Theme of the lecture
advertisement

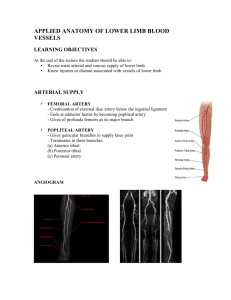
TASHKENT MEDICAL ACADEMY DEPARTMENT OF FACULTY AND HOSPITAL SURGERY OF MEDICAL FACULTY "CONFIRM" Vice Rector of TMA Professor Teshaev O.R. _______________________ «27» august 2015 y. Theme of the lecture: VARICOSE DISEASE For the students of V course of the treatment faculty Tashkent – 2015 1 TASHKENT MEDICAL ACADEMY DEPARTMENT OF FACULTY AND HOSPITAL SURGERY OF MEDICAL FACULTY "CONFIRM" Dean of the medical faculty Professor Zufarov P.S. _______________________ «27» august 2015 y. Theme of the lecture: VARICOSE DISEASE For the students of V course of the treatment faculty Considered and approved on surgical meeting section of CPC Protocol number 1 of «27» august 2015 y. Tashkent – 2015 2 Varicose disease TECHNOLOGY of TRAINING Number of student – 60-120 Time – 90 min. Form of lesson Lecture - Visualization Plan of the lecture 1. Anatomy and blood flow through the veins of the lower extremities. 2. Research methods veins. 3. Etiology, pathogenesis, classification, clinical features, diagnosis, differential diagnosis, treatment and prevention of varicose veins. 4. Complications of varicose disease, methods of treatment. The purpose of the training session: To introduce students with varicose disease, teach them to clinical features, course, complicated forms, differential diagnosis, optimal treatment, postoperative care, rehabilitation patients. Task of the teacher: Learning outcomes: 1. Introduce the etiology and pathogenesis Student should master: of varicose diseases. 1. Clinical anatomy of veins and the 2. Writing a modern classification of the reason of current of blood on veins. disease. 2. Etiology, pathogenesis, 3. Cause methods of diagnostics of classification of varicose disease. disease, modern treatment options and the 3. Diagnostics of varicose disease. methods of prevention. 4. Principles of treatment, methods of 4. Introduce the complications of the fight against complications of varicose disease, methods of treatment. disease. 5. Principles of prevention of a disease. Methods and techniques of trained Lecture - Imaging Technology: quiz, focusing questions, the technique of "yes-no". Learning tools Laser projector, visual materials, information technology. Forms of learning Collective. Conditions of learning Audience adapted to work with the audio video equipment. 3 TECHNOLOGY CARD OF THE LECTURE Stages, time Activities teachers students Stage 1 1. Notifies the theme, purpose, 1. Listen. Introduction expected results and lectures of his (5 min) plan/ 2.1. In order to update the 2.1. Respond to questions Stage 2 Actualization of knowledge of the students asks questions focusing repeat knowledge knowledge of anatomy, normal and (20 min) pathological physiology. Conducts blitz poll. 2.2. Slide show and comments. 2.2. Contains a study of the slides. Stage 3 3.1. Consistently presents the 3.1. Discuss the content of Information material lectures on plan uses visual the proposed materials, (55 min) materials with incremental clarify, ask questions. responses to questions focusing. Focuses on key topics, offers to Write down the main write them down. points. Stage 4 4.1. Asks a series of questions to 4.1. Respond to questions. Final secure the material using certain (10 min) clinical situations. 4.2. Gives a task for independent 4.2. Listen, write. work. 4 Justification of the theme: Venous disorders are important. They range in severity from trivial telangiectasias to death-dealing pulmonary embolization. Their impact on disability is enormous. Their cost to the economic system in Western countries is almost incalculable. This volume intends to bring under a single hardcover the major facets that have changed the face of diagnosis and treatment of venous disorders during the last quarter century. Much of the activity in diagnosis and treatment of venous disorders actually has been in investigations toward causation. These are well represented in this volume, where the molecular basis of venous insufficiency is discussed as well as in the inappropriate activity of leukocytes and how this impacts on venous function. This is discussed further in the chapters on physiologic testing of venous disorders. All of this, of course, is based on the anatomical findings and the newly uncovered pathologic anatomy revealed by ultrasound testing. Classification of venous dysfunction has undergone some revisions and has helped in making treatment of venous insufficiency more of a science. The fundamental basis for all treatment in venous disorders is compression therapy, and this is thoroughly discussed in this volume by a preeminent authority in this field. The purpose of the lecture: To introduce students with varicose disease, teach them to clinical features, course, complicated forms, differential diagnosis, optimal treatment, postoperative care, rehabilitation patients. Educational objectives of the lecture: Instilling students need adequate timely development operations to severe complications, and in their development familiarity with the most informative and modern methods of diagnosis and surgical treatment of patients, familiarity with possible complications out of operation and operational period, their, prevention. Clinical development of students thinking. Developing a modern look at the problem from the perspective of the global issue of medicine and general practice. Objectives of the lecture: Writing the concept of varicose diseases, explain the causes and mechanisms of development of complications to clinical characteristics and possible options for the disease, differential diagnosis with other diseases, to acquaint students with modern and most informative methods of examination of patients. Issues to be parsed with the time allotted to the question: 1. Motivation, rationale topic – 5 min. 2. Brief anatomical and physiological data, methods of examination of patients – 20 min. 3. Etiopathogenesis of varicose diseases – 10 min. 4. Clinical picture – 20 min. 5. Diagnosis and differential diagnosis – 10 min. 6. Treatment – 15 min. 7. Prevention of disease – 10 min. 5 INTRODUCTION It is generally agreed that varicose veins afflict from 40 to 60% of women and 15 to 30% of men. Therefore, it is surprising that the etiology and development of this common problem remain obscure. Actually, in Western populations the incidence of varicose veins varies with the definition applied. Most investigators favor the definition of Arnoldi, who said that varicosities are “any dilated, elongated, or tortuous veins, irrespective of size.” Thus, some epidemiologic studies would include telangiectasias and reticular veins, while others would exclude these entities. The definition of Arnoldi is particularly useful because it presents a unifying concept for reticular varicosities, telangiectasias, and major varicose veins. Since all three are elongated, dilated, and have incompetent valves, they probably have a common origin and respond to the same physical forces and acquired influences. The dilation and elongation implies that these abnormal veins have been responsive to effects of pressure. The causes and results of venous valve dysfunction are more subtle. They might be explained simply by the dilation of a vein and valve annulus, which stretches beyond the capability of its leaflets to close together. However, such a simplistic view does not explain the disappearance, perforation, and splitting of valves noted by several authorities or the stunted valves observed angioscopically in refluxing vein segments. Dodd and Cockett defined varicose veins, saying “a varicose vein is one which has permanently lost its valvular efficiency.” They further explained varicosities by suggesting that it was pressure over a course of time that causes a varix to become elongated, tortuous, pouched, and thickened. Venous pressure appears inextricably linked to development of venous insufficiency. In assessing the etiology and pathogenesis of varicose veins, it would be best to accept the definition of Arnoldi and search for the factor or factors that best fit research observations and clinical experience. It is the purpose of this presentation to offer evidence for a common factor of causation and development of the three forms of primary venous insufficiency and then suggest proper intervention based upon this etiology. RISK FACTORS: THE PATHOLOGIC SUBSTRATE In the mind of the lay public, heredity is the single most important factor in causation of varicose veins. Yet evidence for the role of genetic predisposition in development of varicose veins is limited. There are many methodologic difficulties in interpreting available data. However, Cornu-Theґnard and colleagues performed a very carefully planned study, which presents useful conclusions. Their investigations were based on prospective evaluation of 67 patients with varicose veins compared to 67 controls. The patients, controls, and parents of each study group were all examined for varicose stigmata. This provided a total of 402 subjects. This study demonstrated a prominent role for hereditary in development of varicose veins. If both parents had varicose veins, the children had a 90% chance 6 for developing varicosities. When one parent was affected, the risk was 25% for males and 62% for females. This contrasted with a risk of only 20% when neither parent was afflicted with varicose veins. Interestingly, in this study both the total number of pregnancies and the presence of constipation were found to influence development and aggravation of varicosities. Although a third generation of individuals was not examined, an autosomal recessive type of inheritance with variable penetrance would be compatible with the results observed. A dominant mode of transmission has been discussed by many authors, as has crossed transmission, but there is little evidence for either of these. It is generally agreed that varicose veins may appear for the first time during pregnancy and may be exacerbated by pregnancies. The widely held view is that varicose veins are caused by pressure of the gravid uterus, which obstructs venous return. That concept has been refuted by the observation that a majority of varices appear during the first 3 months of pregnancy when the uterus is not large enough to cause mechanical obstruction. Multiparous women frequently note that the first sign of another pregnancy is the sudden appearance of a new cluster of varicosities or a major telangiectatic blemish. This could be explained by an hormonal effect due to the profound elaboration of progesterone by the corpus luteum. Progesterone inhibits smooth-muscle contraction in the uterus and vein wall. While that is logical, the physiologic venous function study performed by Struckmann’s group failed to find a statistical correlation between venous muscle pump function and changes in hormone concentrations of estradiol, estriol, or progesterone. These authors felt that venous insufficiency developing during pregnancy was caused primarily by mechanical obstruction or other causes not related to the hormone studied. Also, the immunocytochemical investigations of Sadick and Niedt failed to find estrogen or progesterone receptors in lower extremity telangiectasias. Despite that negative evidence, intuitively one would implicate progesterone as an etiologic factor in the development of varicose veins. As progesterone does inhibit smooth-muscle contraction, it could have a profound down regulating effect on function of the smooth muscle of the venous wall. Miller et al. speculated on hormonal effects on venous function, saying that the excitatory effects of some agents might be more prominent when estrogen and progesterone are elevated, as would be found during some phases of the menstrual cycle. Vin and coworkers established that functional venous symptomatology is aggravated upon the appearance of new varicose veins or blue telangiectatic veins during the course of progestogen or estroprogestogen treatment with oral contraceptives. Therefore, heredity and female hormones, principally progesterone, can be thought of as the substrate for action by physical factors that elongate and dilate superficial veins. INTRINSIC FACTORS Among the theories that have been proposed to explain the cause of varicose veins is the hypothesis invoking a weakness in the vein wall. A significantly reduced vein wall elasticity has suggested that the role of venous valves in 7 development of varicose veins is secondary to changes in the elastic properties of the vein wall. Such abnormality of vein wall function could very well be the result of progesterone effectively inhibiting smooth-muscle contraction. In vitro evaluation of varicose veins has shown that maximal contractions in response to stimulation by potassium chloride, norepinephrine, and endothelin are reduced compared to normal control veins. Endothelium-dependent relaxations were also attenuated, which suggests that endothelial and smoothmuscle function is impaired in vein segments removed from patients with primary varicose veins. Thulesius et al. showed a functional disturbance of smooth-muscle function in varicose veins. Therefore, the combination of venous wall abnormalities and hormonal effect could be acting interdependently. Scanning electron microscopy has shown varying degrees of thinning of the media of the varicose venous wall. These areas of thinning coincide with areas of varicose dilation. Replacement of smooth muscle by collagen is also a characteristic of varicose veins. Interestingly, the collagen content and wall area is greater proximally compared to distally in varicose saphenous veins compared to controls. There is a higher content of smooth muscle and elastin in varicose veins proximally as compared to distally. This suggested to Travers and his group that varicose veins were a dynamic response to venous hypertension rather than a passive thinning of venous wall components, as has been postulated by others. Our own work in this area has been based on the hypothesis that both the venous valve and the venous wall are affected by the elements that cause varicose veins. We and others have observed absence of the subterminal valve in saphenous veins in patients with venous varicosities requiring surgical intervention. As mentioned earlier, perforation, splitting, and atrophy of venous valves has been seen during intraoperative saphenous angioscopy, and angioscopy has revealed that approximately 50% of varicose saphenous veins have no functional valves between the groin and the knee. Other direct observations have shown that a widening of the space between valve leaflets is the first and most common cause of reflux in venous varicosities. These observations led us to explore the possible role of leukocyte infiltration of venous valves and the venous wall in genesis of varicosities. Our investigations identified the presence of leukocytes in the venous valves, in the venous wall, and in the base of the valve leaflet. CD-64 monoclonal antibody staining revealed their precise identification as monocytes that become macrophages on penetration into the wall. These were found in greater numbers on the proximal side of the valve cusp and in the proximal venous wall than on the distal surface of the valve and distal venous wall and were associated with increased ICAM-1 expression. But the greatest number of tissue macrophages were seen in the base of the valve. Liberation of toxic products by the monocyte could very well explain both venous wall weakness and venous valvular destruction. If true, this would suggest that vein wall weakening and valve destruction are occurring simultaneously. 8 Why obesity would be associated with varicose veins is unclear. Nevertheless, in case-controlled studies, obesity defined as greater than 20% over ideal weight was much more common in patients with truncal varicosities and telangiectasias than in controls. Even population-based studies have shown a positive relationship between body mass and varicose veins. Multivariant analyses, including weight and height, have revealed weight to be significantly related to occurrence of varicose veins in women but not in men. Central obesity, often referred to as android obesity, is more dangerous than peripheral or gynoid obesity. This is believed to be secondary to increased fat metabolism which leads to hyperglycemia, diabetes, insulin-induced sodium reabsorption, and hypertension. These factors together are known as syndrome X, and it has been found that this type of obesity is associated with a marked increase in intraabdominal pressure. Urinary bladder pressure can be used as an estimate of intraabdominal pressure, and such pressures plotted against abdominal diameter display a straight-line relationship with an r value of +0.67. For decades, a standing occupation has been implicated in causation of varicose veins, yet it is only with development of duplex scanning that this has been proven to be true. In a study of 387 male workers with a standing occupation, venous insufficiency was present in 29% of the individuals and correlated with age, weight, and duration of standing work. Of some interest to surgeons was examination of the distribution and extent of venous reflux in the lower extremities of vascular surgeons as compared to volunteers. Vascular surgeons were chosen because they are typically thought to lead a life of prolonged standing. In symptom-free vascular surgeons, there was greater superficial venous reflux than in controls. Even deep venous reflux and perforator incompetence was greater in the vascular surgeons. Fortunately, in other studies, such changes in reflux have been reversed by physical conditioning. This has been shown to improve venous hemodynamics. In contrast to that improvement in venous function during physical therapy has been the observation that deterioration of venous function is seen while patients await corrective surgery. Other risk factors for varicosities include the wearing of tight undergarments, which presumably produce proximal limb venous hypertension, a low-fiber diet, which supposedly predisposes to constipation and increased abdominal straining, and raised toilet seats, which prevent squatting during defecation. Similarly, the Western habit of chair sitting rather than squatting during resting has been thought by some to increase hydrostatic pressure and blood pooling in the legs. All of these theoretical and unproven theories are related to venous hypertension, which itself is linked to development of venous insufficiency. PHYSICAL FORCES It is acknowledged that the hydrostatic weight of the column of blood transmitted from the level of the right atrium through valveless vena cava and iliac 9 veins is exerted directly on the femoral vein and the saphenofemoral junction. This hydrostatic pressure can easily be increased by intraabdominal pressure as implied in theories which link constipation, low-fiber diet, and raised toilet seats to development of varicosities. An increasing body of evidence suggests that fluid shear stress serves as a mechanism to control inflammatory reactions in endothelial cells and circulating leukocytes. A shift of fluid shear stresses from normal physiological levels as well as mechanical stretching of the venous wall or the leaflet tissue may serve to promote inflammatory gene products. Alterations of venous or valve leaflet tissue stresses may serve as mechanisms that promote the inflammatory state and the high propensity for venous valve failure. The process may be further enhanced in the presence of humoral inflammatory mediators which serve to suppress the antiinflammatory reaction of normal physiological fluid shear stresses. If such venous hypertension were to be the stimulus for activation of leukocytes, such activation would introduce a cascade of events. Leukocytes would roll rather than flow through the venous stream and would adhere to venous endothelium. Such activation and adherence producing leukocyte-endothelial interaction could explain the presence of monocytes on the endothelium in the valves and in the venous wall in specimens removed at varicose vein surgery. It is but a short step in thought to ascribe valve destruction and vein wall thinning and weakening to such monocyte infiltration. Thus, hydrostatic forces are easily linked to development of varicosities. But more subtle is the effect of hydrodynamic forces. These derive from muscular contraction and can be transmitted through failed perforator vein valves to unsupported subcutaneous and intradermal venulectasias. According to Faria and Moraes, telangiectasias were first thought to be associated with elevated venous pressure by Meischer in 1919. Faria showed that telangiectasias are linked to varicose veins. His observations support the view that telangiectasias, like varicose veins, are related to high venous pressure. Using injection of radiographic contrast media into telangiectasias, Moraes et al. showed direct connections of the telangiectasias with named veins such as the greater saphenous and other superficial veins. Communicating veins directly connecting the telangiectasias to the saphenofemoral junction were also demonstrated, but the most important observations were that telangiectasias were directly connected to perforating veins. Thus, even telangiectasias and venulectasias were shown to be connected to the two most important sources of venous hypertension. The contrast studies allowed visualization of the deep venous system in 61 of 74 limbs. The greatest number of perforating veins were found between 15 and 35 cm below the greater trochanter, where 102 of the total of 142 perforating veins were identified. It should be emphasized that contraction of muscles within their muscular compartments causes tremendous elevation of intracompartmental pressure and that these elevations are transmitted through failed check valves in the perforating veins. These direct demonstrations of interconnections of intradermal venous abnormalities with venous hypertension 10 support the theory that telangiectasias and varicose veins are pathologically identical and only differ in size. Muscular compartmental forces during exercise are enormous. These range between 100 and 300 mmHg. The effects of these pressures on weakened vein walls would be to further dilate and elongate the affected conduits. Further, the dilation of the venous wall could easily render the contained valve incompetent. SUMMARY OF ETIOLOGY A unifying concept that links telangiectasias, reticular varicosities, and varicose veins to hereditary and hormonal factors is intriguing. On a hereditaryhormonal substrate, the factors of hydrostatic and hydrodynamic forces interact to affect vein valves and vein walls weakened by unknown factors, which remain to be discovered. The etiology of primary varicose veins may be diverse, but it can be simplified. First, elongated, dilated, and tortuous veins with incompetent valves, regardless of size, are varicose, and this definition includes telangiectasias, reticular varicosities, and varicose veins. Next, a hereditary predisposition acted upon by female hormones and venous hypertension may produce the pathologic changes. Venous hypertension is the sum of hydrostatic pressure caused by gravity and hydrodynamic forces from muscular compartment pressure and intraabdominal straining. INDICATIONS FOR INTERVENTION Indications for intervention in primary venous insufficiency are listed in Table 67-2. Often it is the appearance of telangiectatic blemishes or protuberant varicosities that stimulates consultation. Ultimately, this may be the only indication for intervention. Unfortunately, negative physician perception regarding availability and efficacy of treatment of varices may deny the patient the precise care which is sought. Furthermore, symptoms of primary venous insufficiency may be present but not recognized by the patient until asked for during thorough history taking. The characteristic symptoms include aching pain, easy leg fatigue, and leg heaviness, all worsening as the day progresses so that the patient sits down in the afternoon and elevates the legs for some relief. These symptoms will be maximal on the first day of a menstrual period. The symptoms may not be recognized by the patient as being due to the varicose veins or telangiectasias. They must be asked for by the examining physician. Neither the patient nor the physician may understand that these symptoms arise from telangiectatic blemishes just as from venous varicosities. However, this is true. Some 50% of patients with telangiectasias will have such symptoms, and 85% will be relieved of them by appropriate therapy. Other indications for intervention for venous varicosities include superficial thrombophlebitis in varicose clusters, external bleeding from high-pressure venous blebs, or advanced changes of chronic venous insufficiency. These include the 11 severe ankle hyperpigmentation, subcutaneous lipodermatosclerosis, atrophie blanche, or frank ulceration of chronic venous insufficiency (CVI). Specific findings on physical examination which lead toward groin-to-knee stripping rather than phlebectomy include the finding of axial reflux in the greater saphenous or lesser saphenous vein. Experience teaches that any therapy of varicosities in the persistent presence of saphenous reflux will fail. Large varices of the medial thigh lend themselves to phlebectomy rather than to sclerotherapy for two reasons. First, large varices, in general, respond better to removal if only to avoid superficial thrombophlebitis after injection. Second, large varices in this location are usually markers for large incompetent mid-thigh (Hunterian) or distal thigh (Dodd) perforating veins which are detached by phlebectomy. These are difficult to compress after sclerotherapy. OPTIONS IN INTERVENTION: LIGATION VERSUS STRIPPING Objectives of treatment should be ablation of the hydrostatic forces of axial reflux and removal of the hydrodynamic forces of perforator vein reflux. In surgery these should be combined with phlebectomy of vein clusters in as cosmetic a fashion as possible. Sclerotherapy should be used to ablate highpressure, feeding reticular vessels in combination with injection of their targets, the epidermal veins, telangiectasias. The objectives of sclerotherapy and surgery are identical. These are ablation of high-pressure conduits and excision or obliteration of their target vessels. Since a variety of surgical approaches have been used, discussion of these is appropriate. Retrospective reviews of surgical experience are looked upon with disfavor today. Yet a thread of wisdom can be found in some such studies. Lofgren et al. examined limbs of patients up to 5 years following high saphenous ligation or complete ankle-to-groin stripping and found 94% excellent or good results after stripping compared to 40% after high tie. This and other studies were dismissed by Dormandy as he presented his prospective, randomized study. He compared saphenous vein stripping and stab avulsion to saphenous high tie and mid-thigh perforator interruption, finding no differences in either patient or physician evaluation at 3 years. Results of this study were criticized because evaluation was entirely subjective rather than objective. FATE OF SAPHENOUS LIGATION Fate of the long saphenous vein after proximal ligation has been defined by duplex scanning. Rutherford’s evaluation showed that only 8–10 cm of proximal saphenous vein was obliterated after high ligation. He found postligation that all saphenous veins were patent. Friedell et al. studied patients in Florida at a mean follow-up of 10 months and found 78% of saphenous veins completely patent, 15% with less than 10 cm obliterated, and only 7% with a greater length obliterated. In Cardiff, 75 limbs were assessed at a mean follow-up of one year by duplex scans. In 49 limbs (65%) the entire saphenous vein was patent from ankle to groin, 12 and in two additional limbs less than 5 cm was lost proximally. In a physiological evaluation of high saphenous ligation using duplex scans and PPG, the Middlesex group found persistent reflux in 24 of 52 limbs with successful proximal ligation: “A satisfactory outcome was associated with absence of reflux down the long saphenous vein after operation” concluded the authors, who added that proximal ligation “fails to control functionally significant reflux within the long saphenous vein in a high proportion of cases.” RECURRENT VARICOSITIES Some lessons can be learned from studies of recurrent varicose veins. These have been termed “a national problem” by Thomson of Gloucester. Using duplex venous imaging and a careful search for sources of reflux, Thibault and Lewis studied 122 limbs with recurrent varicose veins. Of these, 71.3% had recurrent, incompetent superficial thigh veins in the long saphenous distribution. Redwood and Lambert studied 127 limbs with recurrent varicose veins and found that when a single site of reflux was found, it was at the saphenofemoral junction in 58% of limbs. They concluded that “the high recurrence rate from the groin emphasizes the need for meticulous dissection around the saphenofemoral junction at the time of the initial surgery.” Patterns of incompetence in 100 limbs with recurrent varicose veins were studied by duplex ultrasonography in Adelaide, Australia. Saphenofemoral or recurrent groin tributary incompetence was present in 44 limbs and an incompetent long saphenous remnant in 20, with the authors saying, “the high incidence of long saphenous remnant incompetence in which no other site of incompetence was detected . . . supports the argument for stripping.” Ruckley’s group studied 128 limbs with recurrent varicose vein by varicograms and provided a useful classification of recurrent varices. That is, a Type 1 recurrence required reexploration of the groin, while a Type 2 recurrence allowed operation to be performed without reexploration of the groin. In 75 of 128 limbs, the long saphenous vein was present in the thigh, and in 37 it was present to groin level. Clearly, in all but 24 instances of the 128 limbs, the site of recurrence could have been prevented by more complete primary procedure. Another important observation deriving from this paper is that saphenofemoral tributaries must be removed beyond their primary or even secondary tributaries in order to avoid leaving behind a network of collaterals that can defeat the primary operation. Ruckley concludes, “it is clear from this and previous work that a large number of patients are undergoing inadequate first-time varicose vein surgery. The importance of accurately identifying and ligating the saphenofemoral venous complex, of obliterating all tributaries back to the secondary tributary points, and of stripping the long saphenous vein in the thigh and preventing recurrent varices must be emphasized yet again.” Ligation of the saphenous vein at the saphenofemoral junction has been practiced widely in the belief that this would control gravitational reflux while 13 preserving the vein for subsequent arterial bypass. It is true that the saphenous vein is largely preserved after proximal ligation. However, reflux continues and hydrodynamic forces are not controlled. Recurrent varicose veins are more frequent after saphenous ligation than after stripping of the saphenous vein in the thigh. Also, recurrent varicose veins are more frequent after saphenous ligation and sclerotherapy than after stripping and sclerotherapy. Prospective, randomized trials comparing proximal saphenous ligation and stab avulsion of varices to stripping of the thigh portion of the saphenous vein and stab avulsion of varices have shown superior results for the latter procedure. Careful duplex evaluation of proximal saphenous vein ligation 2 years following intervention showed that, “a large group of patients (33%) had developed significant collateral veins at the level of the operative site.” Ninety-five percent of the saphenous veins were patent to within 10 cm of ligation, and 88% were incompetent. In studying recurrent varicose veins, preservation of patency of the saphenous vein and continued reflux in the saphenous vein have been found to be the most frequent elements in such recurrence. In patients presenting for surgical relief of recurrent varicosities, it has been found that two thirds required removal of the saphenous vein as part of the repeat procedure. VARICOSE VEIN SURGERY Following the principle that surgical removal of large varicose veins is superior to sclerotherapy, operation is offered to patients if the symptoms are attributable directly to varicose veins and after evaluation that indicates that the patient will be likely to benefit from removal of the varicose veins. Formerly, as indicated previously, the operation of saphenous vein removal was performed as a stripping procedure from ankle to groin, often placing the intraluminal stripper through the ankle incision. Several problems were encountered using that technique. The most common was saphenous nerve injury due to avulsion of saphenous nerve 7–13 cm below the knee joint crease. Less frequently, the stripper entered the femoral vein through angulated perforating veins, and cases have been reported of stripping of the superficial femoral vein because of this. Because the objective of saphenous vein removal is to detach perforating veins which carry superficial venous blood from the saphenous system to the deep system, stripping of the saphenous vein need only be done from groin to knee. Introduction of the stripper can be done from above as incompetence of saphenous valves has been proven by preoperative duplex scanning. Sometimes, when subcutaneous fat is excessive, it is difficult to identify the stripper within the saphenous vein at knee level. The saphenous vein can be identified preoperatively and marked at the medial aspect of the popliteal space where the saphenous vein is relatively constant. This allows placement of a cosmetically satisfactory incision on the medial aspect of the knee. However, with preoperative duplex testing confirming saphenous vein reflux, the stripper can be placed from above downward to exit at a cosmetically placed 14 incision on the medial aspect of the knee. Exposure of the saphenous vein at its termination is done through a proximal incision, either in the upper thigh skin fold or 1 cm above it where the saphenous vein regularly enters the femoral vein. The more proximal incision can be made much shorter because it is directly over the termination of the saphenous vein. Exposure must allow exposing the femoral vein 1 cm above and 1 cm below the saphenous vein. This is to verify that no other tributaries are entering the femoral vein rather than the saphenous vein. Regularly, the epigastric and circumflex iliac tributaries can be seen to enter the saphenous vein and are markers for its location. Less important are the pudendal veins, but of greatest importance are the medial posterior and lateral anterior tributaries. Ruckley advocates dissecting all tributaries to the saphenous junction beyond their primary tributaries in order to avoid leaving a network of veins behind. Our own practice is to bring each tributary into the incision, avulsing the tributaries as this is done, and applying pressure wherever tributary bleeding is troublesome. While many surgeons advocate clipping or ligating tributaries to the saphenous vein, we find that electrocoagulation is perfectly satisfactory. A good rule is to cannulate with the internal stripper each major vein entering the groin incision. In this way, very large lateral anterior tributary veins can be stripped to knee level and even the posteromedial tributary can be stripped to mid-thigh or below. Leaving these large tributaries behind contributes to recurrent varicosities. Increasingly, the technique of inversion stripping is being taken up. Our own technique adds the hemostatic pack, which has been described elsewhere. If saphenopopliteal incompetence is found, attention must be paid to the short saphenous vein. Because of the specter of sural nerve injury, many surgeons turn to proximal short saphenous ligation rather than stripping, but the inversion technique described by Oesch has simplified short saphenous stripping and has corrected the problem of sural nerve injury. That injury was found to be associated with an ankle incision and exposure of the short saphenous vein posterior to the lateral malleolus. In any event, imaging of the saphenopopliteal junction must be carried out, and, increasingly, duplex scans are favored rather than the intraoperative films advocated by Hobbs. When varicose veins are simply huge or if operations are done for varicose veins of the Klippel-Trenaunay syndrome, an Esmarch tourniquet is used to exsanguinate the limb and an orthopedic tourniquet inflated to 250mmHg is utilized. Interestingly, the orthopedic tourniquet will not only allow the passage of the saphenous vein intraluminal stripper, but also the stripping can be done while the tourniquet is inflated. The use of the Esmarch exsanguination and proximal tourniquet markedly reduces blood loss and tidies up the performance of the operation. Distal phlebectomies can be done under direct vision without troublesome bleeding. In the United States, it is not routine to give deep venous thrombosis prophylaxis nor is any attention paid to the taking of oral contraceptive pills or hormone 15 replacement therapy. However, in England and on the European continent, very often prophylaxis for deep venous thrombosis is employed, using either subcutaneous unfractionated heparin 5000 units twice a day or the equivalent fractionated heparin dose. Our practice is to avoid hospitalization of the patients. Many surgeons have taken up the techniques developed by dermatologists as these colleagues have provided surgeons with satisfactory instrumentation, which minimizes the size of the incisions. In particular, the Muller hooks are of value, as are the Varaday dissectors. Targets of surgical phlebectomy are major varices fed by refluxing perforating veins as well as reticular veins which nourish telangiectasias. The extraction of these veins is performed through minimal skin incisions, 1–3 mm, or even puncture with a No. 18 needle. Definitive removal of the veins is far more successful than sclerotherapy, and dermatologic ambulatory phlebectomy has proven itself to be complementary to stripping of the greater saphenous vein and its tributary varices. MODERN SAPHENOUS ABLATION Prolonged exposure to high-frequency alternating current [radiofrequency (RF)] energy results in total loss of vessel wall architecture, disintegration, and carbonization. Application of this knowledge has allowed treatment of the greater saphenous vein by intraluminal techniques. The preliminary results obtained in 389 patients treated with RF energy were clouded by third-degree burns of the skin, saphenous nerve injury, periphlebitis, peroneal nerve injury, and wound infection. Elimination of saphenous vein reflux is done using RF heating. The VNUS vein treatment system utilizing the Closurew catheter (VNUS Medical Technologies, Sunnyvale, California) is the most used system in this country and in Western Europe. This system uses electrodes specifically designed for treatment of the saphenous vein and includes monitoring of electrical and thermal effects of the catheter. Clinically, the device produces precise tissue destruction with minimal formation of thrombus. Bipolar electrodes are used to heat the vein wall. The net effect is venous spasm and collagen shrinkage, which produces maximal physical contraction. In practice, elimination of venous flow is accomplished by Esmarch bandaging and proximal saphenofemoral junction compression. Saphenous vein ablation has been performed using intravenous sedation and tumescent anesthesia alone and with general anesthesia with and without proximal saphenofemoral ligation. Acute closure has been achieved in 93% of 141 saphenous veins in the first large series to be reported and 1-year continued closure exceeds 90% with only a small fraction of the original anatomic failures requiring retreatment. Surgical series have shown that undesirable outcomes after saphenous stripping are evident quite early. It is acknowledged that surgical stripping results in recurrent truncal vein reflux in 20% of limbs and that 73% of limbs destined for recurrent varicosities at 5 years have already done so at 1 year. Thus, the 1-year 16 results of VNUS Closure seem destined to be comparable to stripping in the long term. Goldman, who has taken the lead in endovenous closure in our office, uses large amounts of tumescent anesthesia containing 0.1% lidocaine with epinephrine. Intraoperative ultrasound monitoring ensures that the greater saphenous vein is separated from the skin by the tumescent anesthesia, thus avoiding skin burns. Performing endovenous obliteration of the saphenous vein without dissection of the saphenofemoral junction violates a cardinal rule in saphenous vein surgery. This holds that each of the tributaries must be individually divided. It is advocated by some that each of the tributaries should be dissected back beyond their primary and even secondary tributaries. Careful duplex evaluation of saphenous obliteration by Pichot has revealed marked shrinking and obliteration of the saphenous vein itself but with preservation of tributaries to the saphenofemoral junction. A discussion of this point occurred at the annual meeting of the American Venous Forum in February 2000. Sixty limbs treated with saphenofemoral junction ligation and division of tributaries were compared to 120 limbs treated without high ligation. Of the 49 high ligation limbs followed a sufficient length of time, 2% developed recurrent reflux by 6 months and in the 97 non–high ligation limbs followed for that length of time, 8% developed recurrent reflux. In limbs followed to 12 months, no new instances of reflux developed. Actuarial recurrence curves were not different with or without saphenofemoral ligation, and the experience predicted a greater than 90% freedom from recurrent reflux and varicosities at 1 year for both groups. The issue is not settled, but it is acknowledged that should a tributary develop reflux and prove to be a source of recurrent varicosities, the problem can be managed without further surgery by using sclerotherapy. Many surgeons would prefer ambulatory phlebectomy at this point, but in either event the problem is not a major deterrent to use of endovascular saphenous vein obliteration by radiofrequency energy without saphenofemoral ligation. SCLEROTHERAPY Sclerotherapy is an integral part of treatment of venous insufficiency and should not be regarded as a substitute for surgery but instead as supplemental. Virtually any vein can be sclerosed, but recanalization is the rule in large veins that may be the target of venous hypertension. Before sclerotherapy is initiated, all sites of reflux from the deep venous system into the superficial varicosities should be divided. This implies that the major named perforating veins referred to elsewhere should not be the source of reflux, nor should the saphenofemoral or saphenopopliteal junctions be refluxing. Sclerosing agents themselves have the ability to denature biologic molecules within the vein wall. Their full categorization, chemical structure, and biologic activity is described elsewhere. It is commonly and erroneously thought that the result or objective of satisfactory sclerotherapy is formation of an intravascular 17 thrombus. In fact, the opposite is true. Such thrombosis increases the likelihood of superficial thrombophlebitis and recanalization of the treated vein. General principles guiding successful sclerotherapy include the fact that it is essential that the vein be empty of blood as the sclerosant is injected. Usually the extremity in a horizontal position achieves nearly complete emptying of superficial veins. If the needle is inserted while the limb is dependent, the vein is more distended and easier to cannulate. However, before injection the limb should once again be horizontal or perhaps elevated 5–30 degrees. The second general principle is that no more than 0.5–1mL of sclerosant per injection site should be used. For sodium tetradecyl sulfate and sodium morrhuate, the concentration of such solution should range from 0.25–0.5%. Admittedly, higher concentrations and larger volumes are required for varicosities greater than 3mm in diameter. However, moderate sclerotherapy limits this technique to vessels of smaller diameter. The maximum quantity to be used in any one treatment session should be equivalent to 10–15mL of 1% solution, depending on the patient’s body weight. The third general principle in the practice of successful sclerotherapy is compression. The importance of such compression was recognized as early as the 1930s by some. External compression attempts to prevent the return of blood into the vein lumen. The duration and degree of compression depends on the size of the varicosity, with compression of telangiectasias and reticular varicosities requiring only a few hours and, conversely, up to 6–8 weeks for very large varicosities. Clearly, large varicosities with great intramural pressures require longer durations of compression, but it is exactly these veins which can best be removed by ambulatory phlebectomy. Compression is easily achieved by application of gradient compression, 30– 40 mmHg, at the ankle. If sclerotherapy is considered an adjunct to surgery, success is to be expected. Sources of gravitational reflux should be attended to by surgery. Sources of hydrodynamic pressure from muscular compartments should be dealt with surgically and large varicosities removed. This will leave telangiectasias and reticular varicosities smaller than 3mm for subsequent sclerotherapy. It is exactly these targets that are most susceptible to sclerotherapy. Literature I. Basic literature: 1. Хирургик касаликлар. Ш.И.Каримов. Тошкент, 2005. 2. Хирургические болезни. Ш.И.Каримов. Ташкент, 2005. 3. Hirurgik kasalliklar. Sh.I.Karimov. Toshkent, 2011. 4. Хирургик касаликлар. Ш.И.Каримов, Н.Х.Шамирзаев. Тошкент, 1995. 5. Хирургические болезни. Под ред. М.И.Кузина. Медицина, 2002. 6. Методическое пособие по госпитальной хирургии. Назыров Ф.Г. с соавт. Ташкент, 2004. 18 7. Клиническая хирургия. Под ред. Ю.М.Панцырева. Москва, «Медицина», 1988. 8. Справочник практического врача (в 3х томах). А.Воробьев. 1990. 9. Клиническая хирургия. Р.Конден, Л.Нейхус. Москва, «Практика», 1998. 10. Справочник-путеводитель практикующего врача. Ф.Г.Назиров, И.И.Денисов, Э.Г.Улугбеков. Москва, 2000. 11. Руководство по хирургии. Под ред. Б.В.Петровского. (в 12 томах). Москва, «Медицина», 1959-1966. II. Additional literature: 12. Практическое руководство по хирургическим болезням. В.Г.Астапенко. Минск, 2004. 13. 50 лекции по хирургии. В.С.Савельев. Москва, 2004. 14. Основы оперативной хирургии. Под ред. С.А.Симбирцева. 2002. 15. Диагностический справочник хирурга. В.Н.Астафуров. 2003. 16. Хирургическая операция. Расстройство гомеостаза, предоперационная подготовка. И.Я.Макшанов. 2002. 17. Internet: www.rmj.net; www.mediasphera.ru; www.medmore.ru; www.consilium-medicum.com; www.medilexicom.com; www.encicloperdia.com. 19