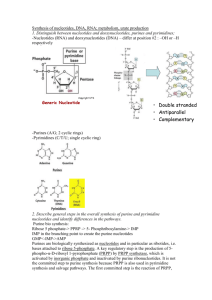
Metabolism of Nucleotides
advertisement

Biochemistry Ⅱ Zhihong Li(李志红) Department of Biochemistry Main Topics Metabolism of Nucleotides (4h) DNA replication(4h); RNA transcription(4h); Protein synthesis (4h) Gene expression and regulation (4h); Recombinant DNA technology (4h) Signal transduction(4h); Oncogene(2h); Gene and disease (2h) Diabetes mellitus (2h); Lipoproteins Metabolism (4h) Cholesterol Metabolism (2h); Bile acids Metabolism (2h) Plasma Proteins and Immuno Proteins (2h) Inter-assesment Free Radicals and Antioxidants (2h) ; Mineral Metabolism(2h) Water and Electrolyte Balance(2h); Acid Base Balance (2h) Heme Synthesis (2h); Bile Pigments Metabolism (2h) Liver function tests (2h); Metabolism of xenobiotics (2h) Hormones (6h); Biochemical changes during Pregnancy (2h) Biochemistry of Cerebrospinal fluid(CSF)(2h) Lecture 1 Metabolism of Nucleotides N N N N N H N Contents • • • • • • Review: Structure of nucleic acid Degradation of nucleic acid Synthesis of Purine Nucleotides Degradation of Purine Nucleotides Synthesis of Pyrimidine Nucleotides Degradation of Pyrimidine Nucleotides Nucleoside and Nucleotide Nucleoside = Nitrogenous base ribose Nucleotide = Nitrogenous base ribose phosphate Purines vs Pyrimidines Structure of nucleotides pyrimidine OR purine N-b-glycosyl bond Ribose or 2-deoxyribose Section 1 Degradation of nucleic acid Degradation of nucleic acid Nucleoprotein In stomach Gastric acid and pepsin Nucleic acid In small intestine Protein Endonucleases: RNase and DNase Nucleotide Nucleotidase Phosphate Nucleoside Nucleosidase Base Ribose Significances of nucleotides 1. Precursors for DNA and RNA synthesis 2. Essential carriers of chemical energy, especially ATP 3. Components of the cofactors NAD+, FAD, and coenzyme A 4. Formation of activated intermediates such as UDP-glucose and CDP-diacylglycerol. 5. cAMP and cGMP, are also cellular second messengers. Section 2 Synthesis of Purine Nucleotides There are two pathways leading to nucleotides • De novo synthesis: The synthesis of nucleotides begins with their metabolic precursors: amino acids, ribose-5-phosphate, CO2, and one-carbon units. • Salvage pathways: The synthesis of nucleotide by recycle the free bases or nucleosides released from nucleic acid breakdown. § 2.1 De novo synthesis • Site: – in cytosol of liver, small intestine and thymus • Characteristics: a. Purines are synthesized using 5phosphoribose(R-5-P) as the starting material step by step. b. PRPP(5-phosphoribosyl-1-pyrophosphate) is active donor of R-5-P. c. AMP and GMP are synthesized further at the base of IMP(Inosine-5'-Monophosphate). 1. Element sources of purine bases N10-Formyltetrahydrofolate N10-Formyltetrahydrofolate First, synthesis Inosine-5'-Monophosphate, IMP FH4 (or THF) N10—CHO—FH4 2. Synthesis of Inosine Monophosphate (IMP) • Basic pathway for biosynthesis of purine ribonucleotides • Starts from ribose-5-phosphate(R-5-P) • Requires 11 steps overall • occurs primarily in the liver OH Step 1:Activation of ribose-5-phosphate Committed step 1 ATP AMP 2 ribose phosphate pyrophosphokinase Step 2: acquisition of purine atom N9 Gln:PRPP amidotransferase •Steps 1 and 2 are tightly regulated by feedback inhibition 5-磷酸核糖胺,PRA Step 3: acquisition of purine atoms C4, C5, and N7 3 glycinamide synthetase 甘氨酰胺核苷酸 •Step 4: acquisition of purine atom C8 4 GAR transformylase 甲酰甘氨酰胺核苷酸 Step 5: acquisition of purine atom N3 5 甲酰甘氨咪核苷酸 •Step 6: closing of the imidazole ring 6 5-氨基咪唑核苷酸 Step 7: acquisition of C6 7 AIR carboxylase Carboxyaminoimidazole ribonucleotide (CAIR) 5-氨基-4-羧基咪唑核苷酸 Step 8: acquisition of N1 Carboxyaminoimidazole ribonucleotide (CAIR) SAICAR synthetase 5-氨基-4-(N-琥珀酸) -甲酰胺咪唑核苷酸 Step 9: elimination of fumarate adenylosuccinate lyase 5-氨基-4-甲酰胺咪唑核苷酸 Step 10: acquisition of C2 AICAR transformylase 5-甲酰胺基-4-甲酰胺咪唑核苷酸 Step 11: ring closure to form IMP • Once formed, IMP is rapidly converted to AMP and GMP (it does not accumulate in cells). N10-CHOFH4 N10-CHOFH4 3. Conversion of IMP to AMP and GMP Note: GTP is used for AMP synthesis. Note: ATP is used for GMP synthesis. IMP is the precursor for both AMP and GMP. 4. ADP, ATP, GDP and GTP biosynthesis kinase kinase AMP ATP ADP ADP ATP ATP kinase ADP kinase GTP GDP GMP ATP ADP ATP ADP 5. Regulation of de novo synthesis The significance of regulation: (1) Meet the need of the body, without wasting. (2) AMP and GMP control their respective synthesis from IMP by a feedback mechanism, [GTP]=[ATP] • Purine nucleotide biosynthesis is regulated by feedback inhibition § 2.2 Salvage pathway • Purine bases created by degradation of RNA or DNA and intermediate of purine synthesis can be directly converted to the corresponding nucleotides. • The significance of salvage pathway : – Save the fuel. – Some tissues and organs such as brain and bone marrow are only capable of synthesizing nucleotides by salvage pathway. • Two phosphoribosyl transferases are involved: – APRT (adenine phosphoribosyl transferase) for adenine. – HGPRT (hypoxanthine guanine phosphoribosyl transferase) for guanine or hypoxanthine. Purine Salvage Pathway . adenine phosphoribosyl transferase Adenine PRPP AMP PPi O N O N 2-O N N N Hypoxanthine O N N hypoxanthine-guanine phosphoribosyl transferase (HGPRT) PRPP N N Guanine NH2 3POH2C O N N N HO OH IMP O PPi N 2-O 3POH2C O N N N NH2 HO OH GMP . Absence of activity of HGPRT leads to Lesch-Nyhan syndrome. Lesch-Nyhan syndrome • first described in 1964 by Michael Lesch and William L. Nyhan. • there is a defect or lack in the HGPRT enzyme • Sex-linked metabolic disorder: only males • the rate of purine synthesis is increased about 200-fold – Loss of HGPRT leads to elevated PRPP levels and stimulation of de novo purine synthesis. • uric acid level rises and there is gout • in addition there are mental aberrations • patients will self-mutilate by biting lips and fingers off Lesch-Nyhan syndrome § 2. 3 Formation of deoxyribonucleotide • Formation of deoxyribonucleotide involves the reduction of the sugar moiety of ribonucleoside diphosphates (ADP, GDP, CDP or UDP). • Deoxyribonucleotide synthesis at the nucleoside diphosphate(NDP) level. P P O CH2 O Base ribonucleotide reductase P P O CH2 O Mg2+ Base H2O OH H S SH thioredoxin thioredoxin dNDP NDP S SH ATP £¨N=A, G, C, U£© kinase FAD + + NADPH + H NADP thioredoxin ADP reductase dNTP OH OH Deoxyribonucleotide synthesis at the NDP level § 2. 4 Antimetabolites of purine nucleotides • Antimetabolites of purine nucleotides are structural analogs of purine, amino acids and folic acid. • They can interfere, inhibit or block synthesis pathway of purine nucleotides and further block synthesis of DNA, RNA, and proteins. • Widely used to control cancer. 1. Purine analogs • 6-Mercaptopurine (6-MP) is a analog of hypoxanthine. OH SH N N N N H hypoxanthine N N N N H 6-MP • 6-MP nucleotide is a analog of IMP de novo synthesis - amidotransferase - 6-MP IMP 6-MP nucleotide - AMP and GMP - HGPRT - salvage pathway 2. Amino acid analogs • Azaserine (AS) is a analog of Gln. O H2N NH2 C CH2 CH2 O N N CH2 C CH COOH Gln NH2 O CH2 CH COOH AS 3. Folic acid analogs • Aminopterin (AP) and Methotrexate (MTX) NH2 N N H2N N CH2 R O N C NH C CH2 H OH H2N H N N CH2 COOH N MTX R=CH3: TXT R=H: AP N COOH CH2 N O COOH C NH C H CH2 CH2 N folic acid COOH NADPH + H+ NADP+ folate FH2 reductase - NADPH + H+ FH2 NADP+ FH2 reductase FH4 - AP or MTX •The structural analogs of folic acid(e.g. MTX) are widely used to control cancer (e.g. leukaemia). •Notice: These inhibitors also affect the proliferation of normally growing cells. This causes many side-effects including anemia, baldness, scaly skin etc. Section 3 Degradation of Purine Nucleotides NH2 Adenosine N Deaminase C N C O C HN C N CH CH HC C HC N N C N O N Ribose-P Ribose-P IMP AMP C HN CH HC C C N HN C C O C N H N H Hypoxanthine O C HN C N Xanthine Oxidase O C N H Uric Acid (2,6,8-trioxypurine) C N O CH C O C N H N N H GMP Xanthine The end product of purine metabolism Uric acid • Uric acid is the excreted end product of purine catabolism in primates, birds, and some other animals. • The rate of uric acid excretion by the normal adult human is about 0.6 g/24 h, arising in part from ingested purines and in part from the turnover of the purine nucleotides of nucleic acids. • The normal concentration of uric acid in the serum of adults is in the range of 3-7 mg/dl. GOUT • The disease gout, is a disease of the joints, usually in males, caused by an elevated concentration of uric acid in the blood and tissues. • The joints become inflamed, painful, and arthritic, owing to the abnormal deposition of crystals of sodium urate. • The kidneys are also affected, because excess uric acid is deposited in the kidney tubules. The uric acid and the gout Out of body In urine Hypoxanthine Xanthine Uric acid Over 8mg/dl, in the plasma Diabetese nephrosis …… Gout, Urate crystallization in joints, soft tissue, cartilage and kidney Advanced Gout Clinically Apparent Tophi 2 1 1 1. Photos courtesy of Brian Mandell, MD, PhD, Cleveland Clinic. 2. Photo courtesy of N. Lawrence Edwards, MD, University of Florida. 3. ACR Clinical Slide Collection on the Rheumatic Diseases, 1998. 3 Allopurinol – a suicide inhibitor used to treat Gout O O C C HN C N HN C H C N CH HC C N H Hypoxanthine N HC C N Allopurinol Xanthine oxidase Xanthine oxidase N H Section 4 Synthesis of Pyrimidine Nucleotides § 4.1 De novo synthesis • shorter pathway than for purines • Pyrimidine ring is made first, then attached to ribose-P (unlike purine biosynthesis) • only 2 precursors (aspartate and glutamine, plus HCO3-) contribute to the 6-membered ring • requires 6 steps (instead of 11 for purine) • the product is UMP (uridine monophosphate) 1. Element source of pyrimidine base C Gln N3 4 5C Asp CO 2 C2 1 N 6C Step 1: synthesis of carbamoyl phosphate •Carbamoyl phosphate synthetase(CPS) exists in 2 types: •CPS-I, a mitochondrial enzyme, is dedicated to the urea cycle and arginine biosynthesis. •CPS-II, a cytosolic enzyme, used here. It is the committed step in animals. Step 2: synthesis of carbamoyl aspartate ATCase: aspartate transcarbamoylase •Carbamoyl phosphate is an “activated” compound, so no energy input is needed at this step. Step 3: ring closure to form dihydroorotate Step 4: oxidation of dihydroorotate to orotate CoQ QH2 (a pyrimidine) Step 5: acquisition of ribose phosphate moiety Step 6: decarboxylation of OMP The big picture 3. UTP and CTP biosynthesis kinase kinase UMP UDP ATP ADP ATP UTP ADP 4. Formation of dTMP The immediate precursor of thymidylate (dTMP) is dUMP. The formation of dUMP either by deamination of dCMP or by hydrolyzation of dUDP. The former is the main route. UDP dUDP dCMP dCDP dUMP N5,N10-methylenetetrahydrofolic Acid dTMP synthetase dTMP ATP ATP ADP dTDP dTTP ADP dTMP synthesis at the nucleoside monophosphate level. dUDP H2O O O Pi NH3 O H2O dCMP thymidylate synthase HN HN O N CH3 N 5 10 FH2 N , N -CH2-FH4 d R 5' P d R 5' P FH2 dTMP dUMP NADPH reductase + H+ FH4 NADP+ § 4. 2 Salvage pathway uridine-cytidine kinase uridine cytidine + ATP deoxythymidine + ATP deoxycytidine + ATP uracil thymine + PRPP orotic acid thymidine kinase deoxycytidine kinase pyrimidine phosphate ribosyltransferase UMP + ADP CMP dTMP + ADP dCMP + ADP UMP dTMP + PPi OMP § 4. 3 Antimetabolites of pyrimidine nucleotides • Antimetabolites of pyrimidine nucleotides are similar with them of purine nucleotides. 1. Pyrimidine analogs • 5-fluorouracil (5-FU) is a analog of thymine. O F HN O O N H 5-FU CH3 HN O N H thymine 2. Amino acid analogs • Azaserine (AS) inhibits the synthesis of CTP. 3. Folic acid analogs • Methotrexate (MTX) inhibits the synthesis of dTMP. 4. Nucleoside analogs • Arabinosyl cytosine (ara-c) inhibits the synthesis of dCDP. NH2 NH2 N N O CH2OH O H N OH H H H OH O CH2OH O H ara-c N H H H OH OH cytosine Section 5 Degradation of Pyrimidine Nucleotides O NH2 N O H2O N H cytosine NH3 O HN O uracil HN N H O HOOC O N H N H thymine HOOC NH2 CH2 ¦Â-ureidopropionate CH3 CH2 NH2 CH CH3 O CH2 ¦Â-ureidoN isobutyrate H H2O H2N CH2 CH2 COOH ¦Â-alanine H2O CO2 + NH3 H2N CH2 CH COOH CH3 ¦Â-aminoisobutyrate Highly soluble products Summary of purine biosynthesis dADP dATP AMP ADP ATP GMP GDP GTP dGDP dGTP IMP Summary of pyrimidine biosynthesis dTTP dTMP dTDP dUMP dUDP UMP UDP UTP CDP CTP dCMP dCDP dCTP Summary of Nucleotide Synthesis • Purines built up on ribose – PRPP synthetase: key step – First, synthesis IMP • Pyrimidine rings built, then ribose added – CPS-II: key step – First, synthesis UMP • Salvage is important Points • Synthesis of Purine Nucleotides – De novo synthesis: Site, Characteristics, Element sources of purine bases – Salvage pathway: definition, significance, enzyme, LeschNyhan syndrome – Formation of deoxyribonucleotide: NDP level – Antimetabolites of purine nucleotides: • Purine, Amino acid, and Folic acid analogs • Degradation of Purine Nucleotides – Uric acid, gout • Synthesis of Pyrimidine Nucleotides – De novo synthesis: Characteristics, Element sources of pyrimidine bases – Salvage pathway – Antimetabolites of pyrimidine nucleotides • Catabolism of Pyrimidine Nucleotides