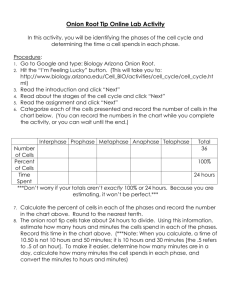
36 Osmosis Onion Lab lab plasmolysis red onion 1415

.9% nacl = 9 gm/ L
Onion Plasmolysis Lab
Introduction
Have you ever seen a plant with wilted leaves? What causes this to happen? Plant leaves wilt in response to a loss of water from individual cells. When the cell loses too much water, the cell membrane will shrink away from the cell wall in a process called plasmolysis. As you know, water moves across the plasma membrane by osmosis, from an area of high water concentration to an area of low concentration. This movement of water affects cell homeostasis
– the maintenance of equilibrium between an organism and its environment.
Experimental question How do onion cells behave in different concentrations of salt water?
Materials
Red onion soaking in distilled water
0.9% NaCl in H
2
O
2% NaCl in H
2
O
7% NaCl in H
Paper towels
2
O
disposable pipets
microscope slide and a cover slip
Compound light microscope
Procedure
1. Formulate a hypothesis explaining which solution you think will be isotonic to the onion cells and why.
Remember to use the “If…then…because” format.
2. Prepare a Wet Mount : Using your fingernail, peel off a small, very thin piece (approximately1 cm 2 ) of the outer purple layer from the onion. Mount this, cut side down, onto the COVERSLIP. Put the coverslip down on the slide so the onion is sandwiched between the two pieces of class.
3. Focus the slide under low power and then switch to medium power. DO NOT USE HIGH POWER. Adjust the diaphragm so the amount of light allows you to visualize the cell wall and the vacuole of the cell. a. Count the total number of cells you see and then the number of plasmolyzed onion cells you see. b. Calculate the percentage plasmolyzed. c. Take a photograph or make a labelled sketch of what you see. If you take photos, be sure to share them with your partner. They must be printed and labeled in your notebook.
4. Adding the 0.9% solution : It is extremely important that you do not get any salt solution on the lenses!
Gently put pressure on the left side of the coverslip to lift the opposite side a bit. Place a drop of the 0.9% salt solution under the right side of the coverslip. Place a small piece of paper towel on the left side of the coverslip to draw the salt water across the slide. This will expose the onion epidermis to the salt solution.
5. Observe the effects of the 0.9% salt solution on the onion cells. You may notice the movement of particles in the water as the water moves to the opposite side of the slide. Do not let these particles distract your attention! Keep focused on what is occurring inside the onion cells. Notice any changes in color, size, or shape of the cell structures. a. Count the number of plasmolyzed cells you see. b. Calculate the percentage plasmolyzed. c. Take a photograph or make a labelled sketch of your observations in the space in the hand-in sheet.
6. Adding the other solutions . Now, using the same piece of onion, repeat the process for 2% and then 7% salt solution.
7. Cleanup: When finished, remove the slide from the stage of the microscope, wash all slides and cover slips to make sure all the salt has been removed. Place them on the paper towel on your lab station. If you have gotten any salt on your lenses, clean them with lens paper.
Post Lab Analysis:
1. What concentration of salt solution appears to be isotonic to the onion cells?
2. Before the onions were placed in the various concentration of salt water, they were soaked in distilled water. What is
the relationship between the onion cells and the distilled water?
3. List 3 sources of inaccuracy in this experiment.
4. Why don't all the cells in the same piece of onion have the same osmotic potential?