Membranes and membrane transport
advertisement

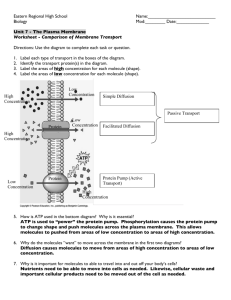
Major Ways Molecules Move Across Cell Membrane Name five different ways that molecules can move across the cell membrane: Cell Membranes and Fluid Balance • Composition of cell membrane? • Intracellular vs. interstitial vs. extracellular fluid compartments • Composition of the fluids in these compartments? • How is composition maintained? • Osmolarity vs. tonicity Extracellular Figure taken from: http://sorrel.humboldt.edu/~jlg21/Zoo%20310/Lab%202%20ADAM%20electrolytes/fluid%20compartments.gif What are the major differences in composition of the intracellular and extracellular fluid? Between the interstitial fluid and the plasma? Figure 5-3b 1. Simple diffusion Characteristics: Figure 5-5 Figure 5-6 Table 5-1 2. Facilitated Diffusion Characteristics: Figure 5-7 (3 of 3) Figure 5-9 - Overview Figure 5-10 Figure 5-14 Figure 5-15 Figure 5-11 3. Primary Active Transport Characteristics: Figure 5-4 Table 5-2 Figure 5-16 Figure 5-17 - Overview 4. Secondary Active Transport Characteristics: Figure 5-18 - Overview Figure 5-12 - Overview Table 5-3 Figure 5-19 The glucose transporters demonstrate common characteristics of all protein transporters: Table 5-4 1. Similarly shaped/charged molecules can compete for transport Figure 5-20 2. Some similarly shaped molecules can bind, but when they do, transport is stopped (competitive inhibition) Figure 5-21 - Overview 3. When the number of transporters is fixed, increasing the concentration of the transported molecule will eventually saturate the transporters, and the rate will remain at its maximum (unless more transporters are made). Figure 5-22 Test Your Knowledge: 1. Steroid hormones pass directly through the cell membrane to activate protein production. What type of transport is this? Why? 2. Why can urea diffuse freely from plasma into cells when most cholesterol, although lipid-soluble, must cross a membrane by receptor-mediated endocytosis? 3. From the graph below, what transport method appears to be used for the transfer of substance Z into a cell? Explain. How could you test whether it was primary or secondary active transport? [X] ECF ICF 4. Choose A if the statement refers to active transport, B if the statement refers to passive transport, and C if it can refer to both. • Movement of molecules from an area of high concentration to an area of low concentration. • Movement of molecules via proteins embedded in the cell membrane, requires ATP. • Movement of molecules against the concentration gradient. • A co-transporter is involved in molecule movement. • Movement of molecules that requires ATP. • This tends to create an equilibrium state. Receptor mediated endocytosis Figure 5-24 - Overview Transepithelial transport Figure 5-25 Figure 5-26 How do cells maintain their volume? What factors influence their ability to maintain their volume? Movement of water toward the area of highest solute concentration Osmolarity of the cell and solution surrounding it Tonicity of the solution outside the cell The solution with the highest solute concentration has the highest osmotic pressure Figure 5-29 - Overview Osmolarity = total number of dissolved particles in solution Osmolarity = molar concentration X number of particles in solution Osmolarity of 2M glucose? 1M NaCl? 1M MgCl2? Table 5-6 Tonicity describes the volume change that occur if a cell were placed in that solution, after the cell has come to equilibrium with the solution. Tonicity is not measured in units, it is only a comparative term. Can osmolarity tell you whether a solution is hypo-, iso-, or hyper-tonic? Table 5-7 Figure 5-30a Figure 5-30b Figure 5-31a Figure 5-31b Figure 5-31c Figure 5-31d Table 5-8 Table 5-9 Test Your Knowledge: 1. Which of the following solutions have the most water per unit volume: 1 M glucose, 1 M NaCl, or 1OsM NaCl ? How do you know? Two compartments are separated aby a membrane that is permeable to water and urea but not to NaCl. Which way will water move when the following solutions are placed in the two compartments? Compartment A Membrane Compartment B a) 1 M NaCl | 1 OsM NaCl b) 1 M urea | 2M urea c) 1 OsM NaCl | 1 OsM urea You have a patient who lost 1 liter of blood, and you need to restore volume quickly while waiting for a blood transfusion to arrive from the blood bank. a) Which would be better to administer: 5% dextrose in water or 0.9% NaCl in water? Defend your choice. b) How much of your solution of choice would you have to administer to return blood volume to normal? Resting Membrane Potential The relative charge difference (electrical gradient) between the intracellular and extracellular compartments. What can influence the charges on the inside or outside of the cell? • Charge-charge interactions • Activity of active transport pumps • Relative concentration of each ion Figure 5-32 - Overview Figure 5-33 Figure 5-34a Figure 5-34b Figure 5-34c Figure 5-35 Figure 5-36 Figure 5-37