potato core osmosis 2014
advertisement

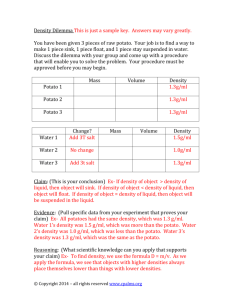
Name__________________________________________ Osmolarity in Potato Cores (Practical 2) Date___________ An intro to a similar procedure: http://www.youtube.com/watch?v=GQAVND5rtcw&noredirect=1 Osmolarity of potato tissue can be estimated by looking at the percent change in mass of potato samples after soaking them in solutions of different molarities. Day 1: Work in groups of 3. Read the procedure and discuss the reasons for the controls. Divide tasks so that time is used efficiently. Ask questions as needed before you begin. 1. Pour a constant volume of each sucrose solution (0.0 – 1.0 M) (enough to submerge your potato cores) into a container. (This may be a beaker / plastic cup / test tube.) 2. Label each beaker / plastic cup with the molarity of the solution you added. 3. Get ONE potato and place on a cutting board or paper towels. 4. Using a cork borer, take parallel cores of the potato and place them in a petri dish. You may wish to use a knife cut off the bottom of the potato as shown in the diagram below but on the long side so you waste less potato. Do not allow the cores to cross into each other. 5. Trim the cores with a knife or scalpel so that no potato skin remains. You need 12 cores of equal length from your ONE potato. 6. Quickly find the total mass of 2 potato cores by taring the electronic balance with an empty petri dish on top, and then placing the potato cores in the petri dish. 7. Add the cores to the solution in one labeled container. Do not leave the potato cores out for a long time before submerging them – work as a team. 8. Record the data for initial mass and qualitative data in the chart provided. Add data to excel. 9. Repeat for the remaining solutions, using 2 cores each time. 10. Cover the beakers. Dispose of used potato. Wash and clean up all materials. Day 2: 1. 2. 3. 4. 5. Remove the potato cores from one beaker and GENTLY blot them with a paper towel. Promptly determine the total mass of the 2 cores and record the data in the chart below. Repeat for the remaining solutions. Add data to excel. Dispose of potato cores in the trash and the solutions in the sink. Cleanup all materials. Some factors that affect change (or rate of change) in mass of potato: Starting osmolarity of potato tissue Tonicity of solution (hypertonic, hypotonic) Surface area : volume ratio Temperature Time Permeability of membrane Some controls to consider: Use of a single potato Squared potato for easier parallel coring Constant diameter and length of cores Only cores that have not intersected with other cores Prompt use of cores (not left out in the air for long), blotting cores after soaking but before massing Volume of solution Percent change in mass (rather than just change in mass) Soaking for a constant amount of time Day 3: In groups, download the excel file and create formulas to calculate the values needed to fill in the chart below. Together, use the processed data and the logger pro graphing program to estimate the osmolarity of the potato tissues. In your graph, consider different types of lines-of-best-fit and appropriate error bars. As a group submit one copy of: 1. The completed charts below – copy and paste (with your group’s data) 2. A screen shot from excel showing sample formulas your created 3. Your final graph created in logger pro Potato core data collection: Molarity of Initial sucrose solution Mass (±0.02 M) (±0.01g) Molarity of sucrose solution (±0.02 M) Final Mass (±0.01g) Change in mass (±0.02g) Percent change in mass Class average % change in mass ± standard deviation Qualitative data Before soaking After soaking Individually, on a separate sheet of paper, write and submit a concluding paragraph that addresses the following: 1. Your quantitative estimate of the osmolarity of the potatoes used in class 2. Your level of confidence in this result 3. The reasoning behind the technique you used to determine the osmolarity 4. The justification, as a cellular level, for why the potatoes had the percent changes in mass that were observed. The justification should use appropriate vocabulary related to membranes and diffusion.