ATOMIC STRUCTURE
advertisement

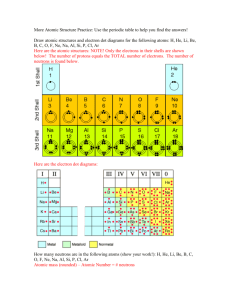
ATOMIC STRUCTURE P+ = PROTON No = NEUTRON E--- = ELECTRON DIFFERENT WAYS TO WRITE ELEMENTS & THEIR SYMBOLS Phosphorus—31 AND 3115P ***NEUTRONS = mass # -- atomic # ***isotopes--- atoms of the same element but with different numbers of neutrons (different mass #s) Ex: P—32 and P—30 Ex: phosphorus—31 Protons = 15 Electrons = 15 Neutrons = 16 NUMBER THESE COLUMNS ON YOUR PERIODIC TABLE: 1 2 13 16 17 (1A)(2A) (3A) (7A) 14 18 (4A) (8A) 15 (5A) (6A) Vertical columns—groups or families --- there are 18 total (8A groups and 10B groups) Horizontal rows—periods or series ---there are 7 rows total VALENCE ELECTRONS-- the outermost electrons having the highest amount of energy and are farthest from the nucleus and the ones involved in chemical reactions ** # of valence electrons = “A” column # on the periodic table (“A” group #) ** # of energy levels = row # on the periodic table Ex: Beryllium (Be): column 2A= 2 valence electrons Row 2 = 2 energy levels Ex: Phosphorus (P): column 5A = 5 valence electrons Row 3 = 3 energy levels ATOMIC MATH PROBLEM 1. Take the # of protons in oxygen X the # of electrons in fluorine = _______________ 2. Take this number --- # of neutrons in chlorine = _________________ 3. Take this number + silicon’s atomic # = ____________ 4. Take this number / # of neutrons in helium = _____________ 5.Take this number --- aluminum’s mass # = _____________ Atomic Math Problem #2 1. 2. 3. 4. 5. 6. Take sodium’s mass # and subtract its proton number. Take this number and divide by carbon’s neutron number. Take this number and multiply by calcium’s electrons. Take this number and divide by boron’s proton number. Take this number and multiply oxygen’s neutron number. Take this number and subtract zinc’s mass number. Calculating average atomic mass using % abundance information atomic mass-the average mass of all of an element's isotopes; you round this to actually get the mass number ***every element has at least 2 isotopes * **the most abundant isotope matches the mass # from the periodic table!! Ex: Which is most common? O-16 O-17 O-18 Hydrogen has 3 known isotopes: H-1 (protium) occurs 99.9885% of the time with an atomic mass of 1.0078 amu H-2 (deuterium) occurs 0.0115% of the time with an atomic mass of 2.0141 amu H-3 (tritium) is synthetically made MASS OF EACH ISOTOPE X DECIMAL FORM OF THE % OF THE TIME IT OCCURS (1.0078 X 0.999885 ) + (2.0141 X 0.000115) 1.007684103 + 0.000231622 1.007915725 amu Calculate avg. atomic mass for “element Z” Z—10 occurs 75.5% of the time Z—11 occurs 20.5 % of the time Z—12 occurs 4.0 % of the time (10 x 0.755) + (11 x 0.205) + (12 x 0.040) 10.285 amu