Regualtion of Cycle & Signalling
advertisement

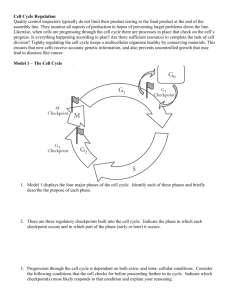
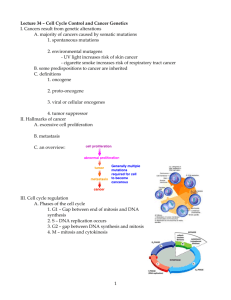
Cell Cycle 3 – Regulation of Cycle and Signalling Anil Chopra 1. Explain in molecular terms the concept of a tumour suppressor, using as an example the retinoblastoma (Rb) susceptibility gene product. 2. Explain in molecular terms the concept of a proto-oncogene and provide examples. 3. Explain how apoptosis can limit inappropriate cell division and discuss the role of cytoprotective pathways (e.g. the phosphoinositide 3' kinase pathway) in facilitating cell proliferation. The turnover of haematopoietic cells in a 75kg adult is around 500 000 000 000 (500 billion) cells a day. A small number of progenitor stem cells (1% of marrow cells) maintains production. The stem pool responds to external stresses e.g. in hypoxia, number of red blood cells increases. In bacterial infection granulopoiesis increases. With the use of proprium iodide staining, we know that the most DNA is in the cell when it is in G1. E2F is a transcription factor that regulates genes. When the DP1/E2F1 heterodimer is bound to pRB (a tumour suppressor), it cannot initiate transcription, however, when certain cdks (cyclin dependent kinases) bind with certain cyclins, then they phosphorylate the pRB causing the DP1/E2F1 to detach from the pRB leaving the DP1/E2F1 to initiate transcription. In this way pRB acts as a tumour suppressor. Regulation of DP1/E2F1 heterodimer during the cell-cycle pRB G1 DP1 E2F1 Transcriptional inactive/ DNA bound cyclinD/cdk4 or 6 cyclinE/cdk2 cyclinA/cdk2 pRB DP1 G1/ S Transcriptional active/ DNA bound E2F1 cyclinA/cdk 2 DP1 E2F1 S/ G2 Transcriptional not DNA inactive/ bound In order to control the cell cycle we need to use cyclin-dependent kinase inhibitors (CKI’s) of which there are 2 types: INK family – cause separeation of the CDK from the cyclins. Cip/Kip family - bind to and inhibit the action of the CDK-cyclin complex. M G1 Cyclin BCDC2 P pRb E2F G2 P Ink4b X P P Cyclin D-CDK4/6 pRb E2F S p21Waf-1 p27Kip-1 p57Kip-2 Cyclin E-CDK2 p21Waf-1 p27Kip-1 p57Kip-2 Cyclin A-CDK2 At restriction point “R” in the cell cycle, the presence of growth factors is needed for G1 to progress. In the presence of growth factors, cyclin D binds to CDK 4 and phosphorylates the pRB (tumour suppressor). Later in the mid/late G1 phase, the complex formed when cyclin E binds to CDK 2 causes the pRB to be phosphorylated again resulting in its release of the DP1-E2F1 complex. Early G1 CDK4 pRB G1/S Mid/Late G1 Cyclin D P CDK2 pRB Cyclin E P pRB P P P DP1 DP1 E2F1 D DP1 P E2F1 E2F1 Growth factor Cyclin E Cyclin E The binding of CDK2 to cyclin E also causes the breakdown of the CDKI p27. All E2F genes Mid/Late G1 CDK2 Cyclin E P p27 p27 An oncogene is a mutant gene which promotes cell proliferation. A proto-oncogene is the normal cellular gene corresponding to the oncogene. There are a number of proto-oncogenes that are involved in normal cell division, that once mutated become oncogenes and allow uncontrolled proliferation of cells (i.e. cancer). These include: The EGF receptor Ras which may become the oncogene V12Ras following a Gly12Val mutation, or L61Ras following Gln61Leu mutation. C-Raf which becomes the oncogene v-Raf upon deletion of it regulatory domain. C-Jun which becomes the oncogene v-Jun upon deletion of its regulatory domain. Mutations usually result in cell cycle arrest and apoptosis. P53 is a tumour suppressor gene, and it also promotes apoptosis. For unregulated cell signals to occur, one of the following must occur: o There must be an active proliferative pathway o Cell cycle arrest must be inhibited o Cell survival signals must be activated. The ERK cascade is cytoprotective as it produces a protein called p90RSK which can phosphorylate Bad. This means that Bad is bound to the protein and can therefore not be part of apoptosis. This leads to the proliferation of the cell. PI3K (phosphatidylinositol 3-kinase) promotes cell survival. A extracellular survival signal activates a receptor tyrosine kinase, which recruits and activates PI3K. It results in the phosphorylation of the Bad protein, which holds inhibitory proteins. These are released and can block apoptosis, thereby promote cell survival. The diagram shows the activation and inactivation of Ras. However, V12Ras prevents GAP binding to it, so once the Ras has been switched on, it cannot be switched off, therefore Ras will continuously stimulate the ERK cascade which could lead to uncontrolled cell proliferation. A similar thing occurs with L61Ras – it prevents GTP hydrolysis (i.e. stops the GAP from doing its job) and it to will therefore remain active continuously.