here - IMSS Biology 2014
advertisement

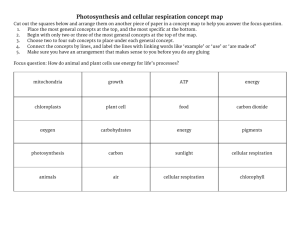
EVOLUTION OF METABOLISM: CELLULAR RESPIRATION IMSS BIOLOGY ~ SUMMER 2012 LEARNING TARGETS • To understand the evidence for the evolutionary origin of cellular respiration. • To understand the impact of oxygen on the evolution of multicellular life on Earth. • To understand the processes by which cellular respiration harvests chemical energy from food and converts it into ATP that fuels cellular work. • To understand the distinctions between aerobic & anaerobic respiration • To understand the role respiration plays on a cellular, organismal, & ecological scales. FOLLOW UP: PHOTOSYNTHESIS Q’S • All plants and most other photosynthetic organisms contain chlorophyll (primary pigment is chl a), but • At least one group of bacteria (halobacteria) rely solely on bacteriorhodopsin (derived from carotene) for photosynthesis THEORY OF ENDOSYMBIOSIS • First proposed by Lynn Margulis in 1960s • Much evidence to support eukaryotic cellular respiration originated via endosymbiosis of aerobic purple bacteria (alphaproteobacteria) which ultimately became mitochondria. • Note: this drawing shows mitochondria endosymbiosis in BOTH animal and plant cells. Both figures depict endosymbiotic origin of mitochondria. Which important details are portrayed in the one above that are not below (and vice versa)? SUPPORTING EVIDENCE FOR ENDOSYMBIOSIS • Mitochondria have own genome (mtDNA) and are selfreplicating (divide independently of cell they live in) • Genomes are much reduced from alpha-proteobacteria (purple aerobic bacteria) ancestors • mtDNA, mtRNA, ribosomes, and mechanisms of protein synthesis and oxidative metabolism all similar to that of proteobacteria • Mitochondria have double phospholipid bilayers • Evidence supports mitochondria arose ca. 2 bya in a common ancestor of all extant eukaryotes • mtDNA inherited by one’s mother (maternal lineage) Human mtDNA contains 37 genes which code for proteins needed for normal mitochondrial function, e.g. 13 genes that code for enzymes involved in oxidative phosphorylation. MITOCHONDRIA FUNCTIONS – BEYOND OXIDATIVE METABOLISM • Oxidative phosphorylation to make ATP • Greatly increases capacities to make ATP! • Many other cellular activities • Assist in regulation of apoptosis (timed cell death) • Genes code for proteins involved in synthesis of heme and cholesterol • Help keep intracellular levels of Ca+2 low inside neurons – important in cell signaling pathways Nuclear DNA is inherited from all ancestors. Mitochondrial DNA is inherited from a single maternal lineage. The Hunt for mtDNA 15 min. ATMOSPHERIC O2 THROUGH EARTH’S HISTORY • By 1.5 bya, photosynthesis-derived O2 accumulated to significant levels in atmosphere; this in conjunction with mitochondria Cambrian Explosion during which metazoan radiation occurred LIFE IN AN OXYGENATED WORLD • Oxygen is highly reactive and potentially toxic – production of reactive oxygen species (radicals), or ROS causes damage to cell membranes, DNA, RNA, proteins (including enzymes and their cofactors) • Aging = accumulated cellular damage caused by ROS EVOLUTION OF MULTICELLULARITY – FROM A METABOLIC PERSPECTIVE • Chloroplasts & mitochondria meet nrg demands of cell via their electron transport systems which generate ROS oxidative stress and DNA damage in organelles • In unicellular organisms, DNA repair is only way to maintain pristine chromosomal DNA in organelles, but DNA repair is limited. • Better strategy: avoid DNA damage by having separate germ and somatic cell lines • Germ (reproductive) cells: metabolically quiet early in development germ line DNA protected from oxidative damage • Somatic cells: must be metabolically active as responsive to rapid change • Bendich, A.J. 2010.http://www.biology-direct.com/content/5/1/42 ENERGETICS OF GENOME COMPLEXITY • All complex life composed of eukaryotic cells. • Eukaryotic cells arose from prokaryotes just once in 4 billion years. Why haven’t prokaryotes evolved greater complexity? • Lane and Martin (2010) propose that prokaryotic genome constrained by energetics • Endoysymbiosis of mitochondria opened door for 200fold expansion in number of genes that can be expressed. • 75% of cell’s total nrg budget allocated to protein synthesis. • http://blogs.discovermagazine.com/notrocketscience/2010/10/20/the-origin-of-complexlife-%E2%80%93-it-was-all-about-energy RESOURCES • Big themes in evolution of photosynthesis and respiration http://www.shmoop.com/cell-respiration/evolution.html • Origins of development of eukaryotic organisms – mtDNA and cpDNA http://www.biology-direct.com/content/5/1/42 • Evolution 101http://evolution.berkeley.edu/evolibrary/news/071101_geneal ogy INTRODUCTION TO RESPIRATION • Via the circulatory system, O2 is delivered to & CO2 is taken away from tissues • Recall that most metabolic processes of the body depend on ATP • What is the function of O2 in cellular respiration? RESPIRATION • Involves ALL processes that deliver O2 from environment to the tissues (cells), including • breathing • gas exhange between air & blood and between blood & tissues • transport of respiratory gases by blood • use of O2 in cellular respiration (aka “internal respiration”) CELLULAR RESPIRATION - OVERVIEW • occurs primarily in mitochondria • harvests nrg stored in organic “fuel” molecules, e.g. glucose (C6H12O6) by enzymatically breaking these molecules down to release their potential nrg • traps and stores this potential nrg in a form that is usable by the cell – ATP! • uses O2 • produces waste products, CO2 & H2O (used in photosynthesis) • Alternative mode: at ocean’s bottom and other anoxic environments, anaerobic organisms synthesize ATP using nitrate or sulfur rather than oxygen CELLULAR RESPIRATION - OVERVIEW • The cells of aerobic organisms (e.g. plant & animals) perform cellular respiration • Cellular respiration is the primary way chemical nrg is harvested from food and converted into ATP ATP = adenosine triphosphate “Currency” of cellular work To get nrg to do cellular work, cells must break down energycontaining substances to release their potential nrg stored as ATP that can be later used by cell to fuel an endergonic reaction ATPase is enzyme that hydrolyzes this terminal phosphate bond http://faculty.ccbcmd.edu/biotutorials/energy/ atp.html#atp GETTING THE PERSPECTIVE • A HUGE amount of ATP is needed to fuel all the cellular activities of an organism! • Example •Average human at rest uses ~45 kg (99 lbs.) of ATP per day (but has surplus of < 1 g of ATP at any given moment) •Estimated that each cell generates & consumes ~10 x 106 molecules of ATP per sec! • So, ATP production must be an on-going process in order to remain alive • Evidence of cellular respiration indicates life!!! Is it Alive? 30 min. Oxidation Glucose loses electrons (and hydrogen) C6H12O6 Glucose 6 O2 Oxygen 6 CO2 Carbon dioxide 6 H2O Water Reduction Oxygen gains electrons (and hydrogen) • Most common fuel molecule for cell. resp. is glucose (C6H12O6) • Overall redox reaction: glu oxidized (loses electrons) while O2 reduced (gains electrons) ROLE OF OXYGEN IN CELLULAR RESPIRATION • Cellular respiration can produce up to 38 ATP molecules for each glucose molecule consumed • During cell. resp., hydrogen (H) & its bonding electrons change “partners” •H & electrons go from glu O2, forming H2O (& ATP) •This H transfer is why O2 is so vital to cellular respiration CRITICAL REDOX REACTION • When hydrogen and water bind to form water, a “burst” of nrg is released • This nrg released as electrons of hydrogen “fall” into their new bonds with oxygen • But, the process needs to be stepped down for a cell to capture this nrg and use it for cellular work. ROLE OF ELECTRON TRANSFER • Cellular respiration can be considered a controlled fall of electrons that releases nrg in a stepwise way, like walking down a staircase • 1st step: transfer of electrons from glucose to NAD+ (electron acceptor) which reduces it to NADH • Rest of path: electron transport chain • Involves series of redox reactions • Leads to production of lots of ATP UNPACKING CELLULAR RESPIRATION • The chemical reactions involved in cellular respiration are grouped into three main stages • Glycolysis • Citric acid cycle • Electron transport chain THE BIG PICTURE • Electron transport chain functions like a chemical machine • Uses nrg released by “fall” of electrons (“pulled” by O2) to pump hydrogen ions (H+) across inner mitochondrial membrane H+ more concentrated on one side of membrane • This H+ can then rush “downhill” thru membrane protein that “spins” the turbine to activate the enzyme, ATP synthase Fig. 6.11 • Cyanide is a deadly poison, because it • Binds to a protein complex in the chain and prevents passage of electrons to O2 • Stops ATP synthesis SUMMARY OF ATP YIELD DURING CELLULAR RESPIRATION MONOMERS FROM FOOD MACROMOLECULES SERVE AS FUEL FOR CELLULAR RESPIRATION ANAEROBIC RESPIRATION • Some cells can actually work for (short) periods without O2, i.e., anaerobically • Sometimes referred to as fermentation • Involves glycolysis lactic acid • Without O2, electrons “dumped” from NADH onto pyruvic acid in order to regenerate NAD+, so lactic acid is formed CELLULAR RESPIRATION IN YEAST • Yeast (the living organisms in soil sample C of the “It’s Alive” activity) are facultative aerobes. • Undergo aerobic respiration when O2 is present – as was the case during the activity – sugar molecules, CO2 (what caused the bubbling in the sample), water, and a high yield of ATP C6H12O6 + 6O2 6CO2 + 6H2O + ATP (hi yield) • Undergo alcoholic fermentation when O2 is absent – sugar molecules broken down into ethanol, CO2 and a low yield of ATP C6H12O6 6C2H5OH + 2CO2 + ATP (low yield) Nrg flow & chemical cycling in ecosystems FIG. 6.2: NRG FLOW & CHEMICAL CYCLING IN ECOSYSTEMS • Misconception: • Plants only perform photosynthesis • Plants perform BOTH photosynthesis and cellular respiration • Plants must be able to harvest nrg (synthesize ATP) from fuel molecules (e.g. glu) in order to grow & reproduce just as animals do • Animals perform cellular respiration only