5.ETC+ATP-synthase
advertisement

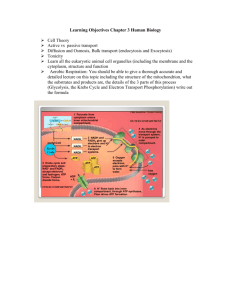
Russian National Research Medical University The respiratory chain: a strategy to recover energy The mitochondrial electron transport chain functioning and control Oxidative phosphorylation Maxim A. Abakumov Moscow, 2014 Energy flow in cell Glicolysis Electron transfer chain Oxidative phosphorilation TCA cycle Outer membrane Inner membrane Intermembrane space Matrix Cristae TCA total energy outcome • Acetyl-CoA + 3 NAD+ + Q(FAD) + GDP + Pi +2 H20 HS-CoA + 3NADH + QH2(FADH2)+ GTP + 2 CO2 + 2 H+ • • • • • Isocitrate Dehydrogenase α-ketoglutarate Dehydrogenase Succinyl-CoA Synthetase Sunccinate Dehydrogenase Malate Dehydrogenase 1 NADH=2.5 ATP 1 NADH=2.5 ATP 1 GTP=1 ATP 1 QH2=1.5 ATP 1 NADH=2.5 ATP • NADH and FADH2is not an ATP (no cash energy) – further conversion is needed NADH and NAD+ • • • • • NAD – Nicotine Adenine Dinucleotide Produced from niacine (vitamine B3) NADH – reduced form NADH – high energy (HE) electron carrier NAD+ – oxidized form • NAD+ + H+ + 2e- → NADH + H+ NADH and NAD+ O H NH2 O HO P O O N HO P O Reduction + H+ + 2eOH OH NH2 N HO P O N O NH2 O + O H H Oxidation O N O OH OH NH2 N HO P O N O O N O OH OH N N O OH OH N FADH2 and FAD • • • • • FAD – Flavine Adenine Dinucleotide Techically not a dinucleotide FADH2 – reduced form (oxidizer) FADH2 – high energy (HE) electron carrier FAD – oxidized form (reductor) • FAD + 2H+ + 2e- → FADH2 FADH2 and FAD O H3C N H3C N NH Flavin Ribitol N H H HO H HO H HO H O NH2 N O O N O P O P O O N - O OH OH OH ADP N FADH2 and FAD FAD FADH2 O H3C N H3C N H3C NH N H H HO H HO H HO H H N H3C O N H NH2 N O O O P O P O - O N N O OH OH OH N O NH N H O Electron transfer chain • Electron transfer chain (ETC) – group of enzymes located on inner membrane of mitochondria • Convert energy from NADH/FADH2 electrons into energy of proton gradient on inner membrane • Proton gradient is needed for further ADP to ATP conversion • Consists of four complexes Oxidative phosphorilation • ADP + Pi→ ATP • Reaction is catalized by ATP-synthase. • ATP-synthase is intramembrane multisubunit enzyme (nanorotor) • ATP-synthase uses energy of proton gradient Electron transfer chain • Consists of four complexes • A lipid soluble coenzyme (UQ, CoQ) and a water soluble protein (cyt c) shuttle between protein complexes • Fe-S, Hem, FMN, Cu atoms acts as a cofactors in different complexes • Electrons generally fall in energy through the chain - from complexes I and II to complex IV Electron transfer chain Coenzyme Q10 (CoQ) • Lipid soluble molecule • Transfer e- from complex I and II to complex III through membrane FMN • FMN –Flavin Mono Nucleotide • Accepts e- from NADH and transfers them to QH2 Complex I • NADH-CoQ Reductase. • Electron transfer from NADH to CoQ • More than 30 protein subunits - mass of 850 kD • Transfers 4H+ Complex I NADH-CoQ Reductase NADH + H+ NAD+ FMN FMNH2 Fe2+S Fe3+S CoQ CoQH2 Complex I - NADH-CoQ Reductase. Complex II • Succinate-CoQ Reductase • Succinate dehydrogenase (from TCA cycle!) • Membrane bound enzyme • Accepts e- from succinate and pass through FADH2 and Fe-S clusters to CoQ • Not a proton pump Complex II Complex III CoQ-Cytochrome c Reductase • CoQ in membrane passes electrons to cyt c in a unique redox cycle known as the Q cycle • 4 H+ are released into intermembrane space • The principal transmembrane protein in complex III is the b cytochrome - with hemes bL and bH • Cytochromes, like Fe in Fe-S clusters, are oneelectron transfer agents Complex III • UQH2 is lipid soluble e- carrier • Cyt c is water soluble e- carrier Complex IV • Cytochrome c oxydase • Electrons from cyt c are used in a four-electron reduction of O2 to produce 2H2O • Oxygen is the terminal acceptor of electrons in the electron transport pathway • Cytochrome c oxidase utilizes 2 hemes and 2 copper sites • Complex IV also transports 2H+ Complex IV ETC ETC summary • 1 NADH gives energy for 10 H+ transport into intramembrane space • 1 FADH2 gives gives energy for 6 H+ transport into intramembrane space • How proton gradient energy is converted into ATP energy? ETC summary • http://www.learnerstv.com/animation/animat ion.php?ani=177&cat=biology How is H+ gradient energy used for ATP synthesis? How is H+ gradient energy used for ATP synthesis? • ATP synthase – nanoscale rotating motor • H+ movement through the channel in ATP synthase leads to ATP synthesis • 3H+ leads to one full circle • 3H+ reguired for 1 ATP synthesis ATP-synthase ATP-synthase ATP-synthase • ATP is synthesized in mitochondria matrix • It must be transported to cytosol • 1H+ is spend for each ATP transfer • 3H+ is spend for each ATP synthesis • Total 4H+ is spend for 1 ATP synthesis • ATP-ADP Translocase ATP-ADP Translocase P/O ratio • How many ATP is made per 2e- • 1 ATP – 4H+ • NADH – 10H+ • P/ONADH = 10/4 = 2,5 • FADH2 = 6H+ • P/OFAD = 6/4 = 1,5 ETC and OP inhibitors • Rotenone inhibits Complex I • Cyanide, azide and CO inhibit Complex IV, binding tightly to the ferric form (Fe3+) • Oligomycin and DCCD (Dicyclohexyl carbodiimide) are ATP synthase inhibitors ETC and OP inhibitors • Rotenone -inhibits the transfer of electrons from iron-sulfur centers in complex I to ubiquinone ETC and OP inhibitors • Oligomycin –blocks proton channel on ATPsynthase (Fo subunit) + e /H transfer uncoupling • Uncoupling of e-/H+ leads to no proton gradient • No ATP synthesis • Affected by lipid soluble H+ acceptors, such as dinitrophenol NADH mitochondria/cytosol transport • Most NADH used in electron transport is cytosolic and NADH doesn't cross the inner mitochondrial membrane • Malate-aspartate shuttle system for NADH transport without actuall NADH transfer NADH mitochondria/cytosol transport Free radicals • Contain unpaired electron on valent level • Highly reactive • Reactive Oxygen Species (ROS) • Reactive Nitrogen Species (RNS) Reactive oxygen species Reactive nitrogen species Lipid peroxides Phagosome NADPH oxidase Antioxidants http://sbxsupplements.com/blog/Complementary-Treatment-ofSchizophrenia-with-Antioxidants Antioxidants Enzymatic Superoxide dismutase Catalase Glutathione peroxidase Glutathione reductase Non-enzymatic Vitamins C and E Carotenoids Flavonoids Minerals Manganese Copper Zinc Selenium Antioxidant enzymes Superoxide Dismutase 2O2- + 2H+ O2 +H2O2 + 2H2O Catalase 2H2O2 O2 + 2H2O Antioxidant enzymes • Glutathione (GSH) – tripeptide 2x GSH-reduced form GSSG-oxidized form Antioxidant enzymes Antioxidants Enzymatic Superoxide dismutase Catalase Glutathione peroxidase Glutathione reductase Non-enzymatic Vitamins C and E Carotenoids Flavonoids Minerals Manganese Copper Zinc Selenium Vitamin E CH3 α-Tocopherol (vitamin E) HO CH3 H3C O CH3 CH3 CH3 CH3 CH3 Free radicals CH3 O CH3 H3C O CH3 CH3 CH3 CH3 CH3 Tocopheroxyl free radical (Oxidized vitamin E) Cell oxidative status