1 atomic structure
advertisement

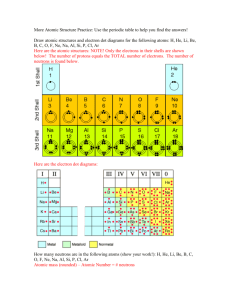
Unit One: Beginning Chemistry 1. Atoms, Elements, Compounds Names & Symbols 2. Atomic structure, volume of atom 3. Periodic Table Introduction: calculating atomic mass, atomic #, p+, e-, n0, carbon-12 4. Isotopes : nuclear symbol, hyphen symbol 5. Ion introduction- symbols Worksheets: (1.1) (1.2) (1.3) Chemistry Tables Structure of the Atom Atom: smallest unit of an element Element: pure substance made of only 1 kind of atom. - pure gold, pure sulfur, pure copper…. all listed on periodic chart. Compound: 2 or more atoms of an element chemically combined.- CO2, NaCl, H20 Symbols for each element: periodic chart First letter always CAPITAL 3 main parts to an ATOM: Symbol Charge Size Mass Location Neutron n0 O 1 1 nucleus Proton p+ positive 1 1 nucleus Electron enegative 0 0 electron cloud Protons and neutrons can be broken down into smaller parts called quarks. 3 quarks from protons, 3 quarks from neutrons Do you think it is possible to walk through walls? If the atomic nucleus were the size of this magnet, the first energy level containing electrons would be on the second tier stadium seats. Photo via permission Dreamstime Unit One: Beginning Chemistry 1. Atoms, Elements, Compounds Names & Symbols 2. Atomic structure, volume of atom Unit One: Beginning Chemistry 3. Periodic Table Introduction: calculating atomic mass, atomic #, p+, e-, n0, carbon-12 Worksheets: (1.1) Periodic Table Look on periodic chart 17 Cl Atomic # is # of protons……identifies element ….if element is neutral then #p+ = #eChlorine has 17 protons or 17 electrons 35.45 Atomic Mass # is # of protons + # neutrons Chlorine has 17 protons and 18 neutrons on the average (Subtract the two numbers) Ws 1.1 What is element 29 with #e-,#p+,no, & atomic mass 1. 2. 3. 4. 5. Ag, Cu, Cu, Ni, Cu, 29,29,60, 107.87amu 29,29,34.55, 63.55amu 29, 29,35, 63.55amu 29, 29, 34, 58.69amu 29,29,60, 107.87amu Calculate atomic mass of magnesium 1. 2. 3. 4. 5. 12 54.94 24.00 24.31 40.08 What is the atomic number for Phosphorus? 1. 2. 3. 4. 5. 6. 19 39.10 15 30.97 16 32.07 Calculate #of neutrons of Arsenic 1. 2. 3. 4. 5. 6. 7. 33 74.92 74 75 42 41.92 40.92 Atomic mass # of all periodic chart is based on carbon-12. Why? All life forms are carbon based = organic So Silicon atomic mass is 28/12 of carbon Germanium is 73/12 heavier than carbon Tin is 118.7 / 12 heavier than carbon Lead is 207 / 12 heavier than carbon What is the weight ratio of vanadium to gold? 1. 2. 3. 4. 5. 6. 1:2 2:3 1:1 1:4 4:2 4:3 Unit One: Beginning Chemistry 3. Periodic Table Introduction: calculating atomic mass, atomic #, p+, e-, n0, carbon-12 Worksheets: (1.1) Unit One: Beginning Chemistry 4. Isotopes : nuclear symbol, hyphen symbol Worksheets: (1.2) (1.3) Isotopes or Nuclides Atoms same # p+ which identify the element … but atoms may have different # of n0. Hydrogen H= H= H= 1p+ 1p+ 1p+ 1e1e1e- Isotopes only concerned with neutrons: “protons do not change” Isotopes are used in cancer detection 0no 1no 2no 2 ways to write an Isotope Nuclear Symbol Ws. 1.2 Ws 1.3 Hyphen 50 8 8 5 1 142 How many p+, e-, no are in the isotope Chlorine-37 1. 2. 3. 4. 37- 37- 35.45 17- 52.45 - 17 17- 17- 35.45 17- 17- 20 How many protons, electrons, neutrons in Calcium- 45 1. 2. 3. 4. 45,45,22 20, 20 45 40.08, 20,20 20,20,25 Isotopes of each element differ in 1. 2. 3. 4. The number of neutrons in the nucleus Atomic number The number of electrons in the highest energy level The total number of electrons Unit One: Beginning Chemistry 4. Isotopes : nuclear symbol, hyphen symbol Unit One: Beginning Chemistry 5. Ion introduction- symbols Ion: an atom or group of bonded atoms that have a positive or negative charge • How do you get a positive or negative charge? By gaining or losing an electron. • Ionization: any process resulting in the formation of an ion. Lost an electron Become positive + called cation K+1 Mg+2 B+3 Gained an electron become negative called Anion F-1 O-2 N-3 All metals become cations OIL RIG Calculate the number of electrons for the cation Si+4. 1. 2. 3. 4. 14 18 4 10 The anion for phosphorus-3 contains 1. 2. 3. 4. 5. 6. Ten electrons Eleven electrons Twelve electrons Thirteen electrons Fourteen electrons Fifteen electrons Calculate number of electrons for the anion Br -1 1. 2. 3. 4. 5. 6. 34 35 36 79 80 81 introduce ions as electrolytes Pure water does not conduct a current. Water containing dissolved salt conducts a current. Pure water does not conduct a current. Water containing dissolved salt conducts a current. Change # of protons… change element Change # of neutrons… it is an isotope Change # of electrons … it is an ion Unit One: Beginning Chemistry 1. Atoms, Elements, Compounds Names & Symbols 2. Atomic structure, volume of atom 3. Periodic Table Introduction: calculating atomic mass, atomic #, p+, e-, n0, carbon-12 4. Isotopes : nuclear symbol, hyphen symbol 5. Ion introduction- symbols Worksheets: (1.1) (1.2) (1.3)