Nonmetal Oxides - BC Learning Network
advertisement

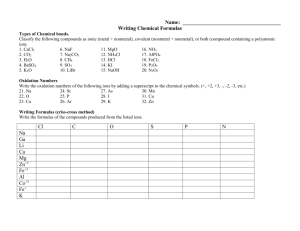
Here we’ll have a closer look at nonmetal oxides, and how they behave in water. Metals Nonmetals Nonmetal Oxides form Acidic Solutions Nonmetal Oxides are oxides of elements on the right side of the staircase on the periodic table. Remember, (click) nonmetal oxides react with water to form acidic solutions. O O S O SO3 We’ll consider the nonmetal oxide sulphur trioxide, SO3. Nonmetals have electronegativity values closer to that of oxygen than metals have. O O S O not an ionic solid So nonmetal oxides are not ionic solids like metal oxides are. O O S O Covalent Bonds Instead the atoms are joined by covalent bonds, where electrons are shared. O O O S O O H H O S OH O H SO3( l ) H 2O( l ) H 2SO4(aq) At room temperature, SO3 is a liquid. When it dissolves in water, O O O S O O H H O S OH O H SO3( l ) H 2O( l ) H 2SO4(aq ) It reacts with water, and through a series of reactions, O O O S O O H H H O S OH O SO3( l ) H 2O( l ) H 2SO4(aq) Forms sulphuric acid, H2SO4. The Lewis structure for sulphuric acid is shown here. If you check, you will see that sulphur, oxygen, and hydrogen atoms are conserved in this reaction. O H O S OH O H 2SO4 O H H H 2O When H2SO4 is in water, (click) a molecule of H2SO4 will collide with a water molecule and donate a proton to it, O– H O S OH O H H O H 24SO4 HSO + H 2H O3O forming a hydrogen sulphate ion and a hydronium ion. + O – H O S O O HSO4 + H O H H H 3O + Because hydronium ions are present, the solution will be acidic. Makes the Solution Acidic Now we’ll look at a few equations we can write for nonmetal oxides reacting with water. Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid 2Cl 2O(g) H 2O( l ) P4O6(s) 6H 2O( l ) P4O10(s) 6H 2O( l ) nitrous acid 2HOCl (aq) hypochlorous acid 4H 3PO 3(aq) phosphorous acid 4H 3PO4(aq) phosphoric acid When we are considering the reactions of nonmetal oxides with water, we usually stick to writing formula equations for these reactions. We’ll look at some of the common ones here. Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid 2Cl 2O(g) H 2O( l ) P4O6(s) 6H 2O( l ) P4O10(s) 6H 2O( l ) nitrous acid 2HOCl (aq) hypochlorous acid 4H 3PO 3(aq) phosphorous acid 4H 3PO4(aq) phosphoric acid The formula equation summarizing the example we just went over is sulphur trioxide or SO3 reacts with water to produce H2SO4, sulphuric acid. Sulphuric acid is a strong acid. Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid 2Cl 2O(g) H 2O( l ) P4O6(s) 6H 2O( l ) P4O10(s) 6H 2O( l ) nitrous acid 2HOCl (aq) hypochlorous acid 4H 3PO 3(aq) phosphorous acid 4H 3PO4(aq) phosphoric acid The other common oxide of sulphur, sulphur dioxide, SO2, reacts with water to produce H2SO3, or sulphurous acid. Sulphurous acid is a weak acid. Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid 2Cl 2O(g) H 2O( l ) P4O6(s) 6H 2O( l ) P4O10(s) 6H 2O( l ) nitrous acid 2HOCl (aq) hypochlorous acid 4H 3PO 3(aq) phosphorous acid 4H 3PO4(aq) phosphoric acid A common oxide of nitrogen is nitrogen dioxide, NO2. This reacts with Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid 2Cl 2O(g) H 2O( l ) P4O6(s) 6H 2O( l ) P4O10(s) 6H 2O( l ) Water to produce both nitrous acid 2HOCl (aq) hypochlorous acid 4H 3PO 3(aq) phosphorous acid 4H 3PO4(aq) phosphoric acid Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid 2Cl 2O(g) H 2O( l ) P4O6(s) 6H 2O( l ) P4O10(s) 6H 2O( l ) nitrous acid 2HOCl (aq) hypochlorous acid 4H 3PO 3(aq) phosphorous acid 4H 3PO4(aq) phosphoric acid Nitric acid, HNO3, which is a strong acid, Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid 2Cl 2O(g) H 2O( l ) P4O6(s) 6H 2O( l ) P4O10(s) 6H 2O( l ) nitrous acid 2HOCl (aq) hypochlorous acid 4H 3PO 3(aq) phosphorous acid 4H 3PO4(aq) phosphoric acid And nitrous acid, HNO2, which is a weak acid. Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid Cl 2O(g) H 2O( l ) P4O6(s) 6H 2O( l ) P4O10(s) 6H 2O( l ) nitrous acid 2HOCl (aq) hypochlorous acid 4H 3PO 3(aq) phosphorous acid 4H 3PO4(aq) phosphoric acid An oxide of the nonmetal chlorine, Dichlorine monoxide, Cl2O, reacts with water to produce the weak acid HOCl, hypochlorous acid. This is an equilibrium reaction. Oxides of the other halogens have similar Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid nitrous acid Cl 2O(g) H 2O( l ) 2HOCl (aq) hypochlorous acid P4O6(s) 6H 2O( l ) 4H 3PO3(aq) phosphorous acid P4O10(s) 6H 2O( l ) 4H 3PO4(aq) phosphoric acid One of the common oxides of phosphorus, P4O6, reacts with water to produce the weak acid H3PO3, phosphorous acid Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid nitrous acid Cl 2O(g) H 2O( l ) 2HOCl (aq) hypochlorous acid P4O6(s) 6H 2O( l ) 4H 3PO3(aq) phosphorous acid P4O10(s) 6H 2O( l ) 4H 3PO4(aq) phosphoric acid Another oxide of phosphorus, P4O10, reacts with water to produce the weak acid H3PO4, phosphoric acid Formula Equations for Reactions of Nonmetal Oxides with Water SO3( l ) H 2O( l ) H 2SO4(aq) sulphuric acid SO2(g) H 2O( l ) H 2SO3(aq) sulphurous acid 2NO2(g) H 2O( l ) HNO3(aq) HNO2(aq) nitric acid nitrous acid Cl 2O(g) H 2O( l ) 2HOCl (aq) hypochlorous acid P4O6(s) 6H 2O( l ) 4H 3PO3(aq) phosphorous acid P4O10(s) 6H 2O( l ) 4H 3PO4(aq) phosphoric acid You should be familiar with these reactions, especially the reactions for the oxides of sulphur and nitrogen. Nonmetal Oxides Are also Called Acidic Anhydrides You should also know that nonmetal oxides (click) are also called (click) acidic anhydrides. Nonmetal Oxides Are also Called Acid Anhydrides Or simply “acid anhydrides”. It’s important to remember that nonmetal oxides, or acid anhydrides react with water to produce acidic solutions.