIL INSTRUCTED REMINDER: Worksheet on Pressure is due on the 26th, beginning of class. DAILY POINTS • As of today, you will have the opportunity to earn daily points. 5 points a day will be available for meeting classroom expectations: • 1. Phones /headphones/electronics put away when asked. And they stay away. • 2. Not sleeping in class. • 3. Doing the opening/closing exercises and participating in class. • 4. Being on time and ready to work • 5. No side conversations. OPENING EXERCISE • Why does a helium balloon explode when it reaches the upper atmosphere? PREDICTING GAS BEHAVIOR LESSON OBJECTIVES: Describe and apply the relationship between pressure and volume of a gas sample at constant temperature. TODAY’S LESSON OBJECTIVES: Practicing using Boyle’s law to solve problems. CONSTANTS One last piece of Boyles Law. When temperature and moles of gas are constant, the product of the volume and pressure will be constant. You can use this idea to check your answers as you work through the worksheet. P₁ V₁ = P₂ V₂ ↓ = ↓ CONSTANTS We did this problem yesterday: If initially a gas sample occupies a volume of 8.0 mL and exerts a pressure of 1.0 atm, how would the pressure of the gas sample change if its volume were increased to 10.0 mL? 1 atm 8.0 mL= P₂ 10.0 mL CONSTANTS We did this problem yesterday: If initially a gas sample occupies a volume of 8.0 mL and exerts a pressure of 1.0 atm, how would the pressure of the gas sample change if its volume were increased to 10.0 mL? 1 atm 8.0 mL= .8 atm 10.0 mL CONSTANTS When you have found the missing factor, you can re-arrange the formula back to it’s original arrangement. Multiply each side; the products should match. (1 atm) (8.0 mL)= (0.8 atm) (10.0 mL) ↓ = ↓ 8 atm mL 8 atm mL ONE LAST THING… As a note…. Boyle’s law can also be written using all letters instead of numbers. Pᵢ Vᵢ = Pf Vf The “i” stands for “initial” and the “f” stands for “final” WORKSHEETS IN FOLDERS • These are a classroom assignment, so won’t be turned in, but you will earn points CLOSING EXERCISE What would happen to the size of a helium balloon if you took it to the bottom of the ocean?