Renal Physiology for Lab
advertisement

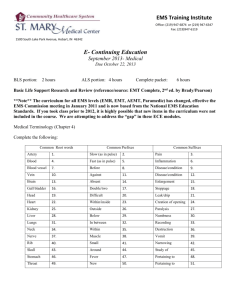
Renal Physiology for Lab Cut out the characters to practice moving them into and out of the tubules. Nutrients are green, electrolytes are yellow, waste is black, hormones and medicines are purple. 1/3 water 2/3 water 2/3 Salt Amino Acids Potassium Water Water Salt Salt Water Water Salt Salt 1/3 Salt Glucose Potassium Phosphate Potassium Magnesium Potassium Magnesium 2 Calcium Calcium Parathyroid hormone Calcium Hormones that regulate K+ Bicarbonate Bicarbonate Bicarbonate Bicarbonate Sodium channel blockers Hydrogen ions Hydrogen ions Hydrogen ions Hydrogen ions Furosemide (Lasix) Urea Urea Urea Aldosterone/ADH Glomerulus Bowman’s capsule Proximal convoluted tubule (Priority City Tunnel) Descending limb (Disneyland) Loop of Henle (tip) (Low Hat Tipping) Thick Ascending Limb (Tall Tales movie theater) Early Distal convoluted tubule (Education Department) Late Distal convoluted tubule (Lucky District with casinos) Collecting duct (Criminal district) This area has the maximally hyperosmotic fluid when ADH levels are high. This area is the site of hormonally regulated potassium Secretes creatinine, antibiotics, diuretics, H+ and uric acid. Macula densa is found here. Tubular fluid here is hyperosmotic to plasma, and is not hormone sensitive Reabsorbs bicarbonate and urea; Secretes K+ This area is impermeable to water unless a hormone like aldosterone or ADH allows it. This area is acted upon by parathyroid hormone. No water is reabsorbed unless ADH or aldosterone is present. No salt is reabsorbed unless aldosterone is present. This area Is sensitive to sodium channel blockers like amiloride and aldosterone inhibitors. Intercalated cells here secrete hydrogen and make “new” bicarbonate. Is sensitive to the diuretic furosemide (Lasix). Capillary hydrostatic pressure is very high here. Reabsorbs salt and H2O water if aldosterone and ADH are present. Secreted into the lumen here is 50% Urea, small amount of sodium and water This area is acted upon by aldosterone and ADH colloid osmotic pressure here is essentially zero Intercalated cells here secrete hydrogen and make “new” bicarbonate. Print this page. Add the yellow labels on the previous page first, then run the pictures on the previous page through the tubule and make them exit and enter at the right locations. Then place the white text boxes in the locations where they belong. 1 Glomerulus 2 Bowman’s capsule 8 Late Distal convoluted tubule 3 Proximal convoluted tubule 7 Early Distal convoluted tubule 9 Collecting duct 6 Thick Ascending Limb 4 Descending limb 5 Loop of Henle (tip) 6 1 Glomerulus Capillary hydrostatic pressure is very high here. 2 Bowman’s capsule colloid osmotic pressure here is essentially zero 3 Proximal convoluted tubule Secretes creatinine, antibiotics, diuretics, H+ and uric acid. 7 6 Thick Ascending Limb Is sensitive to the diuretic furosemide (Lasix). This area is impermeable to water unless a hormone like aldosterone or ADH allows it. 4 Descending limb Secreted into the lumen here is 50% Urea, small amount of sodium and water 5 Loop of Henle (tip) Tubular fluid here is hyperosmotic to plasma, and is not hormone sensitive 8 Late Distal convoluted tubule Reabsorbs salt and H2O if aldosterone and ADH are present. 7 Early Distal convoluted tubule Macula densa is found here. This area is acted upon by aldosterone and ADH This area is acted upon by parathyroid hormone. This area is the site of hormonally regulated potassium Intercalated cells here secrete hydrogen and make “new” bicarbonate. This area Is sensitive to sodium channel blockers like amiloride and aldosterone inhibitors. 9 9 Collecting duct No water is reabsorbed unless ADH or aldosterone is present. No salt is reabsorbed unless aldosterone is present. Reabsorbs bicarbonate and urea; Secretes K+ Intercalated cells here secrete hydrogen and make “new” bicarbonate. This area has the maximally hyperosmotic fluid when ADH levels are high. Imagine It…. • Each of these particles is like a person who works in a factory in Glomerulus City. • They all leave work at 5pm and have to take the same freeway home (the Tubular Freeway). • Each of them get off at different freeway exits. 11 The People THE GOOD NUTRIENTS Water Salt (NaCl) Glucose Amino acids THE GOOD ELECTROLYTES Potassium (K+) Calcium (Ca2+) Magnesium (Mg2+) Phosphate (PO4) Bicarbonate (HCO3-) H+ THE WASTE PRODUCTS Urea Creatinine Antibiotics Diuretics Uric acid THE LAW ENFORCEMENT OFFICIALS Aldosterone ADH Parathyroid hormone Furosemide (Lasix) Hormones that regulate potassium (aldosterone and insulin) Sodium channel blockers like Amiloride (a potassium sparing diuretic) and aldosterone inhibitors 12 The Freeway Exits • Proximal Convoluted Tubule (PCT) – • Descending limb (DL) – • City of Education (this is where the schools are) Late Distal convoluted tubule (LD) – • City of TALL TALES (this is where all the movie theaters are) Early Distal convoluted tubule (ED) – • City of Low Hats (and they TIP their hats to you…very polite!) Thick Ascending Limb (TAL) – • Disneyland! Loop of Henle, tip (LOHT) – • Priority City Tunnel (these people are rich and have a Fast Track pass!) Lucky District (this is where the gambling casinos are) Collecting duct (CD) – The criminal district 13 The Workers and their exits THE GOOD NUTRIENTS – Salt (NaCl) (picture a salt shaker) • PCT (2/3 leaves) • DL (1/3 leaves) • TAL (leaves if aldosterone present) • ED (leaves if aldosterone present) • LD (leaves if aldosterone present) • CD (leaves only if aldosterone is present) – Water (picture a water delivery man) • PCT (2/3 of water exits) • DL (1/3 leaves, but it could enter) • TAL (leaves only if aldosterone or ADH present) • ED (leaves only if aldosterone or ADH present) • LD (leaves only if aldosterone or ADH present) • CD (leaves only if aldosterone or ADH present) – Glucose (picture a lollipop) • PCT – Amino acids (picture a cow) • PCT Leaving the freeway means reabsorption Entering the freeway means secretion 14 The Workers and their exits THE GOOD ELECTROLYTES – Potassium (K+) allows for muscle contraction (picture a body builder) • • • • – PCT (leaves) DL (leaves) LD (enters the freeway here) CD (enters the freeway here) Calcium (Ca2+) for strong bones (picture a skeleton) • • • – PCT (leaves) LOHT (leaves) ED (leaves) Magnesium (Mg2+) for muscle relaxation (picture a massage therapist) • • – PCT (leaves) LOHT (leaves) Phosphate (PO4) for ATP (picture a race car driver) • – PCT (leaves) Bicarbonate (HCO3-) (picture a fire extinguisher) • • • • – TAL (leaves) ED (leaves) LD (leaves) CD (enters or leaves here) H+ (picture an acid-squirting monster) • • • • PCT (enters the freeway here) ED (enters the freeway here) LD (enters the freeway here) CD (enters or leaves here) Leaving the freeway means reabsorption Entering the freeway means secretion 15 The Workers and their exits THE WASTE PRODUCTS – – – – – Urea (a waste product of protein metabolism; picture a Rhea bird, similar to an ostrich) • PCT (leaves) • DL (enters the freeway here) • LD (enters the freeway here) Creatinine (a waste product of protein metabolism) • PCT (enters the freeway here) Antibiotics • PCT (enters the freeway here) Diuretics • PCT (enters the freeway here) Uric acid • PCT (enters the freeway here) Leaving the freeway means reabsorption Entering the freeway means secretion 16 The Workers and their exits HORMONES AND MEDICINES (LAW ENFORCEMENT) – Aldosterone • • • • – TAL (acts on this site) ED (acts on this site) LD (acts on this site) CD (acts on this site) ADH • • • • – TAL (acts on this site) ED (acts on this site) LD (acts on this site) CD (acts on this site) Sodium channel blockers like Amiloride (a potassium sparing diuretic) and aldosterone inhibitors; (City salt monitor…no more salt allowed to leave) • • • • – TAL (acts on this site to prevent sodium from being reabsorbed) ED (acts on this site to prevent sodium from being reabsorbed) LD (acts on this site to prevent sodium from being reabsorbed) CD (acts on this site to prevent sodium from being reabsorbed) Parathyroid hormone (school milk man) • – ED (acts at this site to increase calcium reabsorption) Hormones that regulate potassium (aldosterone and insulin); (school banana monitor) • – ED (acts on this site to cause potassium to either enter or leave) Furosemide (Lasix) • TAL (acts on this site to block water reabsorption) Leaving the freeway means reabsorption Entering the freeway means secretion 17 Acids must be buffered, transported away from cells, and eliminated from the body. These are the most important buffers. Phosphate: important renal tubular buffer HPO4- + H+ H2PO4 Ammonia: important renal tubular buffer NH3 + H+ NH4+ Proteins: important intracellular and plasma buffers H+ + Hb HHb Bicarbonate: most important Extracellular buffer and is also another important renal tubular buffer. H2O + CO2 H2CO3 H+ + HCO3 - Buffering Systems • The three different buffering systems are: 1) Respiratory buffering system • Uses bicarbonate 2) Blood buffering system • Uses bicarbonate, phosphate, and protein 3) Renal buffering system • Uses bicarbonate, phosphate, and ammonia BUFFERING SYSTEMS BUFFERS USED BY THE BUFFERING SYSTEMS Bicarbonate Respiratory System Proteins Bicarbonate Phosphate Blood Bicarbonate Kidneys Phosphate Ammonia pH Imbalances • Acidosis –Can be metabolic or respiratory • Alkalosis –Can be metabolic or respiratory Acidosis • Acidosis is excessive blood acidity caused by an overabundance of acid in the blood or a loss of bicarbonate from the blood (metabolic acidosis), or by a buildup of carbon dioxide in the blood that results from poor lung function or slow breathing (respiratory acidosis). Respiratory acidosis • Respiratory acidosis is due to an accumulation of CO2 in the blood stream. This pushes the carbonic anhydrase reaction to the right, generating H+: carbonic anhydrase H2O + CO2 H2CO3 HCO3(-) + H+ Respiratory acidosis • Cause • The increase in CO2 in the blood is often caused by hypoventilation. • This can be caused by asthma, COPD, and overuse of sedatives, barbiturates, or narcotics such as valium, heroin, or other drugs which make you sleepy. • It can also be caused by other things wrong with the lungs: an accident were the breathing muscles are damaged (causing decreased ventilation), airway obstruction, or lung disease (pneumonia, cystic fibrosis, emphysema, etc.). Respiratory acidosis • Compensation • Even if the peripheral receptors sense the change in pH, the lungs are unresponsive. • The kidneys will compensate by secreting H+. • If H+ excretion cannot restore the balance, the kidneys will also generate bicarbonate. • Since the primary abnormality is an increase in pCO2, the compensatory response is intracellular buffering of hydrogen (by hemoglobin) and renal retention of bicarbonate, which takes several days to occur. Respiratory acidosis • Symptoms • May have no symptoms but usually experience headache, nausea, vomiting, and fatigue. • Breathing becomes deeper and slightly faster (as the body tries to correct the acidosis by expelling more carbon dioxide). • As the acidosis worsens, people begin to feel extremely weak and drowsy and may feel confused and increasingly nauseated. • Eventually, blood pressure can fall, leading to shock, coma, and death. • The most common clinical intervention is IV sodium bicarbonate and applying an oxygen mask. Respiratory acidosis • Treatment • Treatment is aimed at the underlying disease, and may include: • Bronchodilator drugs to reverse some types of airway obstruction • Noninvasive positive-pressure ventilation (sometimes called CPAP or BiPAP) or a breathing machine, if needed • Oxygen if the blood oxygen level is low • Treatment to stop smoking Metabolic acidosis • Metabolic acidosis is the gain of acid or the loss of bicarbonate. • Cause • Usual causes are the generation of ketone bodies in uncontrolled diabetes mellitus, diarrhea (loss of bicarbonate), excess protein consumption (breakdown products are amino ACIDS), or excess alcohol consumption: (alcohol formaldehyde acetic acid). • Can also be caused by ingestion of an acid (aspirin, ethanol, or antifreeze). • Exercise creates a milder, transient metabolic acidosis because of the production of lactic acid. Metabolic acidosis • Compensation • The body will compensate with hyperventilation and increased bicarbonate reabsorption in the kidney. • Since the primary abnormality is a decrease in HCO3, the compensatory response includes extracellular buffering (by bicarbonate), intracellular buffering (by phosphate and proteins), respiratory compensation and renal hydrogen excretion. • Metabolic acidosis stimulates an increase in ventilation (reducing pCO2). • This hyperventilation is called Kussmaul's respiration. Metabolic acidosis • Symptoms • Most symptoms are caused by the underlying disease or condition that is causing the metabolic acidosis. • Metabolic acidosis itself usually causes rapid breathing. • Confusion or lethargy may also occur. • Severe metabolic acidosis can lead to shock or death. • In some situations, metabolic acidosis can be a mild, chronic (ongoing) condition. Metabolic acidosis • Treatment is give i.v. of sodium bicarbonate. • The HCO3- deficit can be calculated by using the following equation: • HCO3- deficit = deficit/L (desired serum HCO3- - measured HCO3-) x 0.5 x body weight (volume of distribution for HCO3-) • This provides a crude estimate of the amount of HCO3that must be administered to correct the metabolic acidosis; the serum HCO3- level or pH should be reassessed frequently. Alkalosis • Alkalosis is excessive blood alkalinity caused by an overabundance of bicarbonate in the blood or a loss of acid from the blood (metabolic alkalosis), or by a low level of carbon dioxide in the blood that results from rapid or deep breathing (respiratory alkalosis). Respiratory alkalosis • Respiratory alkalosis is generally caused by hyperventilation, usually due to anxiety. The primary abnormality is a decreased pCO2. • Cause • Caused from a decrease in CO2 in the blood because the lungs are hyperventilating (anxiety, but not panting). • Fever or aspirin toxicity may also cause respiratory alkalosis. Respiratory alkalosis • Compensation • The body will reduce the breathing rate if the respiratory problem is from anxiety. • The kidney will excrete bicarbonate. • The kidney will also stop excreting H+, and may also reabsorb more H+. Respiratory alkalosis • Compensation • The compensatory response to a respiratory alkalosis is initially a release of hydrogen from extracellular and intracellular buffers. • This is followed by reduced hydrogen excretion by the kidneys. • This results in decreased plasma bicarbonates. • In chronic respiratory alkalosis, compensatory measures can make the pH revert to normal. Respiratory alkalosis • • • • Symptoms Irritability Muscle twitching Muscle cramps Respiratory alkalosis • Treatment • Treatment for hyperventilation is to breathe into a paper bag for a while, as the person breathes carbon dioxide back in after breathing it out. • For severe cases, need to replace the water and electrolytes (sodium and potassium). Metabolic alkalosis • Metabolic alkalosis is due to the gain of base or the loss of acid. The primary abnormality is having an increased HCO3 seen in the blood test. • Cause • Caused from an increase in bicarbonate in the blood because of ingestion of excess bicarbonate in the form of an antacid (Tums), eating excess fruits (vegetarian diets and fad diets*), loss of acid from vomiting, or loss of potassium from diuretics. Metabolic alkalosis • Compensation • This is initially buffered by hydrogen buffers (such as plasma proteins and lactate). • Chemoreceptors in the respiratory center sense the alkalosis and trigger hypoventilation, resulting in increased pCO2. • The respiratory system will hypoventilate but this will not be effective because CO2 will accumulate and the CO2 receptors will override the pH receptors. Metabolic alkalosis • Compensation • Naturally, the extent of respiratory compensation will be limited by the development of hypoxia with continued hypoventilation. The kidney will make more of a difference by not reabsorbing bicarbonate. • In addition to respiratory compensation, the kidneys excrete the excess bicarbonate. However, this takes several days to occur. Metabolic alkalosis • • • • • • • • Symptoms Confusion (can progress to stupor or coma) Hand tremor Light-headedness Muscle twitching Nausea, vomiting Numbness or tingling in the face, hands, or feet Prolonged muscle spasms (tetany) Metabolic alkalosis • Treatment is to give an anti-emetic if the problem is from vomiting. If not, give an i.v. of normal saline to increase the blood volume. • If potassium is also low, would have to add that to the i.v. Interpreting Arterial Blood Gases (ABG) • This blood test is from arterial blood, usually from the radial artery. • There are three critical questions to keep in mind when attempting to interpret arterial blood gases (ABGs). First Question: Does the patient exhibit acidosis or alkalosis? Second Question: What is the primary problem? Metabolic? or Respiratory? Third Question: Is the patient exhibiting a compensatory state? Assessment Step 1 • Step One: Determine the acid/base status of the arterial blood. • If the blood's pH is less than 7.35 this is an acidosis, and if it is greater than 7.45 this is an alkalosis. You may hear nurses or doctors say: "The patient is 'acidotic' or 'alkalotic' • If pH is normal, use the patient history to determine if it is respiratory or metabolic. To figure out if it is alkalosis or acidosis, check the table. Know the normal ranges for pH ACID BASE PARAMETERS (The problem chemical is in yellow) Respiratory Acidosis PH Respiratory Alkalosis PH PCO2 HCO3 If compensating Or normal if not compensating PCO2 HCO3 If compensating Or normal if not compensating Metabolic Acidosis PH Metabolic Alkalosis PH PCO2 HCO3 If compensating Or normal if not compensating PCO2 If compensating Or normal if not compensating HCO3 Assessment Step 2 • Once you have determined the pH, you can move on to determine which system is the 'primary' problem: respiratory or metabolic. • To do this, examine the pCO2 and HCO3 levels. • If only the pCO2 is abnormal, it is a respiratory problem. • If only the HCO3 is abnormal, it is a metabolic problem. • If both are abnormal, they are either both high or both low. That means they are compensating. One will never be high and the other low. Check the table to see if it is respiratory or metabolic. Assessment Step 3 • Determine if the body is attempting to compensate for the imbalance or not. • If they are compensating, both CO2 and HCO3 will be high, or both will be low. To know if they have fully compensated, see if pH has returned to normal or not. Review the three essential steps of ABG analysis • Number One: Determine if the patient is demonstrating an acidotic (remember: pH less than 7.35) or alkalotic (pH greater than 7.45). • Number Two: • What is the 'primary problem? • If the patient is acidotic with a pC02 greater than 45 mmHg it is RESPIRATORY • If the patient is alkalotic with a pC02 less than 35 mmHg it is RESPIRATORY • If the patient is acidotic with a HC03 less than 22 mEq/L it is METABOLIC • If the patient is alkalotic with a HC03 greater than 26 mEq/L it is METABOLIC Know the normal ranges for pCO2 and HCO3 Review the three essential steps of ABG analysis • Number Three: Is the patient compensating? • Are both components (HCO3 and pCO2) shifting in the same direction? • Up or down the continuum? • Above or below the normal ranges? • If this is noted, you know that the patient’s buffering systems are functioning and are trying to bring the acidbase balance back to normal. http://www.wikihow.com/Interpret-Blood-Gas-Results Arterial Blood Gas problems when compensation is present pH Respiratory Acidosis Acid Metabolic Alkalosis Base Metabolic Acidosis Acid Respiratory Alkalosis Base PCO2 HCO3 59 Condition pH Resp CO2 Bicarb Compensating? Resp acidosis Low Hypoventilating High High Yes Resp acidosis Low Hypoventilating High Norm No Resp alkalosis High Hyperventilating Low Low Yes Resp alkalosis High Hyperventilating Low Norm No Metab acidosis Low Normal Low Low Yes Metab acidosis Low Normal High Norm No Metab Alkalosis High Normal High high Yes Metab Alkalosis High Normal Low Norm No Kussmaul Breathing • Kussmaul breathing is a form of hyperventilation often associated with severe metabolic acidosis, particularly diabetic ketoacidosis (DKA) but also renal failure. Case Study 1 A patient recovering from surgery in the post-anesthesia care unit is difficult to arouse two hours following surgery. The nurse in the PACU has been administering Morphine Sulfate intravenously to the patient for complaints of post-surgical pain. The patient’s respiratory rate is 7 per minute and demonstrates shallow breathing. The patient does not respond to any stimuli! The nurse assesses the ABCs (remember Airway, Breathing, Circulation!) and obtains ABGs STAT! The STAT results come back from the laboratory and show: pH = 7.15 (low) C02 = 68 mmHg (high) HC03 = 22 mEq/L (normal) 1. 2. 3. 4. Compensated Respiratory Acidosis Uncompensated Metabolic Acidosis Compensated Metabolic Alkalosis Uncompensated Respiratory Acidosis Answer • The answer is #4 uncompensated respiratory acidosis Case Study 2 • An infant, three weeks old, is admitted to the Emergency Room. The mother reports that the infant has been irritable, difficult to breastfeed and has had diarrhea for the past 4 days. The infant’s respiratory rate is elevated and the fontanels are sunken. The Emergency Room physician orders ABGs after assessing the ABCs. • The results from the ABGs come back from the laboratory and show: pH = 7.37 (normal) C02 = 29 mmHg (low) HC03 = 17 mEq/L (low) 1. 2. 3 4 Compensated Respiratory Alkalosis Uncompensated Metabolic Acidosis Compensated Metabolic Acidosis Uncompensated Respiratory Acidosis Answer • Answer is #3 • Compensated Metabolic Acidosis Case Study 3 • • 1. 2. 3. 4. A patient, 5 days post-abdominal surgery, has a nasogastric tube. The nurse notes that the nasogastric tube (NGT) is draining a large amount (900 cc in 2 hours) of coffee ground secretions. The patient is not oriented to person, place, or time. The nurse contacts the attending physician and STAT ABGs are ordered. The results from the ABGs come back from the laboratory and show: pH = 7.52 (high) C02 = 35 mmHg (normal) HC03 = 29 mEq/L (high) Compensated Respiratory Alkalosis Uncompensated Metabolic Acidosis Compensated Metabolic Acidosis Uncompensated Metabolic Alkalosis Answer • • Answer is #4 Uncompensated Metabolic Alkalosis Case Study 4 • A patient is admitted to the hospital and is being prepared for a craniotomy (brain surgery). The patient is very anxious and scared of the impending surgery. He begins to hyperventilate and becomes very dizzy. The patient looses consciousness and the STAT ABGs reveal: • • • • The results from the ABGs come back from the laboratory and show: pH = 7.57 (high) C02 = 26 mmHg (low) HC03 = 24 mEq/L (normal) 1. 2. 3. 4. Compensated Metabolic Acidosis Uncompensated Metabolic Acidosis Uncompensated Respiratory Alkalosis Uncompensated Respiratory Acidosis Answer • • The answer is #3 Uncompensated Respiratory Alkalosis Case Study 5 • A two-year-old is admitted to the hospital with a diagnosis of asthma and respiratory distress syndrome. The father of the infant reports to the nurse that he has observed slight tremors and behavioral changes in his child over the past three days. The attending physician orders routine ABGs following an assessment of the ABCs. The ABG results are: • • • pH = 7.36 (normal) C02 = 69 mmHg (high) HC03 = 36 mEq/L (high) 1. 2. 3. 4. Compensated Respiratory Alkalosis Uncompensated Metabolic Acidosis Compensated Respiratory Acidosis Uncompensated Respiratory Alkalosis Answer • Answer is #3 • Compensated Respiratory Acidosis Case Study 6 • • 1. 2. 3. 4. A young woman, drinking beer at a party, falls and hits her head on the ground. A friend dials "911" because the young woman is unconscious, depressed ventilation (shallow and slow respirations), rapid heart rate, and is profusely bleeding from both ears. Which primary acid-base imbalance is this young woman at risk for if medical attention is not provided? metabolic acidosis metabolic alkalosis respiratory acidosis respiratory alkalosis Answer • Correct answer is #3 • Respiratory Acidosis Case Study 7 • • 1. 2. 3. 4. An 11-year old boy is admitted to the hospital with vomiting (losing acid!), nausea and overall weakness. The nurse notes the laboratory results: potassium: 2.9 mEq (low). Which primary acid-base imbalance is this boy at risk for if medical attention is not provided? Note: Potassium makes blood more acidic. metabolic acidosis metabolic alkalosis respiratory acidosis respiratory alkalosis Answer • Correct Answer is #2 • Metabolic Alkalosis Case Study 8 • An elderly gentleman is seen in the emergency department at a community hospital. He admits to taking many tablets of aspirin (salicylates) over the last 24-hour period because of a severe headache. He complains of an inability to urinate. His vital signs are: Temp = 98.5; apical pulse = 92; respiration = 30 and deep. • Which primary acid-base imbalance is the gentleman at risk for if medical attention is not provided? 1. 2. 3. 4. metabolic acidosis metabolic alkalosis respiratory acidosis respiratory alkalosis Answer • Correct Answer is #1 • Metabolic Acidosis Case Study 9 • • 1. 2. 3. 4. A young man is found at the scene of an automobile accident in a state of emotional distress. He tells the paramedics that he feels dizzy, tingling in his fingertips, and does not remember what happened to his car. Respiratory rate is rapid at 34/minute. Which primary acid-base disturbance is the young man at risk for if medical attention is not provided? metabolic acidosis metabolic alkalosis respiratory acidosis respiratory alkalosis Answer • Correct Answer is #4 • Respiratory Alkalosis Case Study 10 12 year old diabetic presents with Kussmaul breathing • • • • pH : 7.05 (low) pCO2: 12 mmHg (very low) pO2: 108 mmHg (normal) HCO3: 5 mEq/L (low) – What is the diagnosis? Is he compensating? What caused the problem? Answer 12 year old diabetic presents with Kussmaul breathing • • • • pH : 7.05 (low) pCO2: 12 mmHg (very low) pO2: 108 mmHg (normal) HCO3: 5 mEq/L (low) – Compensating metabolic acidosis without hypoxemia due to ketoacidosis Case Study 11 17 year old w/severe kyphoscoliosis, admitted for pneumonia. Elevated respiratory rate. • • • • pH: pCO2: pO2: HCO3: 7.37 (normal) 25 mmHg (low) 60 mmHg (low) 14 mEq/L (low) – What is the diagnosis? Is he compensating? What caused the problem? Case Study 11 17 year old w/severe kyphoscoliosis, admitted for pneumonia. Elevated respiratory rate. • • • • pH: pCO2: pO2: HCO3: 7.37 (normal) 25 mmHg (low) 60 mmHg (low) 14 mEq/L (low) – Compensated respiratory alkalosis due to chronic hyperventilation secondary to hypoxia Case Study 12 9 year old w/hx of asthma, audibly wheezing x 1 week, has not slept in 2 nights; presents sitting up and using accessory muscles to breathe w/audible wheezes • • • • pH: pCO2: pO2 HCO3: 7.51 (high) 25 mmHg (low) 35 mmHg (very low) 22 mEq/L (normal) – What is the diagnosis? Is he compensating? What caused the problem? Case Study 12 9 year old w/hx of asthma, audibly wheezing x 1 week, has not slept in 2 nights; presents sitting up and using accessory muscles to breathe w/audible wheezes • • • • pH: pCO2: pO2 HCO3: 7.51 (high) 25 mmHg (low) 35 mmHg (very low) 22 mEq/L (normal) – Uncompensated respiratory alkalosis with severe hypoxia due to asthma exacerbation Case Study 13 7 year old post-op presenting with chills, fever and hypotension • • • • pH: pCO2: pO2: HCO3: 7.25 (low) 36 mmHg (low) 55 mmHg (low) 10 mEq/L (low) – What is the diagnosis? Is he compensating? What caused the problem? Case Study 13 7 year old post-op presenting with chills, fever and hypotension • • • • pH: pCO2: pO2: HCO3: 7.25 (low) 36 mmHg (normal) 55 mmHg (low) 10 mEq/L (low) – Uncompensated metabolic acidosis due to low perfusion state and hypoxia causing increased lactic acid Urinalysis Lab • 1. What would most likely be the cause of a urine sample with a positive test for nitrites, leukocytes and a slightly higher than normal pH? Urinary tract infection • 2. Are proteins or blood normally found in urine? If they are present, how might they get there? No; kidney infection, glomerulonephritis, trauma, kidney stones • 3. If the urine sample tests positive for ketones and glucose, for what disease should the patient be checked? Diabetes mellitus • 4. Elevated levels of urobilinogen and bilirubin may indicate problems with what organ? Liver • • 5. What dietary habits may cause an acidic urine sample (more acidic than normal)? High proteins or acidic foods • 6. What would cause a basic urine sample? vegetarian diet Urinalysis Lab 7. Normal urine is usually yellow to amber in color, due to the presence of what pigment? Urochrome 8. What blood protein (like all other proteins) is too large to pass through the glomerular filtration membrane, and so is not normally found in the urine? Albumin 9. Circle the correct term: Hematuria / Ketonuria, the appearance of red blood cells in the urine, indicates pathology. 10. The appearance of bile pigments in the urine is known as __________________, and indicates liver disease. Bilirubinuria 11. Circle the correct term: Casts / Caliculi are hardened cell fragments formed in the DCT and collecting ducts and flushed out of the urinary tract. 12. When determining the presence of inorganic constituents such as sulfates, phosphates, and chlorides, you look for the formation of a precipitate. What is a precipitate? A cloudy solid that forms Urinalysis