Models of the Atom Notes Template
advertisement
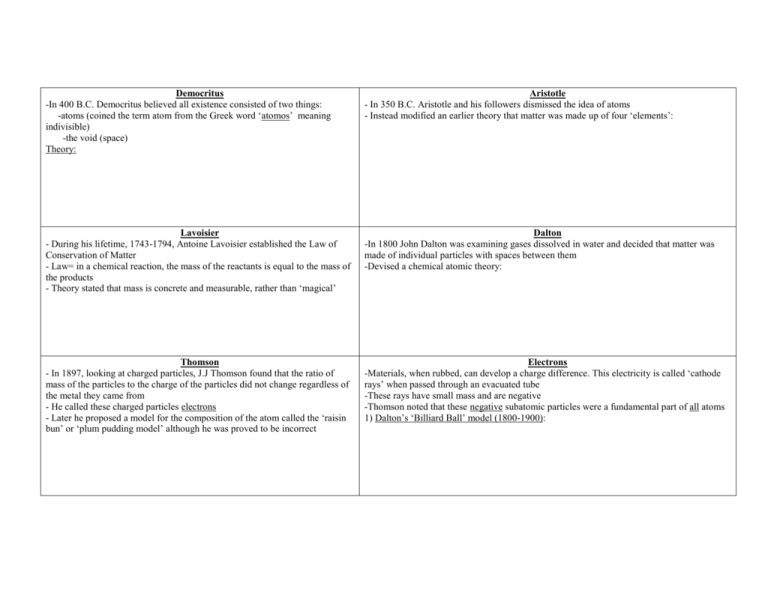
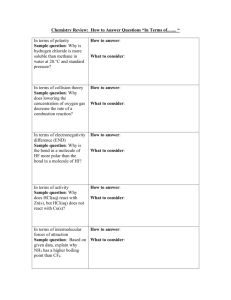
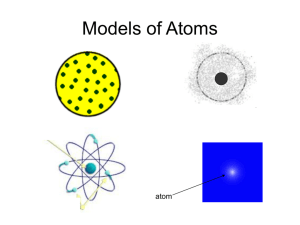
Democritus -In 400 B.C. Democritus believed all existence consisted of two things: -atoms (coined the term atom from the Greek word ‘atomos’ meaning indivisible) -the void (space) Theory: Aristotle - In 350 B.C. Aristotle and his followers dismissed the idea of atoms - Instead modified an earlier theory that matter was made up of four ‘elements’: Lavoisier - During his lifetime, 1743-1794, Antoine Lavoisier established the Law of Conservation of Matter - Law= in a chemical reaction, the mass of the reactants is equal to the mass of the products - Theory stated that mass is concrete and measurable, rather than ‘magical’ Dalton -In 1800 John Dalton was examining gases dissolved in water and decided that matter was made of individual particles with spaces between them -Devised a chemical atomic theory: Thomson - In 1897, looking at charged particles, J.J Thomson found that the ratio of mass of the particles to the charge of the particles did not change regardless of the metal they came from - He called these charged particles electrons - Later he proposed a model for the composition of the atom called the ‘raisin bun’ or ‘plum pudding model’ although he was proved to be incorrect Electrons -Materials, when rubbed, can develop a charge difference. This electricity is called ‘cathode rays’ when passed through an evacuated tube -These rays have small mass and are negative -Thomson noted that these negative subatomic particles were a fundamental part of all atoms 1) Dalton’s ‘Billiard Ball’ model (1800-1900): Electrons Cont. 2) Thomson’s ‘Plum Pudding’ Model (1900): Rutherford - Used radiation to unlock clues to the atom; alpha particle (α) -positive charge and beta particle(β) -negative charge - Ernest Rutherford short alpha (α) particles at gold foil - Most particles passed through; therefore, atoms are mostly empty - Some positive alpha deflected or bounced back! - Thus, a ‘nucleus’ is positive and holds most of an atom’s mass 3) The Rutherford Model (around 1910): Millikan - In 1907 Robert Millikan found that all electrons are identical - Millikan was able to find the charge of a single electron Bohr - Around 1913 Niels Bohr discovered that electrons orbit the nucleus in ‘shells’ - Electrons can be bumped up to a higher sell if hit by an electron or a photon of light - It is when electrons fall back down energy level(s) that they release a photon