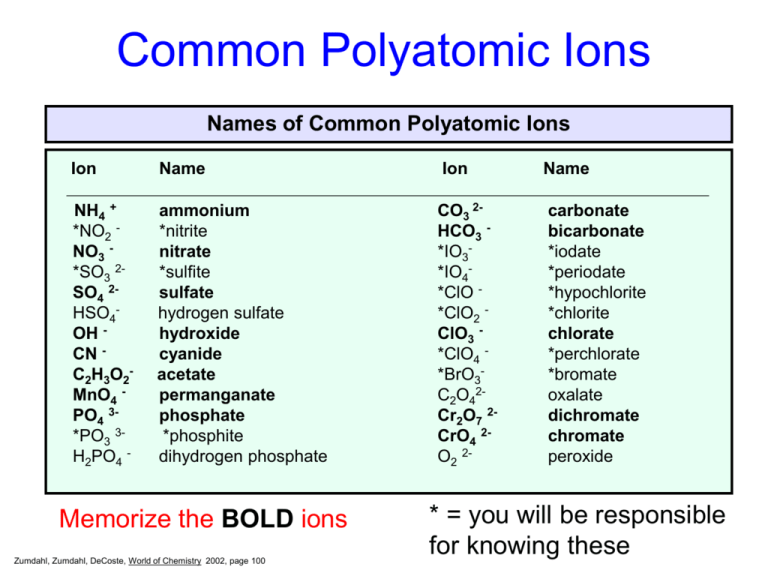
Common Polyatomic Ions
Names of Common Polyatomic Ions
Ion
Name
Ion
Name
NH4 +
*NO2 NO3 *SO3 2SO4 2HSO4OH CN C2H3O2MnO4 PO4 3*PO3 3H2PO4 -
ammonium
*nitrite
nitrate
*sulfite
sulfate
hydrogen sulfate
hydroxide
cyanide
acetate
permanganate
phosphate
*phosphite
dihydrogen phosphate
CO3 2HCO3 *IO3*IO4*ClO *ClO2 ClO3 *ClO4 *BrO3C2O42Cr2O7 2CrO4 2O2 2-
carbonate
bicarbonate
*iodate
*periodate
*hypochlorite
*chlorite
chlorate
*perchlorate
*bromate
oxalate
dichromate
chromate
peroxide
Memorize the BOLD ions
Zumdahl, Zumdahl, DeCoste, World of Chemistry 2002, page 100
* = you will be responsible
for knowing these
Polyatomic Ion Trends
*Charges NEVER change between different forms
+1 oxygen
“Per____ate”
BrO4perbromate
ClO4-
perchlorate
IO4periodate
“Normal”
“–ate” form
BrO3bromate
ClO3chlorate
IO3iodate
NO3nitrate
CO32carbonate
SO42sulfate
PO43phosphate
-1 oxygen
“-ite” form
-2 oxygens
“hypo___ite”
BrO2-
BrO-
bromite
hypobromite
ClO2-
ClO-
chlorite
hypochlorite
IO2-
IO-
iodite
hypoiodite
NO2nitrite
CO22carbonite
SO32sulfite
PO33phosphite
Atomic Theory
Review
Electron Cloud Model
• Orbital (“electron cloud”) instead of “orbits”
• Region in space where there is 90% probability
of finding an electron
90% probability of
finding the electron
Orbital Shape
Electron Probability vs. Distance
Electron Probability (%)
40
30
20
10
0
0
50
100
150
Distance from the Nucleus (pm)
Orbital
Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem
200
250
Shapes of s, p, and d-Orbitals
each holds 2 electrons (s2)
each of 3 orbitals holds 2 e - = 6 total p electrons (p6)
each of 5 orbitals holds 2 e - = 10 total d electrons (d10)
s, p, and d-orbitals
s orbitals:
Each holds 2
electrons
(outer orbitals of
Groups 1 and 2)
Orbital Filling Video
p orbitals:
Each of 3 sets holds
2 electrons = 6
electrons
(outer orbitals of
Groups 3 to 8)
d orbitals:
Each of 5 sets holds
2 electrons
= 10 electrons
(found in elements
in third period
and higher)
Orbitals overlap
each other as
you get farther
from the nucleus
Copyright © 2006 Pearson Benjamin Cummings. All rights reserved.
f-orbitals
7 f orbitals @ 2 e- each = 14 e-
s, p, d and f orbitals
How many g orbitals could exist?
theoretical g-orbitals
9 g orbitals @ 2 e- each = 18 e-
Periodic Patterns for Electron Config
n
1
2
3
4
5
6
7
p
s
8
2
1
3
1s
2s
6
5
2p
d (n-1)
3s
4
3p
4s
3d
4p
5s
4d
5p
6s
5d
6p
7s
6d
7p
f (n-2)
6
7
4f
5f
7
1s
Periodic Patterns
• Example - Hydrogen
1
2
3
4
5
6
7
1
1s
1st Period
# of e- in that
orbital(s)
s-block
Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem
16
Electron Configuration Review
S
32.066
• Longhand Configuration
S 16e- 1s2 2s2 2p6 3s2 3p4
Core Electrons
Valence Electrons
(Highest energy level)
• Shorthand Configuration
S
16e
2
4
[Ne] 3s 3p
Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem
32
Ge
Periodic Patterns
72.61
• Example - Germanium
1
2
3
4
5
6
7
[Ar]
2
4s
10
3d
Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem
2
4p
Shorthand Configuration
22s22p6)
neon's
electron
configuration(1s
A
rd
B 3rd energy level (or 3 period)
[Ne] 3s1
C 1 electron in the s orbital
D orbital shape (s,p,d,f…etc.)
22s22p6] 3s1
[
1s
Na =
electron configuration
Shorthand Configuration
Practice
Element symbol
Electron configuration
Ca
[Ar] 4s2
V
[Ar] 4s2 3d3
F
[He] 2s2 2p5
Ag
[Kr] 5s2 4d9
I
[Kr] 5s2 4d10 5p5
Xe
[Kr] 5s2 4d10 5p6
Fe
Sg
22p64s
[He] 2s[Ar]
3s223d
3p664s23d6
[Rn] 7s2 5f14 6d4
Periodic Patterns Review
• Period # (1-7)
• energy level
• (subtract for d & f)
1
2
3
4
5
6
7
• Group # (1-8…excluding d block)
• total # of valence e-
• Column within Sublevel block
• # of e- in sublevel/orbital
Courtesy Christy Johannesson www.nisd.net/communicationsarts/pages/chem
Periodic Patterns and Charge Trends
n
p
s
+1 +2
d
1
2
3
4
5
6
7
Variable Charge
METALS
f (n-2)
6
7
+3
-3 -2 -1
1s
NONMETALS
Electron Configurations for Cations
• Metals lose e- to attain a noble gas
configuration
• Metals form positive ions
– Cations are “paws”itive
• Metal ions will lose e- from configuration
Na: 1s22s22p63s1
Na+: 1s22s22p6
noble gas configuration
Formation of a Cation
sodium atom
Na
[Ne] 3s1
sodium ion
Na+
[Ne]
ee-
e-
e-
e-
e-
ee-
e-
11p+
ee-
loss of
one valence
electron
e-
e-
11p+
e-
e-
e-
e-
e-
e-
e-
e-
Electron Configurations for Anions
• Non-metals gain electrons to attain a noble
gas configuration
• They form negative ions
• Take a look at the e- configuration
S 1s22s22p63s23p4 (6 valence electrons)
+ 2eS2- 1s22s22p63s23p6 (noble gas configuration)
Formation of an Anion
chlorine atom
Cl
[Ne]3s23p5
e-
chloride ion
Cl –
[Ne]3s23p6 or [Ar]
egain of
one valence
electron
ee-
e-
e-
e-
eee-
e-
e-
e-
e-
ee-
e-
17p+
17p+
e-
e-
e-
e-
ee-
e-
e-
ee-
e-
e-
e-
e-
e-
ee-
e-
e-
Stable Electron Configurations
• All atoms react to achieve noble gas
configuration
• Noble gases have two s and six p electrons
• Eight valence electrons
• Also called the octet rule
Ar
Table salt
Bonding Review and
Writing Ionic Formulas
Formation of Ionic Bond
chloride ion
Cl -
sodium ion
Na+
e-
e-
ee-
e-
e-
e-
e-
e-
ee-
e-
11p+
e-
e-
e-
e-
e-
17p+
e-
ee-
e-
e-
e-
ee-
+
e-
e-
-
e-
Ionic Bonding
• Anions and cations are held together by
opposite charges
• All ionic compounds are called salts
• Simplest ratio is called the formula unit
• The bond is formed through the transfer
of electrons
• Electrons are transferred to fill all
available orbitals, or achieve a noble gas
configuration
K+
e-
e-
potassium atom
BrBr-
bromine atom
K+
O2K+
bromide ion
potassium
potassium
ion
bromide
potassium oxide
K2O
KBr
Br -
Mg2+
Br magnesium bromide
MgBr2
Bonding Practice
N3Pb4+
Na+
OH -
N3-
Al3+
N3-
NaOH
N3-
Pb4+
AlN
Pb4+
OH Mg2+
N3-
Ca2+
OH -
Mg(OH)2
?
CaBr2
Ca(OH)2
CaO
Pb4+
N3Pb3N4
lead (IV) nitride
or
plumbic nitride
Pb4+
N3-
NH4+
Ca2+
O2-
PO4
3-
NH4+
PO43-
Al3+
NH4+
Ca2+
O2-
(NH4)3PO4
PO43Ca2+
Ca3(PO4)2
Al3+
Fe2+
O2-
FeO
O2-
Al2O3
Subscripts, Superscripts and
Coefficients
superscripts
Mg
2+
3PO4
5 Mg3(PO4 )2
coefficient
subscripts
MAGNESIUM PHOSPHATE
Writing Formulas of Ionic Compounds
chemical formula:
• has neutral charge;
• shows types of atoms and how many of each
To write an ionic compound’s formula, we need:
1. the two types of ions
2. the charge on each ion
Na+
Ba2+
Na+
Ba2+
and
and
and
and
F–
O2–
NaF
BaO
O2–
Na2O
F–
BaF2
The Importance of Parentheses
Calcium hydroxide
ide
Ca2+
OH-
CaOH2
Ca - O
H
H
vs.
Ca(OH)2
HO - Ca - OH
The formulas imply two totally different compounds!
Writing Formulas w/Polyatomic Ions
Parentheses are required only when you need more
than one “bunch” of a particular polyatomic ion
Ba2+
and
SO42–
BaSO4
Mg2+
and
NO2–
Mg(NO2)2
NH4+
and
ClO3–
NH4ClO3
Sn4+
and
SO42–
Sn(SO4)2
Fe3+
and
Cr2O72–
Fe2(Cr2O7)3
NH4+
and
N3–
(NH4)3N
Nomenclature - Humor
Fe2+
Fe2+
Fe2+
Fe2+
Fe2+
Fe2+
Fe2+
Fe2+
“Ferrous Wheel”
Fe = iron (Latin = ferrum)
Fe2+ = lower oxidation state = ferrous
Fe3+ = higher oxidation state = ferric
BaNa2
“BaNaNa”
What weapon can you make
from the elements nickel,
potassium and iron?
A KNiFe
Teacher: What is the formula for water?
Student: H, I, J, K, L, M, N, O
Teacher: That’s not what I taught you.
Student: But you said the formula for water was…H to O.
"H-O-H"?! WHAT'S
THAT SPELL?!
mis
WATER?
Website: Dihydrogen monoxide Information Campaign
“Little Johnny took a drink,
Now he shall drink no more.
For what he thought was H2O,
Was H2SO4.”
Under aged Pb walks into a bar and the bartender turns to the gold
Bouncer and says, “Au, get the lead out!”
Four Types of Naming
• Fixed Charge Ionic Compounds
• Contain a metal with a fixed charge and a nonmetal (ionic)
• Variable Charge Ionic Compounds
• Contain a metal with a variable charge and a nonmetal (ionic)
• Acidic Compounds
• Contain H+ with various anions (ionic when aqueous)
• Covalent Molecules
• Contain 2 or more nonmetals only
Fixed charge cations
Variable charge cations
Elemental anions
1+
1
H
1
2
3
7
2
3- 2- 1-
B
C
N
O
F
Ne
3
4
5
6
7
8
9
10
Al
Si
P
S
Cl
Ar
13
14
15
16
17
18
Na Mg
K
12
Ca Sc
Ti
V
Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br
Kr
23
24
35
36
I
Xe
53
54
20
21
22
Rb Sr
Y
Zr Nb Mo Tc Ru Rh Pd Ag Cd
In
39
40
41
42
49
Hf
Ta
W
72
73
74
37
6
1
Be
19
5
3+
He
Li
11
4
2+
H
38
Cs Ba
55
56
Fr
Ra
87
88
*
W
25
43
26
44
Re Os
75
76
27
28
29
47
30
45
46
Ir
Pt Au Hg
Tl
77
78
81
79
48
31
80
32
33
34
Sn Sb Te
50
51
Pb Bi
82
83
52
Po At Rn
84
85
86
Rf Db Sg Bh Hs Mt
104
105
106
107
108
109
La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu
57
58
59
Ac Th Pa
89
90
91
60
U
92
61
62
63
64
65
66
Np Pu Am Cm Bk Cf
93
94
95
96
97
98
67
68
69
70
71
Es Fm Md No Lr
99
100
101
102
103
Fixed Charge Exceptions
3+
• Start with Al
• Go backwards down the stairs
• Decrease the charge after
each stair
3+
Al
13
2+
Zn
30
Ag
47
+
Fixed Charge Compound
Nomenclature
Metals (fixed charge) + Nonmetals
Fixed Charge Compounds
Compounds that contain a metal with a fixed charge
(group 1, 2, and 3, with Zn2+, Ag+, etc.), and a non-metal
To name these compounds, use the name of metal and the
name of the non-metal with the ending replaced by the suffix –ide
Examples:
NaCl
(Na+ Cl-)
sodium chloride
CaS
(Ca2+ S2-)
calcium sulfide
AlI3
(Al3+ 3I-)
aluminum iodide
Cations and Anions
Common Simple Cations and Anions
Cation
Name
H+
hydrogen
Li+
lithium
Na+
sodium
K+
potassium
Cs+
cesium
Be2+
beryllium
Mg2+
magnesium
Al3+
aluminum
Ag+
silver
EXCEPTIONS
Zn2+
zinc
Anion
HFCl Br IO 2S 2N 3P 3-
Name*
hydride
fluoride
chloride
bromide
iodide
oxide
sulfide
nitride
phosphide
*The root is given in color
Zumdahl, Zumdahl, DeCoste, World of Chemistry 2002, page 86
“Perhaps one of you gentlemen would mind telling me just
what is outside the window that you find so attractive..?”
Image courtesy NearingZero.net
Fixed Charge Naming Practice
Formula
Name
BaO
barium oxide
____________________
NaBr
2 ________________
sodium bromide
1
3
MgI2
magnesium iodide
____________________
4
KCl
potassium chloride
____________________
SrF2
5 ________________
strontium fluoride
CsF
6 ________________
cesium fluoride
Fixed Charge Nomenclature
w/Polyatomic Ions
Simply insert the name of the
polyatomic ion into the final name
Ba2+ and
SO42–
BaSO4
barium sulfate
Mg2+ and
NO2–
Mg(NO2)2
magnesium nitrite
NH4+ and
ClO3–
NH4ClO3
ammonium chlorate
Al3+
and
SO42–
Al2(SO4)3
alumnium sulfate
Na+
and
Cr2O72–
Na2Cr2O7
sodium dichromate
N3–
(NH4)3N
ammonium nitride
NH4+ and
Polyatomic Ions Quiz
PO43-
……………
phosphate
SO42-
……………
sulfate
Cr2O72MnO4-
…………..
…………..
CH3COO-
…….
dichromate
permanganate
acetate
Variable Charge Compound
Nomenclature
Metals (variable charge) + Nonmetals
Roman Numeral
Review
Number
1
Roman
Numeral
I
5
V
10
X
50
L
100
C
Greek
Number
1
2
3
4
5
6
7
8
9
10
Roman
Numeral
I
II
III
IV
V
VI
VII
VIII
IX
X
Single charge cations
Variable charge cations
Elemental anions
1+
1
H
2+
3+
Li
Be
B
3
4
1
2
3
Na Mg
11
4
K
19
5
7
Ca Sc
C
N
O
F
Ne
5
6
7
8
9
10
Al
Si
P
S
Cl
Ar
13
14
15
16
17
18
Ti
V
Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br
Kr
23
24
35
36
I
Xe
53
54
Rb Sr
Y
Zr Nb Mo Tc Ru Rh Pd Ag Cd
In
39
40
41
42
49
Hf
Ta
W
72
73
74
55
56
Fr
Ra
87
88
*
W
2
1-
22
Cs Ba
1
2-
21
38
He
3-
20
37
6
12
H
25
43
26
44
Re Os
75
76
27
28
29
47
30
45
46
Ir
Pt Au Hg
Tl
77
78
81
79
48
31
80
32
33
34
Sn Sb Te
50
51
Pb Bi
82
83
52
Po At Rn
84
85
86
Rf Db Sg Bh Hs Mt
104
105
106
107
108
109
La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu
57
58
59
Ac Th Pa
89
90
91
60
U
92
61
62
63
64
65
66
Np Pu Am Cm Bk Cf
93
94
95
96
97
98
67
68
69
70
71
Es Fm Md No Lr
99
100
101
102
103
Variable Charge Ionic Compounds
Compounds that contain a metal with variable charge
(Pb, Sn, and the transition metals—not including Ag or
Zn), and a non-metal
To name, given the formula:
1. Figure out charge on cation
2. Write name of cation
3. Write Roman numerals in ( )
to show cation’s charge
Stock System
of
nomenclature
Variable charge Naming
Practice
4. Write name of anion:
O2–
iron(II) oxide
FeO
Fe?
Fe2O3
2 Fe? 3 O2–
CuBr
Cu?
Br –
copper(I) bromide
CuBr2
Cu?
2 Br –
copper(II) bromide
iron(III) oxide
Variable Charge Cations
Common Type II Cations
Ion
Stock System
Fe 3+
Fe 2+
Cu 2+
Cu 1+
Co 3+
Co 2+
Sn 4+
Sn 2+
Pb 4+
Pb 2+
Hg 2+
Hg2 2+
iron (III)
iron (II)
copper (II)
copper (I)
cobalt (III)
cobalt (II)
tin (IV)
tin (II)
lead (IV)
lead (II)
mercury (II)
mercury (I)
Traditional System
ferric
ferrous
cupric
cuprous
cobaltic
cobaltous
stannic
stannous
plumbic
plumbous
mercuric
mercurous
*Mercury (I) ions are always bonded together in pairs to form Hg2 2+
Zumdahl, Zumdahl, DeCoste, World of Chemistry 2002, page 90
Variable Charge Ionic Compounds
To find the formula, given the name:
1. Write symbols for the two types of ions
2. Balance charges to write formula
cobalt (III) chloride Co3+ Cl–
CoCl3
tin (IV) oxide
Sn4+ O2–
SnO2
tin (II) oxide
Sn2+ O2–
SnO
Naming Variable Charge Ionic
Compounds
Formula
Name
1
Hg2O
mercury (I) oxide
____________________
2
HgO
mercury (II) oxide
____________________
CuF2
3 ________________
copper (II) fluoride
Cu2S
4 ________________
copper (I) sulfide
5
Cr2O3
PbO2
6 ________________
chromium (III) oxide
____________________
lead (IV) oxide
Ionic Compounds with
Polyatomic Ions
Fe3(PO4) 2
1. ________________
iron (II) phosphate
(NH4)2CO3
2. ________________
ammonium carbonate
Al2(SO4)3
3. ________________
aluminum sulfate
4.
V2SO3
vanadium (I) sulfite
____________________
5.
AgCN
silver cyanide
____________________
6.
Ba(ClO3)2
Cu(OH)2
7. ________________
barium chlorate
____________________
copper (II) hydroxide
Acidic Compounds
Oxysalts + H2O Oxyacids
Acidic Compounds
(Nonmetals Dissolved in Water)
Acids(aq) without oxygen
The prefix hydro- is used to represent hydrogen, followed by the root
name of the nonmetal with its ending replaced by the suffix –ic acid.
Examples:
(aq) = aqueous or
dissolved in water
*HCl(aq)
Hydrochloric acid
HBr(aq)
Hydrobromic acid
*The name of this gas would be hydrogen monochloride if it was NOT
dissolved in water.
Acidic Compounds
Nonmetals Dissolved in Water
Acids(aq) WITH oxygen (oxyacids)
Start with the name of the polyatomic ion (with oxygen), and change the
ending based on the following rule:
• If the ion ends in “ate,” change the suffix to –ic acid.
• If the ion ends in “ite,” change the suffix to –ous acid.
Retain any prefixes such as “per” or “hypo” in front of the acid name.
NEVER use “hydro”!
Examples:
HClO3(aq)
HBrO(aq)
ClO3- = chlorate
BrO- = hypobromite
Chloric acid
Hypobromous acid
Oxyacid Naming Rules
An ion with a
name ending in
An acid with a
name ending in
-ite
-ous
-ate
-ic
Hill, Petrucci, General Chemistry An Integrated Approach 1999, page 60
Common Oxyacid Names
The following table lists the most common families of oxy acids.
one more
HClO4
oxygen atom perchloric acid
most
“common”
HClO3
chloric acid
H2SO4
sulfuric acid
one less
oxygen
HClO2
chlorous acid
H2SO3
H3PO3
HNO2
sulfurous acid phosphorous acid nitrous acid
two fewer
oxygens
HClO
hypochlorous acid
H3PO4
phosphoric acid
HNO3
nitric acid
H3PO2
hypophosphorous acid
Acid Nomenclature Practice
Formula
Name
1
HCl
hydrochloric acid
____________________
2
HClO
hypochlorous acid
____________________
H2SO4
3 ________________
sulfuric acid
HF
4 ________________
hydrofluoric acid
5
H3N
HIO4
6 ________________
hydronitric acid
____________________
periodic acid
Covalent Compounds
Nonmetal + Nonmetal
Writing Formulas of Covalent
Compounds
Covalent Compounds
contain two or more non-metals (NO METALS!)
Key: FORGET CHARGES
To Name, given the formula:
1. Use Greek prefixes to indicate how many atoms
of each element, but don’t use “mono” on first
element (least electronegative, excluding H)
2. Add the suffix –ide to the second element (most
electronegative)
Prefixes to
memorize!
1
2
3
4
5
mono
di
tri
tetra
penta
6 hexa
7 hepta
8 octa
9 nona
10 deca
Covalent Nomenclature Practice
EXAMPLES:
1. carbon dioxide
2. CO
3. dinitrogen trioxide
4. SF6
5. carbon tetrachloride
6. NI3
CO2
carbon monoxide
N2O3
sulfur hexafluoride
CCl4
nitrogen triiodide
More Practice with covalent
compounds…
As2S3
1. ________________
diarsenic trisulfide
SO2
2. ________________
sulfur dioxide
3.
P2O5
NH3
4. ________________
diphosphorus pentoxide
____________________
nitrogen trihydride
5.
N2O5
dinitrogen pentoxide
____________________
6.
H2O
dihydrogen monoxide
____________________
Naming Simple Chemical Compounds
Ionic (metal and nonmetal)
Metal
Forms
only one
positive
ion
Use the
name of
element
Forms
more than
one positive
ion
Covalent (2 nonmetals)
Nonmetal
Single
Negative
Ion
Use element
Use the name
name followed
of the
by a Roman
element, but
numeral to
end with ide
show the charge
First
nonmetal
Second
nonmetal
Before
element name
use a prefix
to match
subscript
Use a prefix
before
element name
and end
with ide
Polyatomic
Ion
Use the
name of
polyatomic
ion (ate or
Ite)
Overall Nomenclature Practice
Ionic/variable
Ir F3
iridium (III) fluoride
Ca S
calcium sulfide
Ti S2
titanium (IV) sulfide
HCl(aq)
hydrochloric acid
Acid
BaBr2
barium bromide
Ionic/fixed
N3P2
trinitrogen diphosphide
Covalent
HF(aq)
hydrofluoric acid
Zn I2
zinc iodide
Ionic/fixed
NCl4
nitrogen tetrachloride
Covalent
Au 2O3
gold (III) oxide
Na 3P
sodium phosphide
Ionic/fixed
Ionic/variable
Acid
Ionic/variable
Ionic/fixed
Ionic Nomenclature Practice
Ionic/variable
Ir2 (Cr2O7)3
iridium (III) dichromate
Ca (OH)2
calcium hydroxide
HNO3 (aq)
nitric acid
Pt (CH3COO)2
platinum (II) acetate
HBrO2(aq)
bromous acid
Sr SO4
strontium sulfate
Ionic/fixed
KCN
potassium cyanide
Ionic/fixed
Zn (NO2)2
zinc nitrite
Ionic/fixed
Mn (ClO3)4
manganese (IV) chlorate
Ionic/variable
Au PO4
gold (III) phosphate
Ionic/variable
H2 SO3 (aq)
sulfurous acid
Ionic/fixed
Acid
Ionic/variable
Acid
Acid
Covalent
Ionic (M + NM)
Two or more NM’s
Variable charge cation
carbon
sulfurN
tetrabromide
dichloride
NCl
O35
2
vanadium
niobiumMn
Pt(IO
(V)
(II)
perchlorate
)4
2S
53chromate
Greek prefixes
Ionic (M + NM)
Fixed charge cation
rubidium
sulfate
NH4KI
ClO
barium
oxide
3
Roman
numeral
Roman numeral
for name only
Charge
Polyatomic ions OK
Criss-Cross
Rule
Roman
numeral
Polyatomic ions OK
Where would you file these?
VCrO4
dinitrogen pentoxide
BaO
platinum (IV) iodate
CBr4
ammonium chlorate
Nb(ClO4)5
potassium iodide
SCl2
nitrogen trichloride
Rb2SO4
manganese (V) sulfide
Suffixes have meaning
“-ide”
binary compound
sodium chloride (NaCl)
“-ite” or “-ate”
sulfite (SO32-)
sulfate (SO42-)
“-ol”
polyatomic compound
“-ate” means one more oxygen
than “-ite”
alcohol
methyl alcohol (methanol)
“-ose”
sugar
sucrose
“-ase”
sucrase
enzyme
Extra Slides
O
O
S
O
O
H
H
Compounds Containing Polyatomic Ions
Insert name of ion where it should go in the compound’s name.
Write formulas:
iron (III) nitrate
ammonium phosphide
ammonium chlorite
zinc phosphate
lead (II) permanganate
Fe3+
3 NO31–
Fe(NO3)3
3 NH41+
P3–
(NH4)3P
ClO21–
NH4ClO2
NH41+
3 Zn2+
Pb2+
2 PO43–
Zn3(PO4)2
2 MnO41–
Pb(MnO4)2
Writing Formulas of Ionic Compounds
Write names:
(NH4)2S2O3
ammonium thiosulfate
AgBrO3
silver bromate
(NH4)3N
ammonium nitride
U(CrO4)3
U?6+
Cr2(SO3)3
2 Cr ?3+
3 CrO42–
3 SO32–
uranium (VI) chromate
chromium (III) sulfite
Traditional (OLD) System of Nomenclature
…used historically (and still some today) to name
compounds w/multiple-charge cations
To use:
1. Use Latin root of cation.
2. Use -ic ending for higher charge; (“icky” food is good for you!)
“ -ous “
“ lower “ ; (“delicious” food is not good for you!)
3. Then say name of anion, as usual.
Element
Latin root
-ic
-ous
gold, Au
aur-
Au3+
Au1+
lead, Pb
plumb-
Pb4+
Pb2+
tin, Sn
stann-
Sn4+
Sn2+
copper, Cu
cupr-
Cu2+
Cu1+
iron, Fe
ferr-
Fe3+
Fe2+
Write formulas:
cuprous sulfide
Cu1+ S2–
auric nitride
Au3+ N3–
Write names:
3 Pb? 4 P3–
plumbic phosphide
Pb3P2
3 Pb? 2 P3–
Cu2S
AuN
plumbous phosphide
SnCl4
ferrous fluoride
Fe2+ F1–
Pb3P4
FeF2
Sn? 4 Cl1–
stannic chloride
Polyatomic Ion:
a group of atoms that stay together and have a single, overall charge.
BrO41-
Perbromate ion
CO42ClO41IO41NO41PO53SO521 more oxygen
BrO31-
BrO1-
Bromate ion
BrO21-
Bromite ion
CO32-
CO22-
CO2-
ClO31-
ClO21-
ClO1-
IO31-
IO21-
IO1-
NO31-
NO21-
NO1-
PO43-
PO33-
PO23-
SO42-
SO32-
SO22-
“normal”
1 less oxygen
Carbonate ion
Chlorate ion
Iodate ion
Nitrate ion
Phosphate ion
Sulfate ion
Hypobromite ion
2 less oxygen
Two nonmetals
Multiple-charge cation
Everything else
carbon
sulfurN
tetrabromide
dichloride
NCl
O35
2
vanadium
niobiumMn
Pt(IO
(V)
(II)
perchlorate
)4
2S
53chromate
rubidium
sulfate
NH4KI
ClO
barium
oxide
3
Greek prefixes
Roman numeral
for name only
Roman
numeral
Charge
Polyatomic ions OK
CrissCross
Rule
Roman
numeral
Polyatomic ions OK
Where would you file this?
VCrO4
dinitrogen pentoxide
BaO
platinum (IV) iodate
CBr4
ammonium chlorate
Nb(ClO4)5
potassium iodide
SCl2
nitrogen trichloride
Rb2SO4
manganese (V) sulfide
Two nonmetals
Greek prefixes
Multiple-charge cation
Everything else
Roman numeral
for name only
Roman
numeral
Charge
Polyatomic ions OK
CrissCross
Rule
Roman
numeral
Polyatomic ions
Where would you file this?
VCrO4
dinitrogen pentoxide
BaO
platinum (IV) iodate
CBr4
ammonium chlorate
Nb(ClO4)5
potassium iodide
SCl2
nitrogen trichloride
Rb2SO4
manganese (V) sulfide
Write the compound formed by the following ions:
1) Al3+ S22) Mg2+
PO43-
When a formula is given…write the proper name.
When a name is given…write the proper formula.
3) BaO
4) lithium bromide
5) Ni2S3
6) triphosphorus heptoxide
7) N2O5
8) molybdenum (VI) nitride
Write the total number of atoms that make up each compound.
9) trinitrotoluene (TNT)… CH3C6H2(NO2)3
10) phosphoric acid H3PO4
Extra credit: What is the formula for plumbic iodide? (Hint: lead is Pb2+ or Pb4+)
Write the compound formed by the following ions:
1) Al3+ S22) Mg2+
PO43-
When a formula is given…write the proper name.
When a name is given…write the proper formula.
3) BaO
POP
QUIZ
4) lithium bromide
5) Ni2S3
6) triphosphorus heptoxide
7) N2O5
8) molybdenum (VI) nitride
Write the total number of atoms that make up each compound.
9) trinitrotoluene (TNT)… CH3C6H2(NO2)3
10) phosphoric acid H3PO4
Extra credit: What is the formula for plumbic iodide? (Hint: lead is Pb2+ or Pb4+)






