Ch. 2 Tissue Healing
advertisement

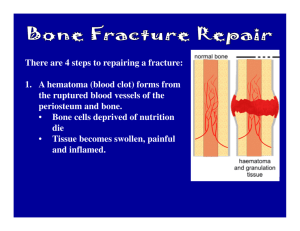
Ch. 2 Tissue Healing Introduction • Tissue healing occurs and is influenced by several factors • There are three phases: – Inflammatory response – Repair/regeneration – remodeling Inflammatory Response • Cells remove debris • Cells create groundwork for repair and regeneration phase Repair/Regeneration Phase • Cells restore the vascular and structural integrity of injured structures Remodeling Phase • Healed tissue adapts to functional loading • Gets stronger based on the stress that is applied to it • The three phases overlap one another • Disruption of any one phase can cause an unsatisfactory outcome Types of Tissue • • • • Epithelial tissue Connective Muscle nervous Epithelial Tissue • • • • Composed of layers of cells Protect organs Secretions (glands) Absorption ( lining of the stomach ) Connective Tissue • • • • • • • Most abundant and variable tissue in the body Attaching organs Support/structure Movement Physical protection Immune response, energy & mineral storage Heat generation and transportation (blood, etc.) Connective Tissue • Subtypes: – Fluid connective tissue=blood and lymph – Fibroconnective tissue=fat, tendons and ligaments – Supportive connective tissue=cartilage and bone Nervous Tissue • Located in brain and spinal cord and nerves • Electrically exciteable • Used to transmit and respond to various impulses and forms of information Muscle Tissue • Skeletal- striated or voluntary • Cardiac – heart muscle • Smooth- linings of organs and blood vessels Common Injuries Puncture and Laceration Incision Acute vs. Chronic • Acute – single forcefull event (ACL tear) • Chronic – occur over a period of time (Tennis elbow) Acute Injuries • Contusions – bruises • Strains – muscle or tendon injuries • Sprains – ligamentous injuries Contusions • Caused by compressive forces • or direct impacts and are graded by degrees of trauma • 1st degree-minimal swelling, no limits • 2nd – pain, bleeding, moderate functional limits • 3rd-hematoma formation,sever limitations Sprains and Strains Fracture Classifications • • • • • • • • • Greenstick Transverse Oblique Spiral Comminuted Avulsion Impacted Depressed Epiphyseal (Salter-Harris classification) Peripheral Nerve Injuries • Neuropraxia-least severe, transient and reversible loss of nerve function • Axontomesis- partial disruption, may cause atrophy or weakness for 2 – 52 weeks • Neurotomesis – complete severance of a nerve resulting in permanent loss of function Chronic Injuries • Blisters – continuous friction • Repetitive overload – tendinosis, tenosynovitis • Chronic irritation –could cause neuralgia or neuroma Soft Tissue Healing • Phase 1: Inflammatory Response • Phase 2: Repair/Regeneration • Phase 3: Remodeling/Maturation Phase 1 • Signs and symptoms: – Redness – Heat – Pain – Swelling – Loss of function Phase 1 con’t • • • • Lasts 7-10 days Initial damage is called “the primary injury” Caused by a release of proteins at the injury site Mast cells produce chemicals (histamines) which promote vasodilation • This causes the redness and heat • Vessels become more permeable which contributes to the swelling Phase 1 • Mast cells also release chemicals that attract neutrophils (immune cells) to come to the injury • Neutrophils clear debris and regulate the early inflammatory process • Injured area becomes ischemic and acidic which may cause secondary damage to otherwise healthy areas around the injury Phase 1 • Neutrophils die and are ingested by macrophages at the injury site • When this happens the macrophages begin to produce proteins that promote tissue repair • Early scar tissue forms • Early rehab should focus on pain management, decreasing swelling, promote tissue healing Phase 1 • • • • Immobilization or protection should be done RICE NSAIDS for decreased inflammatory symptoms Short term analgesic Phase 2: Repair/Regeneration • Repair- articular cartilage, meniscus, spinal cord (new tissue is not identical) • Regeneration- bone, muscle, peripheral nerve, blood vessels (heal with identical tissue) • Patients are still swollen and pain with motion • Blends with the inflammatory phase and remodeling phase • Day 7 to 21 Phase 2 • Fibrin clots form • Fibroblasts proliferate near the injury • Capillary proliferation occurs (more oxygen needed) • Fibroblasts produce fibronectin, collagens, glycoproteins • Fibronectin begins dormant followed by type III collagen then to stronger type I Phase 2 • Initially patient has structural deficiency • Treatment – early controlled mobilization • This aligns collagen with areas of physical stress making it stronger • Prevents atrophy Phase 3: Remodeling/Maturation • Lasts up to 24 months • May have persistent swelling and pain with motion • Fibroblast activity decreases throughout this phase • Capillary density decreases • Cellular matrix becomes more refined Phase 3 • More type I and type III collagen fibers are produced • Tensile strength improves • Rehab can increase the physical demands until patients return to their activities of daily living Fracture Healing • Four phases: – Inflammation – Soft callus formation – Hard callus formation – Bone remodeling Phase 1: Acute • Lasts up to a week • Hematoma formation, inflammation, angiogenesis (new blood vessel formation), soft callus formation • Dominated by immune cells • Phagocytic cells remove debris • Platelets form clots Phase 2: Repair/Regeneration • Lasts 8 to 12 weeks • Remodeling of scar tissue through cartilage formation, calcification and bone formation • Mesenchymal cells become cartilage cells • Proliferate the soft callus • Replace the scar tissue with cartilage • Chondrycytes hypertrophy and release chemicals to promote bone formation Phase 2 • • • • Osteoblasts produce new bone Revascularization of the region occurs Soft tissue is replaced by bone Casting is used to allow time for a proper callus to develop Phase 3: Remodeling • Starts during Phase 2 and continues for several years • Starts 21 days post fracture • Woven bone replaced by cortical or trabecular bone • New bone is remodeled by osteoclasts and osteoblasts based on mechanical loading Fracture Management • Treatment varies by : • type of fixation – – – – • • • • No reduction Closed reduction Open reduction Open reduction internal fixation (ORIF) Location of fx Involved bone Mechanical loads Surrounding soft tissue Fracture Management • ORIF – Surgical fixation – Uses metal implants to stabilize fx’s – May allow rehab to begin within first week after surgery ( edema control, wound management, early motion) – More aggressive rehab can begin in 6-8 weeks Fracture Management • Non-surgical fixation – ROM delayed for 3 weeks – Until callus has enough tensile strength to tolerate movement – At 6-8 weeks patient may begin strengthening exercises and increase mechanical loads – Upper extremity fx’s may begin ROM exercises sooner because of smaller loads and fear of atrophy Delayed/Non Union Fx’s • Causes – – – – – – – – – – – Nutritional deficiencies Diabetes, anemia Smoking Pharmacological drug use Pre-injury vascular status Muscle around fracture Inadequate immobilization Infection High energy fractures Gap distance Nerve injury Delayed / Non Union Fx’s • Susceptible areas – Tibia – Ulna – Femoral neck – Scaphoid (most common) Peripheral Nervous Healing • More proximal injuries result in greater losses • Within 3 -5 days, axons distal to the injury undergo a degenerative process called Wallerian degeneration • This is promote by immune cells, Schwann cells(the cell that normally myelinate axons) and the distal axon Nerve Healing • Produce proteins that produce and inflammatory response and pain • Schwann cells divide to create an optimal environment for regeneration • Surgical repairs can be helpful but are not guaranteed • Only one proximal branch will form a new axon the others will degenerate Muscle Healing • • • • • • • Tissue trauma Hematoma formation Inflammatory cell reaction Phagocytosis Capillary regrowth Scar formation remodeling Muscle Healing • Must balance regeneration with scar formation • Scar tissue is weak and only serves as a “scaffold” for the healing process • Site is still weak • No active motion immediately after injury • Could result in re-rupture • However, prolonged immobilization could result in atrophy Muscle Healing • What do you do? Muscle Healing • Early mobilization and motion may be started within the first 24 hours • It must be pain free to avoid overloading • Pain is your guide Tendon Healing • Ruptured tendons often require surgery • Once repaired the tendon ends can go through their normal healing stages Tendon Healing • • • • • • • Hematoma formation Platelet aggregation Recruitment of inflammatory cells Phagocytosis Angiogenesis Fibroblast proliferation remodeling Tendon Healing • Tensile strength increases at 4 weeks • Continues to improve up to 1 year • Strength of tendon never returns to pre-injury level Factors Affecting Healing • Healing process is variable • Factors: – Extensive trauma – Blood supply – Infection – Diabetes – Age – nutrition Critical Thinking • In a rehab setting, patients want to return ASAP. As a result, some patients do extra exercises, stretch to a point of pain, or try aggressive exercises (e.g. running) too early. What are the potential hazards to this approach? How would you explain your concerns to the patient or athlete? Critical Thinking • Two patients that are 10 weeks post –fractures are exercising next to each other. One of the patients had a distal radius fracture and the other had a scaphoid fracture. Both were treated conservatively with immobilization. The patient with the scaphoid fracture notices that she is still not able to do a lot of the exercises her counterpart is able to perform. She then asks why she is not progressing at the same rate as the other patient. How would you explain the differences in their recovery?