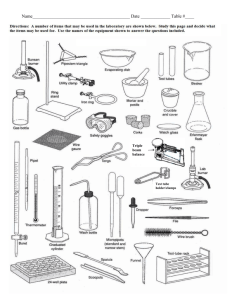
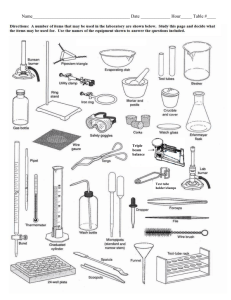
science lab: density, data collection, identification of
advertisement

Laboratory Normal Lab Write-up Contents 1. 2. 3. 4. 5. 6. 7. 8. 9. Names Period Title of Lab Purpose Procedure(optional) Safety Precautions Data(usually in Data Table form) Analysis(answers to questions) Conclusion(answers to questions) Organized Experimentation Problem/Questions: State the purpose of the experiment. Facts/Research: Write the facts that you know or have researched about the problem. Hypothesis: Based upon the facts/research, what do you think the outcome of the experiment will be? Materials: List the materials that you will need to do the experiment. Procedure: List the step you plan to take in the experiment. Data Collection/Tables: Prepare tables and record the data that your experiment will produce. Data Analysis: Find ways to relate the data: graphs, math and others. Conclusion: Application Lab: Observations of a Candle Names _______________________________________ _______________________________________ Period ______________ Date _______________ Purpose: Record as many observations as possible in 10 minutes of a candle. During part of the observation time the candle should be lit. Materials: Candle and any equipment available in the room. Cautions: An OPEN FLAME will be in use. Therefore, long hair MUST be restrained. Care should also be followed to avoid burning human tissue and clothing. Hint: The purpose is not to compile an extensive list of “things that will burn in a candle”. One will do, donkey! Observations: 1. __________________________________________________________________________ 2. __________________________________________________________________________ 3. __________________________________________________________________________ 4. __________________________________________________________________________ 5. __________________________________________________________________________ 6. __________________________________________________________________________ 7. __________________________________________________________________________ 8. __________________________________________________________________________ 9. __________________________________________________________________________ 10. __________________________________________________________________________ 11. __________________________________________________________________________ 12. __________________________________________________________________________ 13. __________________________________________________________________________ 14. __________________________________________________________________________ 15. __________________________________________________________________________ 16. __________________________________________________________________________ 17. __________________________________________________________________________ 18. __________________________________________________________________________ 19. __________________________________________________________________________ 20. __________________________________________________________________________ Instruments and Uncertainties Lab Name: _______________________________________ Station Number 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 Instrument Period: ________________ Smallest Measurement Uncertainty Factor Uncertainty Factors To find Uncertainty Factor of an Instrument: 1. Estimate 1 additional place value beyond the markings 2. Then +/- half of the smallest readable graduation To find smallest readable graduation: 1. Take difference between any two numbers from the middle of the instruments scale 2. Count the number of graduations between the numbers(graduations not lines) 3. Divide difference/graduations = smallest readable graduation To find Uncertainty factor of a Derived Quantity(from a math operation not measurement) 1. During Addition or Subtraction a. Round all numbers to similar decimal places b. Do the math operation needed for the calculation(add or subtract) c. Add all uncertainty factors from all of the involved numbers to get the new uncertainty factor d. Write answer as b +/- c Example: subtract 23.34+/-.5 ml from 125.4+/- 1 ml a. round 23.34 to 23.3 to match decimal placement of 125.4 b. subtract 125.4 - 23.3 = 102.1 c. add uncertainty factors: .5 + 1 = 1.5 d. new answer: 102.1 +/- 1.5 ml 2.During Multiplication or Division a. Round all numbers to similar decimal places b. Do the math operation needed for the calculation(multiply or divide) c. Divide each measurement’s uncertainty factor by the measurement d. Which ever provides the largest answer controls the final uncertaint e. Multiply the largest answer from d. times the math answer in b f. This new number becomes the new +/- uncertainty factor. Example: Find density of 13.45 +/- .05grams and 25.6 +/- .5 ml a. Round 13.45 to 13.5 to match 25.6 decimal number b. c. D = M/V therefore 13.5/25.6 = .527 .05 ------- = .00370 13.5 d. .0195 is largest e. .0195 X .527 = .001 f. .527 +/- .001 g/ml .5 -------- = .0195(largest) 25.6 Chemistry Lab: Aluminum – Copper (II) Chloride Reaction Name: _______________________________ Name: _______________________________ Name: _______________________________ Period: _____ Date: ___________________ Directions: In this laboratory, you will make both quantitative and qualitative observations about the chemical reaction between aluminum and a copper (II) chloride solution. Materials / Equipment: 150 mL beaker thermometer plastic spoon Copper (II) chloride crystals Aluminum foil (about 10 cm x 10 cm) funnel and filter paper Safety Precautions: You must wear safety glasses at all times. Be careful not to get any chemical in your eyes or mouth. Clean up your equipment and area at the end of each period. Store products as instructed. Wash your hands when finished. 1. Put 100 mL of water into the beaker. 2. Record the temperature of the water. 3. Add a spoonful of Copper (II) chloride to the water and stir till dissolved. 4. Take the temperature of the water / Copper (II) chloride solution and write down any qualitative observations that you can. 5. Find and record the mass of a piece Aluminum foil. 6. Place your piece of loosely-crumpled foil into the solution. You may stir with a stirring rod several times. 7. Take the temperature of the mixture and, again, write down any qualitative observations that you can. 8. Allow to react overnight. 9. Obtain a piece of filter paper; get the mass of the filter paper from your instructor. 10. Fold the filter paper into halves and then quarters. Open 1 of the 4 flaps and place into the filter. Squirt with a few drops of distilled water. 11. Filter the content through filter paper. Catch the liquid in an Erlenmeyer Flask. 12. Rinse the filtrate with 20 ml of distilled water. 13. Allow the filtrate to dry in the Drying Oven overnight. 14. Find and record the mass of your filtrate. 15. Clean up your lab area according to your teacher’s instructions. CProcedure: Data: Temperature of water: ______________ Temperature of water / Copper (II) chloride solution: ______________ Mass of the Aluminum foil ______________ Mass of the dried filtrate ______________ Qualitative observations of water / Copper (II) chloride solution: 1. 2. 3. 4. Page two Qualitative observations of mixture: 1. 2. 3. 4. 5. Temperature changes What do you think has caused the temperature changes? ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ Mass changes What do you think is the cause of the change of mass from day 1 to day 3? Be sure to remove the mass of the filter paper first. ____________________________________________________________________ ____________________________________________________________________ ____________________________________________________________________ ____________________________________________________________________ ____________________________________________________________________ ____________________________________________________________________ Lab: Determining Density and Identifying an Unknown You will be required to find the measurements needed to calculate the density with uncertainty factors of the following three objects. You will then calculate the density of an unknown and use the density to identify the unknown material. I. Density of Solid Objects Obtain one each of the following objects: glass, rubber, metal. Use the balance to determine the mass and a ruler (regular objects) or water displacement (irregular objects) to measure the volume of the objects. Object Mass of Object Uncertainty of mass Volume of Object Uncertainty of volume Density of Object Uncertainty of Density Glass Stopper Rubber Stopper Metal II. Density and identification of an unknown liquid Now, you will be a stoppered test tube containing an unknown liquid. Prepare a procedural plan to find the density of the liquid. You may use any equipment available in the lab area. Day 2: The group members must agree on a plan to find the density and identify the unknown record the steps below. Make a list of any SAFETY PRECAUTIONS that need to be followed during this lab. Assume that all chemicals are FLAMMABLE, TOXIC and CAUSTIC. Page 2 Density Lab Density is frequently used to determine the identity of an unknown substance. Find the density of the unknown supplied to your group and identify using the chart of common densities and data below. TABLE OF DENSITY OF SOME COMMON COMPOUNDS ----------------------------------------------------------------------------------------------------COMPOUND DENSITY ----------------------------------------------------------------------------------------------------1,1,2-TRICHLOROTRIFLURO ETHANE 1.565 GLYCEROL 1.261 1,2-DICHLORO ETHANE 1.245 TRIETHANOLAMINE 1.124 ETHYLENE GLYCOL 1.109 ACETIC ANHYDRIDE 1.082 ACETIC ACID 1.049 BENZALDEHYDE 1.042 WATER 1.000 ETHYL ACETATE .894 TOLUENE .870 METHANOL .791 2-PROPANONE (ACETONE) .770 ETHANOL .769 CYCLOHEXANE .757 HEXANE .664 UNKNOWN NUMBER MASS UNCERT. VOLUME UNCERT. DENSITY UNCERT. ----------------------------------------------------------------------------------------------------------------------------------------------| | | | | | ----------------------------------------------------------------------------------------------------------------------------------------------UNKNOWN IDENTITY:____________________________ PERCENTAGE ERROR ((YOUR DENSITY/REAL DENSITY)*100):_______________ (SUBTRACT 100 IF LARGER THAN 100) Lab: Aluminum Foil and Atoms Names _________________________________________________ _________________________________________________ _________________________________________________ Period _______ Purpose: To find the atom thickness in Al foils. Safety Precautions: ______________________________________________________________ Materials: Ruler, Al bar, Regular Al Foil, Heavy Duty Al Foil, scissors, balance Procedures: 1. Find the Density of Al using the bar. 2. Trace two dimensions of the Al bar onto the foils and cut out the traced form. Mass and record the foil pieces. 3. 4. Using the density of Al and the area and mass of the foil sheet, determine the thickness of the foils. Research the atomic radius of Al. Use the radius to determine the number of Al atom thickness for each foil. BEANIUM LAB Directions: Complete the following activity with a partner or partners. Do not work in groups of more than three. Do all calculations carefully and show all work. Please return all beans to the container they came from when you are through. You have been given a sample of newly discovered element – Beanium! It is your job to determine how many isotopes of this element exist, the atomic mass of each isotope, and finally the weighted average that will become the average atomic mass of Beanium. One unique property is that the atoms are very large. Therefore, sorting the isotopes of this element should be accomplished with little difficulty. Repeat for all isotopes. Record this information on your data table. Materials: Beanium sample and Triple beam balance Procedure 1. Sort the beanium atoms into groups, each group representing a different isotope. (Each different bean represents an isotope of the element beanium.) Record the total number of atoms (beans) in your sample as well as the number of each isotope on your data table. 2. On the data sheet, sketch a picture of each isotope clearly showing the differences between each. 3. Determine the atomic mass of each isotope in the following manner: a. Find the total mass of each isotope; record these masses on the data table. b. Divide the total mass of each isotope by the number of atoms(beans) in the sample to find the average mass of a single atom of that particular isotope, and hence its atomic mass 4. Determine the relative abundance of each isotope in the entire sample by dividing the number of atoms of the isotope by the total number of atoms in the sample. Repeat for all isotopes. Record this information on the data table. 5. Determine the average atomic mass for Beanium based on the relative abundances of each isotope and their atomic masses. This is the method to use: Average atomic mass = (Relative abundance of isotope 1 x mass of one atom of isotope 1) + (Relative abundance of isotope 2 x mass of one atom of isotope 2) + ………………………………………………………. Questions – Answer completely and show work where necessary!! 1. What is the basic atomic difference between isotopes of the same element (real elements not beans)? 2. If there are 100 navy beans, 27 pinto beans and 173 black-eye peas in a container, what is the percent composition of the container by bean? (% composition = Part/whole X 100) 3. If your chemistry grade is broken down so that 70% of it is exams, 20% is labs, and 10% is homework, and your average scores out 100 points in each area are: Exams 85, Labs 75, Homework 96 Calculate your weighted average: 4. A student obtained the following data when measuring her Beanium sample: TYPE # OF BEANS MASS OF SAMPLE Bean A 162 61.052 g Bean B 181 54.182 g Bean C 292 59.784 g Calculate the following: a. percent composition (relative abundance) of each bean b. “atomic” mass of each isotope c. average “atomic” mass of beanium for this sample Five Gas Lab Preparation 1 day of PreLab discussion of procedures and SAFETY PRECAUTIONS Prepare 2 stations for each gas. This allows for 10 groups to move freely. Stations for Gases 1-3 Station requirements: 1 trough with attached rubber tubing, glass tubing and stopper of correct size to stopper a 25x200 test tube as the reaction chamber. 2 sample collection 25x200 test tubes and solid stoppers for them. Stand w/ clamp Test Tube Clamp(hand held) Bunsen Burner for Hydrogen and Oxygen Chemicals: Gas 1: Nitric Oxide 3-6 M Nitric Acid (200 ml/class) Copper (shot, turnings, foil any will do) (50 g/class) Gas 2: Hydrogen 3-6 M Hydrochloric Acid ( 200 ml/class) Zinc metal (50 g/class) Gas 3: Oxygen Hydrogen Peroxide (3% or 6% works best) (200 ml/class) Ferric Chloride saturated solution (20 ml/class) Stations for Gases 4-5 Station requirements: 1 trough. 2 sample collection 25x200 test tubes and solid stoppers for them. Test Tube clamp Bunsen Burner Chemicals: Gas 4: Ammonia Concentrated Ammonium Hydroxide (20 ml/class) Red Litmus Paper or Hydrion paper. Gas 5: Hydrogen Chloride Concentrated Hydrochloric Acid (20 ml/class) Blue Litmus Paper or Hydrion Paper. Safety Precautions: All= Goggles, Glassware safety, No Contact lenses or open toed shoes Gas 1. Production: Toxic gas, Acid use Testing: Toxic Gas Gas 2. Production: Acid use, Explosive gas, Purging required(explosion if omitted) Testing: Open flame, Explosive gas Gas 3. Production: Open flame, Iron toxic solution, 6% peroxide is skin irritant Testing: Open flame Gas 4. Production: Strong Base use, Open flame, Toxic vapor Testing: Toxic vapor Gas 5. Production: Strong Acid use, Open flame, Toxic vapor Testing: Toxic vapor Station 1 Nitric Oxide NO Station 1 Nitric Oxide NO Station 2 Hydrogen Gas H2 Station 2 Hydrogen Gas H2 Station 3 Oxygen O2 Station 3 Oxygen O2 Station 3 Oxygen O2 Station 4 Ammonia NH3 Station 4 Ammonia NH3 Station 5 Hydrogen Chloride HCl Station 5 Hydrogen Chloride HCl PRODUCTION and TESTING of FIVE GASES (ALWAYS WEAR GOGGLES) EACH GROUP IS TO PRODUCE 2 TEST TUBES OF EACH GAS. ONE TUBE IS TO RUN A PRACTICE TEST. THE SECOND TUBE IS TO TEST IN FRONT OF TEACHER. WHEN TEST IS SUCCESSFUL, TEACHER WILL SIGN-OFF ON THIS SHEET. *************************************************************************** WATER DISPLACEMENT—The first three gases will require the use of the trough set-up. PURGING(REMOVING AIR) MUST BE DONE BEFORE ATTEMPTING TO CATCH THE GAS. THE CATCH TUBE MUST BE FILLED WITH WATER BEFORE PURGED BUBBLES ARE ALLOWED TO ENTER THE TUBE. SET-UP #1 ____________ NITRIC OXIDE Production--A 5 GRAM SAMPLE OF COPPER IS PUT INTO TEST TUBE. 15 ml OF 6 M NITRIC ACID IS ADDED TO THE REACTION TEST TUBE AND THE STOPPER IS CLOSED QUICKLY. ALLOW TO PURGE FOR 15 SECONDS. CATCH 2 TEST TUBESFULL AND STOPPER THE GAS WITHOUT INHALING. Testing: Oxygen entering tube will convert NO(clear) to NO2(brown). SET-UP #2 ____________ HYDROGEN PRODUCTION--A 5 GRAM SAMPLE OF ZINC IS PUT INTO TEST TUBE. 10 ml OF 6 M. HCl IS ADDED TO THE TEST TUBE AND THE STOPPER IS CLOSED QUICKLY. ALLOW TO PURGE FOR 15 SECONDS. PURGING IS VERY IMPORTANT TO PREVENT EXPLOSION. STOPPER THE COLLECTION TUBE CONTAINING THE GAS. Testing: Holding the inverted test tube with a clamp, introduce a lighted splint to the mouth of the tube and a small “POP” should be heard. SET-UP #3 ____________ OXYGEN PRODUCTION--A 20 ml SAMPLE OF HYDROGEN PEROXIDE IS PUT INTO THE REACTION TEST TUBE. 10 DROPS OF THE FERRIC CHLORIDE SOLUTION (FeCl3) IS ALSO ADDED. COVER TUBE WITH STOPPER, HEAT WITH BUNSEN BURNER AND PURGE THE SYSTEM FOR 15 SECONDS. CATCH AND STOPPER THE GAS. Testing: Holding the inverted test tube with a clamp, introduce a glowing splint inserted 2 inches into the mouth of the tube and the splint should re-ignite. ************************************************************************** SET-UP #4 ___________ AMMONIA Production--ADD 10 DROPS OF AMMONIUM HYDROXIDE TO A TEST TUBE. HEAT OVER A BUNSEN BURNER UNTIL YOU SEE VAPOR RISING OUT OF THE TUBE. STOP HEATING IMMEDIATELY AND STOPPER QUICKLY. DO NOT INHALE VAPORS. Testing: Invert test tube into filled trough. Remove stopper while mouth of tube is submerged. Water should strongly suck up into tube. Restopper tube. Lift tube out of trough and place 1/5 of a stick of Red Litmus Paper into tube. It should turn blue. SET-UP #5 ____________ HYDROGEN CHLORIDE Production--ADD 10 DROPS OF CONCENTRATED HYDROCHLORIC ACID TO A TEST TUBE. HEAT THE TEST TUBE WITH A BUNSEN BURNER UNTIL VAPOR STARTS TO ESCAPE FROM THE MOUTH OF THE TUBE. STOP HEATING IMMEDIATELY AND STOPPER QUICKLY. DO NOT INHALE VAPORS. Testing: Invert test tube into filled trough. Remove stopper while mouth of tube is submerged. Water should strongly suck up into tube. Restopper tube. Lift tube out of trough and place 1/5 of a stick of Blue Litmus Paper into tube. It should turn pink. SAFETY PRECAUTIONS FOR THE 5 GAS LAB FOR ALL SET-UPS: 1. WEAR GOGGLES FOR ALL LABS 2. CAUTION WITH GLASSWARE 3. NO OPEN SHOES 4. NO CONTACTS Gas Preparation Test Nitric Oxide (Gas 1) 1. Toxic Vapor 1. Nitric Acid 2. Toxic Vapor Hydrogen (Gas 2) 1. 2. 3. Oxygen (Gas 3) Toxic chemical FeCl3 1. Open Flame Ammonia (Gas 4) 1. 1. Toxic vapor 2. Toxic chemical (NH4OH) Open Flame 1. 2. Toxic Chemical (HCl) Open Flame 1. Toxic Vapor Hydrogen Chloride (Gas 5) Explosive Purge Hydrochloric acid 1. 2. Explosive Open Flame SET-UP #1: NITRIC OXIDE 1. USE OF NITRIC ACID--ALL ACIDS SHOULD BE HANDLED WITH CARE NOT TO INHALE OR COME IN CONTACT WITH HUMAN TISSUE. 2. CARE WHEN USING Cu TURNINGS TO AVOID CUTTING ONESELF ON SHARP EDGES. 3. WHEN NITRIC ACID AND Cu ARE MIXED A TOXIC GAS IS PRODUCED, DO NOT INHALE VAPORS(BROWN VAPORS). SET-UP #2: HYDROGEN GAS 1. USE OF HYDROCHLORIC ACID--ALL ACIDS SHOULD BE HANDLED WITH CARE NOT TO CONTACT WITH HUMAN TISSUE. 2. PURGING IS A MUST TO REMOVE OXYGEN FROM THE SYSTEM BEFORE TESTING TO OF THE HYDROGEN-OXYGEN MIXTURE. 3. FLAME IS USED TO IGNITE SPLINT, CAUTION AROUND TISSUE AND HAIR. INHALE OR COME IN PREVENT EXPLOSION SET-UP #3: OXYGEN GAS 1. FLAME IS USED TO IGNITE SPLINT, CAUTION AROUND TISSUE AND HAIR. 2. DO NOT ALLOW CHEMICALS USED TO COME IN CONTACT WITH HUMAN TISSUE. SET-UP #4: AMMONIA 1. USE OF AMMONIUM HYDROXIDE--SHOULD NOT BE INHALED OR ALLOWED TO CONTACT SKIN OR OTHER TISSUES. 2. FLAME IS USED TO PRODUCE GAS, CAUTION AROUND TISSUE AND HAIR. SET-UP #5: HYDROGEN CHLORIDE 1. USE OF HYDROCHLORIC ACID--ALL ACIDS SHOULD BE HANDLED WITH CARE NOT TO CONTACT WITH HUMAN TISSUE. 2. FLAME IS USED TO PRODUCE GAS, CAUTION AROUND TISSUE AND HAIR. INHALE OR COME IN DENSITY, DATA COLLECTION, IDENTIFICATION OF UNKNOWN MASS DENSITY = -----------VOLUME STUDENT 1:__________________________________ STUDENT 2:__________________________________ STUDENT 3:__________________________________ PERIOD:_____________________ I. DENSITY OF A REGULAR OBJECT: AT EACH STATION, YOU WILL FIND AN OBJECT WITH REGULAR SIDES. A. MEASURE THE THREE SIDES OF THE OBJECT AND RECORD THE LENGTHS. 1.___________ 2.____________ 3.____________ B. CALCULATE THE VOLUME USING V = L x W x H IN CUBIC CENTIMETERS. VOLUME=________________ C. MEASURE THE MASS OF THE OBJECT IN GRAMS USING THE TRIPLE BEAM BALANCE. MASS=_________________ D. CALCULATE THE DENSITY OF THE OBJECT: _____________________g/ml II. DENSITY OF AN IRREGULAR OBJECT: AT EACH STATION, YOU WILL FIND A RUBBER STOPPER. A. FILL THE OVERFLOW CAN TO THE VERY TOP UNTIL DROPS FALL FROM THE SPOUT. SLOWLY ADD THE OBJECT AND CATCH THE WATER THAT FALLS FROM THE SPOUT IN A GRADUATED CYLINDER. MEASURE AND RECORD THE VOLUME OF THE OBJECT. VOLUME=_____________________ B. MEASURE THE MASS OF THE OBJECT IN GRAMS USING THE TRIPLE BEAM BALANCE. MASS=_____________________ C. CALCULATE THE DENSITY OF THE OBJECT: _____________________g/ml III. DENSITY OF A LIQUID: A. WEIGH AN EMPTY GRADUATED CYLINDER. RECORD THE MASS=____________. B. MEASURE A VOLUME OF LIQUID. RECORD THE VOLUME=____________. C. WEIGH THE FILLED GRADUATED CYLINDER. SUBTRACT THE EMPTY CYLINDER MASS. THE DIFFERENCE IS THE MASS OF THE LIQUID. MASS OF FILLED CYLINDER=__________________ MASS OF EMPTY CYLINDER=__________________ MASS OF WATER CONTAINED=__________________ D. CALCULATE THE DENSITY OF THE LIQUID: _____________________g/ml (REMEMBER TO USE THE MASS AND VOLUME OF THE LIQUID ONLY) IV. IDENTIFY AN UNKNOWN LIQUID FROM THE DATA AVAILABLE: UNKNOWN NUMBER_____________________ DENSITY OF THE UNKNOWN_____________________ IDENTITY OF THE UNKNOWN_____________________ Flame Test Lab Chemistry 1st Semester Introduction: According to the Bohr Theory of the atom, electrons may occupy only specific energy levels (ground state). When an atom absorbs sufficient energy, an electron can “jump” to a higher energy level and if a lower energy level is available, the electron will “fall” back, giving off excess energy in the process. The difference in energy between the two levels is emitted in the form of a Photon of electromagnetic radiation. The energy of each photon is described in terms of quanta, which can be calculated using the equation E = hv, where E is energy, h is Planck’s constant, and v is frequency of the radiation. This equation was derived by Max Planck in the early 1900’s. If the wavelength of the released photon is between 400 micrometers and 700 micrometers, it is considered to be Visible Light. The color of the light depends on the energy lost as the electrons fall back to ground state. White light is known as a continuous spectrum which contains all the colors of the rainbow. An excited atom produces one or more specific color(s) depending on the energy changes of it’s electrons as they fall back down to ground state. Because each element has a specific electron configuration, each element has a unique line spectrum. Flame tests are a quick way of producing characteristic colors of metallic ions. The loosely held electrons of a metal are easily excited in a flame and produce their specific color(s) as the electrons fall back to ground state. The color is a combination of the wavelengths of each electron energy transition and may be used to identify the elements. GOGGLES MUST BE WORN Conclusions/Questions: Include a copy of your Data Table and unknown evaluations. 1. How can you be sure the color is not due to the negative ion? 2. E.T. comes to visit you and brings along some alien crystals. How could you identify the elements in these crystals? 3. When a pan of milk boils over onto the stove the flame turns red-orange. Explain why. 4. A firework contains copper chloride and strontium sulfate. What colors will this explosive produce? 5. What colors would you expect the following compounds to produce: potassium chloride, potassium nitrate, and potassium sulfate? Why? 6. How is spectrometry used to determine the composition of stars? 7. What is the identity of your two unknowns? A. __________________ B. __________________ PERIODIC TABLE LAB--RELATIONSHIPS Student Name: ________________________________________________________ Period: _________ PURPOSES -- Students will: 1. use research methods to find the data needed to determine if relationships occur on the Periodic Table between the elements in row 3 and between the elements in column 17 as related to a particular property. 2. use the information researched to prepare data charts in Excel and then use those spreadsheets to prepare CHARTS applying the graphing techniques in EXCEL. 3. use skills taught in math to interpret the graphs. “As Atomic Number increases, the property…” Each student will pick a different property from the list below. Students will use their books and Internet sources to define and locate data related to their chosen property for row 3 and column 17 elements. Record the data in an Excel Spreadsheet. The student will then use the spreadsheet to prepare a CHART in Excel. They will interpret the data in the graph to look for relationship or trends of the property. DECIDE IF RELATIONSHIPS DO EXIST AND WHY. Internet research will help with finding the reason for the relationship too. Graphs must contain: 2. 3. 4. 5. 6. Main title Labels for X and Y axis Legend Copy of the spreadsheet w/ data Written explanation for the trend TEACHER: Have each student pick a different topic below. GROUP 1-- ATOMIC MASS vs ATOMIC NUMBER GROUP 2-- ATOMIC RADII vs ATOMIC NUMBER GROUP 3-- IONIC RADII vs ATOMIC NUMBER GROUP 4-- DENSITY vs ATOMIC NUMBER GROUP 5-- IONIZATION ENERGY(IP)(1st) vs ATOMIC NUMBER GROUP 6-- SPECIFIC HEAT vs ATOMIC NUMBER GROUP 7-- BOILING POINTS vs ATOMIC NUMBER GROUP 8-- MELTING POINTS vs ATOMIC NUMBER GROUP 9-- ELETRONEGATIVITY vs ATOMIC NUMBER GROUP 10-- ELECTRON AFFINITY vs ATOMIC NUMBER Assessment: Use this cover sheet and staple in the following to turn in to the teacher after you present information to the class: 1. Define your property. 2. Turn in a copy of each spreadsheet with Data and Labels. 3. Turn in a copy of the properly labeled graphs from Excel. 4. Explain what trends you have found from the graphs. 5. Explain why the property has that trend. Periodic Table Relationship lab -- Presentation notes GROUP 1-- ATOMIC MASS GROUP 2-- ATOMIC RADII GROUP 3-- IONIC RADII GROUP 4-- ATOMIC DENSITY GROUP 5-- IONIZATION ENERGY(FIRST) GROUP 6-- IONIZATION ENERGY(SECOND) GROUP 7-- BOILING POINTS GROUP 8-- MELTING POINTS GROUP 9-- ELETRONEGATIVITY GROUP 10-- ELECTRON AFFINITY Types of Chemical Reactions Lab Purpose: The purpose of this lab is to gain a better understanding of the types of chemical reactions by performing them in the lab. You MUST wear goggles and closed –toed shoes AT ALL TIMES!! There are other specific safety precautions throughout this lab that are noted below. Be prepared!! In your lab write up, you will write the reactions that you perform. You will record any Qualitative Observations as the reactions are occurring !!! If a precipitate is formed by any of the reactions, you should use your solubility rules to figure out what the precipitate is!! You will be testing for certain substances in some cases: Positive test for a base: solution turns pink in the presence of Phenolphthalein OR Litmus paper turns blue. Positive test for an acid: solution turns clear in the presence of Phenolphthalein OR Litmus paper turns red Positive test for Carbon Dioxide: burning splint is extinguished in an inverted test tube Positive test for Oxygen: glowing splint is reignited in an inverted test tube Positive test for Hydrogen: Explodes in the presence of Oxygen and energy. Positive test for Bromine: Turns to a gold or orange in the presence of Hexane. Positive test for Iodine: Turns to a pink or purple in the presence of Hexane. Dispose of all liquids down the drain and solids in the trash can unless otherwise noted. **CAUTION: LOOK AWAY FROM BURNING MAGNESIUM. TIE HAIR BECAUSE OF OPEN FLAME** Station 1: You will perform a synthesis reaction by burning Magnesium ribbon (Magnesium in the presence of Oxygen). Use crucible tongs, and hold the Mg in the hottest part of the flame. When major changes occur, DO NOT LOOK DIRECTLY AT THE FLAME!!! Catch the product in an evaporating dish and observe. Important to note: when you light the Bunsen burner, you are performing a combustion reaction. The flame you see is the result of Methane (CH4) combusting. Write this reaction, too. **CAUTION AROUND OPEN FLAME** Station 2: You will perform a single replacement and then a synthesis reaction. Put a few grams of Zinc into an Erlenmeyer Flask. Add 20 ml of 6M HCl and quickly cover the mouth of the flask with a balloon. Allow the balloon to capture the product (H2(g)) until balloon is about 6 inches in diameter. Tie the balloon. Tape the balloon to a meter stick and hold at arm’s length over a Bunsen Burner. You will burn through the balloon and expose the H2 to O2 of the air with a source of energy and produce water vapor. ** CAUTION! Oxygen gas supports combustion** Station 3: You will perform a decomposition reaction. Hydrogen Peroxide (H2O2) will be decomposed into water and Oxygen gas by the presence of a catalyst (Manganese II oxide) The catalyst is not used up in the reaction, it helps speed up the decomposition. - Put about 1 inch of hydrogen peroxide into a test tube along with a pea sized amount of the catalyst, Manganese II Oxide - Immediately place a rubber stopper with a rubber hose into the test tube. - Purge the system by allowing bubbles to escape for 15 seconds. - Lay a test tube on its side under water until all bubbles have moved out the open end. Raise the tube from the closed side so that it is full of water. Slide tube over the hole to catch the bubbles until all liquid is out. - Bring a GLOWING splint to the mouth of the inverted test tube. Stick the GLOWING splint up into the tube and observe. - Stop the reaction in the reaction test tube by adding water. Remember, liquids down the drain, solids in the trash. - Record your observations **CAUTION: COPPER COMPOUNDS ARE TOXIC** Station 4: You will perform a single replacement reaction by reacting Zinc with Copper(II) Sulfate. You will add 1/2 inch of the Copper(II) Sulfate solution into a test tube. Drop a small piece of Zinc into the test tube. Note any changes of appearance in metal. Pour liquid waste into the “Copper Waste” Flask and the solid in the trash can. **CAUTION!! CHLORINE IS TOXIC! DO NOT INHALE. REPLACE CAP IMMEDIATELY AFTER USING** **Discard contents of test tubes into container marked “WASTE”** Station 5: You will perform 2 separate single replacement reactions. In separate test tubes, add the following: a. 1 ml Sodium Iodide and 1ml of Chlorine water b. 1 ml Potassium Bromide and 1 ml Chlorine water To each tube add 1 ml of Hexane and shake vigorously. Observe and record the colors of the layers. Pour contents of tubes into “Hexane WASTE” container. Rinse the test tubes. **CAUTION: DO NOT GET THESE CHEMICALS ON HUMAN TISSUE** Station 6: You will perform a double replacement reaction by putting 10 drops Barium chloride into 2 test tubes with: a) 10 drops of Potassium Chromate into tube A and b) 10 drops of Potassium Dichromate into tube B. You should do the reactions separately in the provided test tubes. Determine whether or not a precipitate forms, and if so, what is the precipitate (indicate with an (s) in the reaction). Rinse out the test tubes. **CAUTION!!! Silver Nitrate can BURN skin and cause discoloration!! Be careful when handling!!** Station 7: You will perform a double replacement reaction by placing 10 drops of Silver Nitrate into two test tubes. C. add 10 drops of Potassium Chromate into tube A D. 10 drops of Potassium Dichromate into tube B. You should do the reactions separately in the provided test tubes. Determine whether or not a precipitate forms and record its formula. Rinse out the test tubes. **CAUTION!! TOXIC!! Lead Compounds! Take great care in handling!** Station 8: You will perform a double replacement reaction by placing 10 drops of Lead (II) Nitrate into two test tubes. b. Then add 20 drops of Potassium Chromate to tube A c. 10 drops of Potassium Dichromate to tube B. You should do the reactions separately in the provided test tubes. Observe to determine whether or not a precipitate forms and record its formula. Discard the Lead residues into the “Lead Waste” container. Rinse out the test tubes. **CAUTION!! Silver nitrate can burn human tissue!! Lead is poisonous!!*** Station 9: You will perform 2 separate double replacement reactions. a. Sodium Phosphate and Silver Nitrate in test tube A b. Sodium Phosphate and Lead (II) Nitrate in test tube B Add 10 drops of each and note your observations. . Discard the Lead residues into the “Lead Waste” container . **CAUTION!! Take great care when working with acids and bases!!** Station 10: This is a neutralization reaction to be performed at this station. Add 10 drops of HCl to your test tube. Then add one drop of an indicator called Phenolphthalein. You should then add the NaOH and COUNT THE DROPS YOU ADD UNTIL A PERMANENT COLOR CHANGE HAS OCCURRED!! Write down your observations. **CAUTION: USE OF AN ACID! DO NOT GET ON YOUR SKIN! CAN BURN!** Station 11: You will perform a miscellaneous reaction by reacting Sodium Carbonate & Acetic Acid. Procedure: Put a test tube in the water and make sure it is completely filled with water and no gas bubbles. You should then add about an inch of the acid to the test tube attached to the ring stand, and then add the Sodium Carbonate. Put the rubber stopper connected to the tubing on top of the test tube. You will purge the gas for around 10 seconds. Place the water filled test tube over the hole in the container to collect the gas. Once the test tube is filled with gas, move it over the rubber stopper and stopper the test tube. Bring the test tube over to the fume hood and put a burning splint into the mouth of the UPRIGHT test tube. Stop the reaction in the test tube by adding water. Note the results. Types of Chemical Reactions Lab -- Write up Safety Precautions and Reactions for the Types of Reactions Lab: KEY Station 1: Station 2: Hydrogen can explode. Acid should not contact human tissue. Open flame causes the need for caution and tied up hair. (1) 2H2 + O2 2H2O (3c) Zn + (6a) 2HCl H2 + ZnCl2 CH4 + 2O2 CO2 + 2H2O Station 3: Station 4: Copper is Toxic, do not allow to come in contact with human tissue. (3a) Zn + CuSO4 Cu + ZnSO4 Station 5: Chlorine and Iodine are Toxic, do not inhale. (3d) Cl2 + 2NaI I2 + 2NaCl (3d) Cl2 + 2NaBr Br2 + 2NaCl Station 6: Chromates and Dichromates should not come in contact with human tissue. (4) BaCl2 + Na2CrO4 BaCrO4(s) + 2NaCl (4) BaCl2 + Na2Cr2O7 BaCr2O7(s) + 2NaCl Station 7: AgNO3 will burn your skin and should not come in contact with human tissue. Chromates and Dichromates should not come in contact with human tissue. (4) AgNO3 + Na2CrO4 Ag2CrO4(s) + 2NaNO3 (4) AgNO3 + Na2Cr2O7 Ag2Cr2O7(s) + 2NaNO3 (4) AgNO3 + Na2Cr2O7 Ag2Cr2O7(s) + 2NaNO3 Station 8: Pb(NO3)2 is toxic and should not come in contact with human tissue. Chromates and Dichromates should not come in contact with human tissue. (4) Pb(NO3)2 + Na2CrO4 PbCrO4(s) + 2NaNO3 (4) Pb(NO3 )2 + Na2Cr2O7 PbCr2O7(s) + 2NaNO3 Station 9: AgNO3 will burn your skin and should not come in contact with human tissue. (4) 3AgNO3 + Na3PO4 Ag3PO4(s) + 3NaNO3 (4) Pb(NO3)2 + Na2Cr2O7 PbCr2O7(s) + 2NaNO3 Station 10: Acids and Bases should not be allowed to contact human tissue because they burn. (5) Station 11: HCl + NaOH H2O + NaCl Names of Group Members Period I. Title of Lab II. Purpose III. Safety Precautions for each individual station ( Stations 1-11). IV. Qualitative observations for each individual station. V. Answer the following questions. Be sure to include both question and answer. Answer using complete sentences. 1. You collected various gases in this lab. Explain how testing with the splint confirmed which gas was present in each case. 2. Explain how you knew when neutralization has been reached by using Phenolphthalein. 3. Explain how the single replacement reactions occurred. 4. Which reactions released gases? 5. Which reactions produced solids? 6. Which station had a quantitative result? 7. What did the color change in Station 5 indicate? VI. Conclusion a. For each station, write the type of equation, word equation(s) and chemical equation(s) that occurred. b. Explain any problems you may have had doing this lab and what you did to remedy them. c. Tell me which reaction you enjoyed doing the most and why. **CAUTION: LOOK AWAY FROM BURNING MAGNESIUM. TIE HAIR BACK BECAUSE OF THE OPEN FLAME** Station 1: You will perform a synthesis reaction by burning Magnesium ribbon (Magnesium in the presence of Oxygen). Use crucible tongs, and hold 1 inch of the Mg in the hottest part of the flame. When major changes occur, DO NOT LOOK DIRECTLY AT THE FLAME!!! Catch the product in an evaporating dish and observe. Important to note: when you light the Bunsen burner, you are performing a combustion reaction. The flame you see is the result of Methane (CH4) combusting. Write this reaction, too. **CAUTION AROUND OPEN FLAME, Long hair should be tied up** **CAUTION: HYDROGEN GAS CAN EXPLODE** **CAUTION: ACIDS SHOULD NOT BE ALLOWED TO COME IN CONTACT WITH HUMAN TISSUE** Station 2: You will perform a single replacement to produce the Hydrogen and then a synthesis reaction while exploding the Hydrogen to produce water. Put a few grams of Zinc into an Erlenmeyer flask. Add 20 ml of 6M HCl and quickly cover the mouth of the flask with a balloon. Allow the balloon to capture the product (H2(g)) until balloon is about 6 inches in diameter. Tie the balloon. Tape the balloon to a meter stick and hold at arm’s length over a Bunsen Burner. You will burn through the balloon and expose the H2 to O2 of the air with a source of energy and produce water vapor. Stop the reaction by adding tap water to the flask. Pour out the water in the sink. Any solid Zn left over can be left in the flask for the next group. ** CAUTION! Oxygen gas supports combustion** Station 3: You will perform a decomposition reaction. Hydrogen Peroxide (H2O2) will be decomposed into water and Oxygen gas by the presence of a catalyst (Manganese II Dioxide) The catalyst is not used up in the reaction, it helps speed up the decomposition. - Put about 1 inch of hydrogen peroxide into a test tube along with a pea sized amount of the catalyst, Manganese II Dioxide - Immediately place a rubber stopper with a rubber hose into the test tube. - Purge the system by allowing bubbles to escape for 15 seconds. - Lay a test tube on its side under water until all bubbles have moved out the open end. Raise the tube from the closed side so that it is full of water. Slide tube over the hole to catch the bubbles until all liquid is out. - Bring a GLOWING splint to the mouth of the inverted test tube. Stick the GLOWING splint up into the tube and observe. - Stop the reaction in the reaction test tube by adding water. Remember, liquids down the drain, solids in the trash. - Record your observations **CAUTION: COPPER COMPOUNDS ARE TOXIC** **Discard the liquid into the “Copper Waste” container.** Station 4: You will perform a single replacement reaction by reacting Zinc with Copper(II) Sulfate. You will add ½ inch of the Copper(II) Sulfate solution into a large test tube. Drop a small pellet of Zinc into the test tube. Note any changes of appearance in metal. Discard the liquid into the “Copper Waste” container and solid in the trash can. **CAUTION!! CHLORINE IS TOXIC! **DO NOT INHALE VAPORS.** **REPLACE CAP IMMEDIATELY AFTER USING** **Discard contents with HEXANE into container marked “WASTE”** Station 5: You will perform 2 separate single replacement reactions. In separate test tubes, add the following: a. 1 ml Sodium Iodide and 1ml of Chlorine water b. 1 ml Potassium Bromide and 1 ml Chlorine water To each tube add 1 ml of Hexane and shake vigorously. Observe and record the colors of the layers. Pour contents of tubes into “WASTE” container. Rinse the test tubes. **CAUTION: DO NOT GET THESE CHEMICALS ON HUMAN TISSUE** Station 6: You will perform a double replacement reaction by putting 10 drops Barium chloride into 2 test tubes with: a) 10 drops of Potassium Chromate into tube A and then b) 10 drops of Potassium Dichromate into tube B. You should do the reactions separately in the provided test tubes. Determine whether or not a precipitate forms, and if so, what is the precipitate (indicate with an (s) in the reaction). Rinse out the test tubes. **CAUTION!!! Silver Nitrate can BURN skin and cause discoloration so it should not be allowed to contact human tissue!! Be careful when handling!!** Station 7: You will perform a double replacement reaction by placing 10 drops of Silver Nitrate into two test tubes. 1. 2. Then add 10 drops of Sodium Chromate into tube A Add 10 drops of Sodium Dichromate into tube B. You should do the reactions separately in the provided test tubes. Determine whether or not a precipitate forms and record its formula. Rinse out the test tubes. **CAUTION!! TOXIC!! Lead Compounds should not contact human tissue!** **Take great care in handling! Discard into Lead waste container** Station 8: You will perform a double replacement reaction by placing 10 drops of Lead (II) Nitrate into two test tubes. 1. 2. Then add 10 drops of Potassium Chromate to tube A Add 10 drops of Potassium Dichromate to tube B. You should do the reactions separately in the provided test tubes. Observe to determine whether or not a precipitate forms and record its formula. Discard the Lead residues into the “Lead Waste” container. Rinse out the test tubes. **CAUTION!! Silver nitrate can burn human tissue!!** **Lead is poisonous and should not contact human tissue!!*** Station 9: You will perform 2 separate double replacement reactions. a. Sodium Phosphate and Silver Nitrate in test tube A b. Sodium Phosphate and Lead (II) Nitrate in test tube B Add 10 drops of each and note your observations. . Discard the Lead residues into the “Lead Waste” container which is on Station 8(next table). **CAUTION!! Take great care when working with acids and bases!! They should not be allowed to contact human tissue!!** Station 10: This is a neutralization reaction to be performed at this station. Add 10 drops of HCl to your test tube. Then add one drop of an indicator called Phenolphthalein. You should then add the NaOH and COUNT THE DROPS YOU ADD UNTIL A PERMANENT COLOR CHANGE HAS OCCURRED!! Write down your observations. **CAUTION: USE OF AN ACID! DO NOT GET ON YOUR SKIN! CAN BURN!** Station 11: You will perform a miscellaneous reaction by reacting Sodium Carbonate & Acetic Acid. Procedure: Put a test tube in the water and make sure it is completely filled with water and no gas bubbles. You should then add about an inch of the acid to the test tube attached to the ring stand, and then add the Sodium Carbonate. Put the rubber stopper connected to the tubing on top of the test tube. You will purge the gas for around 10 seconds. Place the water filled test tube over the hole in the container to collect the gas. Once the test tube is filled with gas, move it over the rubber stopper and stopper the test tube. Bring the test tube over to the fume hood and put a burning splint into the mouth of the UPRIGHT test tube. Stop the reaction in the test tube by adding water. Note the results. Mole Concept Flip Chart Lab Purpose: To produce a flip chart that relates to Mole Concept information to be used to explain mole applications. Safety Precautions: Goggles not needed. Caution using scissors. Materials: 2-4 sheets of white paper, stapler, scissors, colored pencils, markers, tape and Mole Concept information, class notes, possibly text book Instructions: Create a 2-4page hot dog folded flip chart stapled with 4 to 7 partitions based upon your research. When cutting partitions, you should not cut the last page to provide strength to your chart. This chart needs to be able to last for many months. You may want to use colored pencils or markers to create emphasis and an attractive product. You may put anything on the chart about moles EXCEPT examples of worked problems. No dimensional analysis will be visible. You could put a verbal description of solutions but no numbers allowed. I will be checking the final product on the day of the Moles test. Information that should be included (minimum): Name and period Define moles (3 ways) a. Avogadro’s number b. Mass of a mole 1. Atomic Weight 2. Molar Mass c. Volume of a mole under STP conditions 1. Standard temperature 2. Standard pressure How to calculate: a. moles b. molecules c. atoms d. masses of parts e. using molar volume: moles, mass, particles of gas Empirical Formula Molecular formula SILVER NITRATE LAB--DATA & CALCULATIONS NAME___________________________ NAME___________________________ DATE_____________________ NAME___________________________ CLASS _____________________________ SECTION _________ 1. MASS OF ORIGINAL AgNO3 (remove any container mass):___________________ 2. MASS OF DRY Ag CRYSTALS PRODUCED: _____________________________ 3. MASS OF Cu CONSUMED IN LAB (before – after): _________________________ 4. MASS OF AgNO3 RECLAIMED : ________________________________________ 5. MASS OF AgCl PRODUCED: ___________________________________________ 6. MOLES OF ORIGINAL AgNO3: _________________________________________ 7. MOLES OF DRY Ag CRYSTALS PRODUCED: ____________________________ 8. MOLES OF Cu CONSUMED IN LAB: ____________________________________ 9. MOLES OF AgNO3 RECLAIMED : ______________________________________ 10. MOLES OF AgCl PRODUCED: ________________________________________ 11. NUMBER OF ATOMS OF SILVER PRODUCED: _________________________ 12. NUMBER OF ATOMS OF COPPER CONSUMED: ________________________ 13. RATIO: MOLES OF AgNO3(ORIGINAL) ----------------------------------------- = _____________________________ MOLES OF Cu CONSUMED 14. Problems encountered in completing the lab:____________________________________________________________ ____________________________________________________________________________________________ ____________________________________________________________________________________________ ____________________________________________________________________________________________ 15. How I solved the problems encountered above__________________________________________________________ ___________________________________________________________________________________________ ___________________________________________________________________________________________ SILVER NITRATE LAB INSTRUCTIONS Mass of original SILVER NITRATE=____________g Mass of Cu WIRE BEFORE REACTION=________g Mass of Cu WIRE AFTER REACTION=_________g Mass of EMPTY BEAKER=___________________g Mass of Ag CRYSTALS=_____________________g Mass of RECLAIMED AgNO3=________________g Mass of AgCl PRODUCED=___________________g Name_________________________________________ Name_________________________________________ Name_________________________________________ Class ___________________________ Section _______ Day 1 Turning AgNO3 into metallic Silver 1. Mass and Record the mass of the ORIGINAL SILVER NITRATE. Be sure to remove any container mass. 2. Obtain a piece of Cu wire approximately 30 cm long. Coil the wire around a pencil. MASS AND RECORD THE WIRE. 3. Chemistry clean, dry, label, MASS AND RECORD A 250 or 150 ml Beaker. 4. Chemistry clean a test tube(18x150). Pour the AgNO3 crystals into the test tube. 5. To the test tube w/ AgNO3, pour distilled water into test tube until it is no more than ¾ full. Stir with stirring rod, round and round, until TOTALLY dissolved. USE A FEW DROPS OF FROM WASH BOTTLE WATER TO RINSE OFF STIRRING ROD INTO TEST TUBE. 2. BE CAREFUL WITH THE STIRRING ROD. IT CAN TURN YOU BLACK TOO. 3. Insert the Cu wire into the test tube and allow it to sit undisturbed OVERNIGHT. Record any observations of what is happening. DAY 2 Collecting Silver crystals 1. Shake or scrape all remaining crystals off of the Cu into the test tube. MASS AND RECORD THE DRY WIRE. 2. Pour ALL contents of the test tube into the labeled beaker. You may need to hold the test tube inverted and “THUMP” it. You may squirt a minimal amount of distilled water to empty the tube if needed. Allow to sit for 1 minute to allow particles to settle. 3. Gently pour off the liquid while retaining the solid Silver in the beaker. This is called “decanting”. 4. Gently pour 10 ml of distilled water into the beaker, stir and decant again to wash the Silver crystals. 5. Repeat Steps 3 & 4 if needed to remove too much blue color. 6. Put the beaker into the drying oven as instructor designates. DAY 3 – DO NOT WEAR CONTACT LENSES TODAY! Using silver crystals and Nitric Acid to reclaim AgNO3 1. Get your beaker from the drying oven. MASS AND RECORD the Ag after removing beaker mass. 2. Add 20 ml OF 6M HNO3 into the beaker. 3. Place the beaker onto warm hot plate and stir (without boiling) until solids are dissolved. FIZZ BUT DON’T BOIL! DON’T INHALE TOXIC VAPORS WHILE STIRRING. OPEN DOORS, WINDOWS AND FANS WILL EXHAUST ROOM. 4. Place the beaker in the tray by the open window to dry until the next day. A heat lamp will be used to speed the drying. DAY 4 – Turning AgNO3 into AgCl 1. Obtain dry beaker with your RECOVERED SILVER NITRATE from the tray. MASS AND RECORD THE RECLAIMED AgNO3 after removing the beaker mass. 2. Add 20 ml of distilled water to the beaker with AgNO3 and stir until dissolved. Contaminates may not dissolve well. 3. To another beaker, add 3 g Of NaCl. Then measure and add 20 ml of distilled water to the beaker and stir until dissolved. 4. Pour the salt solution beaker into the pre-weighed AgNO3 solution beaker and stir. A solid precipitate will form(suspended). 5. Place beaker onto a HOT PLATE. Heat until it boils and the liquid becomes clear (you can see through the liquid). Most of the solid should clump up. Color does not count. 6. After heating, pour all contents of beaker into a filter and allow the liquid waste to drip out. Use additional (minimal amounts) distilled water if needed to get all solid out of the beaker. 7. Add 20 ml of additional distilled water and swirl to rinse the precipitate. Repeat of STEP 6 might be needed if blueish. 8. Place preweighed beaker with filter paper and solid Silver Chloride into drying oven overnight to dry. . DAY 5 1. Obtain your beaker with AgCl from drying oven. 2. MASS AND RECORD the AgCl residue after removing the beaker mass. 3. After checking the Data and Calculations page with instructor, scrape out AgCl residue into SILVER WASTE CONTAINER. SILVER NITRATE LAB--CONCLUSIONS NAME___________________________ NAME___________________________ NAME___________________________ CLASS : ____________________ SECTION__________ DATE___________________________ SILVER NITRATE LAB --- PERCENTAGE RESULTS CALCULATE THE FOLLOWING AS PERCENTAGES: 1. (| 1 - (MOLES OF SILVER/MOLES SILVER NITRATE)|)*100=__________________ 2. (| 1 - (MOLES OF AgNO3 RECLAIMED/MOLES AgNO3)|) * 100 = ______________ 3. (| 2 - (MOLES SILVER NITRATE/MOLES OF Cu)|/2)*100 =__________________ 4. (| 1 - (MOLES OF AgCl/MOLES SILVER NITRATE)|)*100=__________________ 5. AVERAGE OF PERCENTAGES: ( 1 + 2 + 3 + 4 ) / 4 = ____________________ 6. WHAT IS THE CAUSE OF THE BLUE SOLUTION DURING Ag FORMATION? 7. SPECIFICALLY, WHAT WOULD HAPPEN TO YOUR LAB RESULTS IF SOME OF THE AgNO3 SPILLS FROM PREWEIGHED WEIGHING BOAT AS YOU ARE POURING INTO TEST TUBE? 8. WHAT COULD HAPPEN TO YOUR RESULTS IF DURING THE LAB, YOU NOTICE THAT YOUR FINGERS HAVE TURNED DARK FROM EXPOSURE TO AgNO3? 9. WHAT WOULD HAPPEN TO YOUR RESULTS IF YOUR BEAKER CONTAINING Ag CRYSTALS HAD A LARGE AMOUNT OF BLUE IN IT? 10. WHAT HAVE YOU NOTICED ABOUT THE RELATIONSHIP OF THE MOLES OF BEGINNING AgNO3, THE MOLES OF Ag METAL, THE MOLES OF AgNO3 RECLAIMED AND THE MOLES OF AgCl? Stoichiometry and Reactions of Copper Compounds Objectives: To demonstrate the conservation of matter in a series of chemical reactions. Percent yield can be close to 100% Make observations of chemical change. Learn and practice good laboratory techniques. Introduction: In order for this experiment to have a meaningful result, one must be careful not to lose or discard any of the copper containing materials during the course of this experiment. The following reactions will be carried out sequentially: TO BE BALANCED THE DAY BEFORE THE LAB !!! → Cu+2 (aq) + NO2 (g) + H2O (l) 1. Cu (s) + H+1 (aq) + NO3-1 (aq) Cu+2 (aq) + Cu(OH)2 (s) OH-1 (aq) → → CuO(s) Cu(OH)2 (s) + H2O (l) CuO(s) + H+1 (aq) + HSO4-1 (aq) Cu+2 (aq) + Zn (s) Zn (s) + Cu+2 (aq) + NH3 (aq) 3. → Cu+2 (aq) + SO4-2 (aq) + H2O(l) 4. → Cu (s) + Zn +2 (aq) H+1 (aq) → 2. Zn+2 (s) + H2 (g) → [Cu(NH3)2]+2 5. 6. 7. Experimental Procedure 1. Weigh a 250 mL beaker. Add about 1g Cu and reweigh the beaker. To the copper add 5 mL of concentrated HNO 3 IN THE HOOD !! NITROGEN DIOXIDE IS A BROWN POISONOUS GAS This will oxidize elemental copper to the cupric ion (reaction 1). Heat the solution gently on a ring stand until the copper is completely dissolved. Note the evolution of brown gas. Continue to heat the beaker driving off the excess HNO 3 by evaporation until only several drops of liquid remain and the mixture resembles a thick paste. Do not allow the mixture to turn black. If dryness is reached before all the copper has dissolved, add 1-2 mL of additional HNO3 and continue to heat until all the copper is dissolved and the excess acid has evaporated. Record your color changes and other observations on the data sheet. 2. Cool the beaker. Add 10mL of H2O and slowly add 8mL of 6M NaOH, about 2mL at a time (reaction 2). Record your observations on the data sheet. 3. The precipitate formed in the previous step can be converted to CuO by adding 50mL of distilled H2O and gently boiling for a few minutes. Stir Continuously (reaction 3). Record your observations. When the color change is complete, continue to heat for an additional 5 minutes. This additional heating allows the precipitate particles to become larger and settle more readily. This is known as the digestion of the precipitate. Remove the beaker from the heat and allow the precipitate to settle. Decant the liquid being careful not to discard any of the precipitate. Wash the precipitate once with about 50 mL of distilled water. Decant and discard the wash water. 4. Add 30 mL H2SO4 and heat gently if necessary to dissolve the CuO (Reaction 4). (If the solution was evaporated at this point, CuSO4 would be formed.) Record your observations on the data sheet. 5. In order to prepare elemental copper from this solution, it is necessary to reduce the Cu+2 ion to elemental copper, Cu0. This is accomplished by using any metal higher than Cu in the electromotive series, such as Zn, Al or Mg. For economy’s sake, Zn will be used here. Add 5g of Zn in small pieces, only a few pieces at a time, to the CuSO 4 solution and stir occasionally until the solution is colorless, indicating that the Cu+2 ion is gone from the solution. (May be done in the hood, reaction 5). A more sensitive check is obtained by adding a few drops of the colorless solution to a small test-tube containing 1 mL (20 drops) of concentrated ammonia. A deep blue solution will result if any Cu+2 is present (reaction 7). If no color is formed in this test, carefully decant the liquid and add 10mL of 6M HCl to the solid to use up the excess Zn (reaction 6). When the reaction is complete (no further hydrogen evolution), wash the copper with two 50 mL portions of distilled water followed by two 10mL portions of acetone. Caution: Extinguish all flames before obtaining or using acetone. Acetone is flammable. Dry in the oven for about 30 minutes. Cool and weigh the beaker and the copper. If time permits, dry for an additional 10-15 minutes, cool and reweigh the beaker to be sure that the copper is dry. It may also be dried by placing it in the fume hood with the fan running. The amount of copper recovered is directly proportional to your lab technique. You should be able to recover nearly all of the copper you started with !!! Data 1. 2. 3. 4. Mass of beaker (or flask) _____________________ Mass of beaker and copper _____________________ Mass of copper _____________________ Mass of beaker and copper after all reactions ______________________ 5. Mass of copper recovered ______________________ 6. % Yield _______________________ Substance Color Substance Color _______________ H2SO4 ______________ _______________ Zn (s) ______________ Cu+2 _______________ Zn+2 ______________ NO2 _______________ H2 (g) ______________ Cu(OH)2 (s) _______________ NH3 solution ______________ CuO (s) _______________ [Cu(NH3)4]+2(aq) ______________ Cu (s) HNO3 Observations Step 1: Step 2: Step 3: Step 4: Step 5: Step 6: Step 7: solution Questions for Copper Stoichiometry Lab 1. If you start with 1.25 g of Cu, calculate the amount that should precipitate. 2. If a student collected more Cu than he started with, what explanation(s) could be given for the error? 3. If a student collected less Cu than what she started with, what explanation could be given for the error? 4. Calculate the % error for your lab. 5. Tell which equations are redox equations. Identify the elements that are oxidized and those that are reduced. 6. Why couldn’t nitric acid be used to eliminate the excess zinc instead of using hydrochloric acid to do so? INTERNATIONAL DAY OF THE MOLE CELEBRATION (October 23) PROJECT EVALUATION I. STUDENT NAME____________________________________________________________________________________ CLASS _______________________________________________ SECTION __________________________________ II. DESCRIPTION OF PROJECT:_______________________________________________________________________ ____________________________________________________________________________________________________ ____________________________________________________________________________________________________ ____________________________________________________________________________________________________ III. HOW DOES THIS RELATE TO MOLES?______________________________________________________________ _____________________________________________________________________________________________________ _____________________________________________________________________________________________________ _____________________________________________________________________________________________________ IV. MUCH OF THIS GRADE COMES FROM EFFORT GIVEN IN THIS PROJECT. I NEED YOUR HELP IN DETERMINING THE AMOUNT OF EFFORT GIVEN ON THIS PROJECT. Activity Planning of Project Time involved (minutes) Teacher evaluation (Don’t write here) Research Material gathering Display (quality and clarity) Presentation to class (quality and clarity) 1-2 minutes V. [DESERVED GRADE]STUDENTS ARE TO CHOOSE FROM ONE OF THE FOLLOWING AREAS: (REMEMBER, THIS IS DESERVED GRADE NOT DESIRED GRADE) STUDENT CHOICE (PUT AN "X" ONLY) TEACHER CHOICE 100-96: _______ 95-90: _______ 89-86: _______ 85-80: _______ 79-76: _______ 75-70: _______ _____ _____ _____ _____ _____ _____ DOES NOT HAVE ONE "0" Formula of a Hydrate Lab Purpose: Hydrates have water built into the molecular structure of the compound. The water can be driven away by severe heating. The mass loss is due to water removal. Moles can be calculated and used to determine the actual formula of a Hydrate. Crucibles can be pre-dryed in mass in a drying oven overnight then stored in a desicator. Procedure: 1. Mass and record a clean dry empty crucible. 2. Fill crucible ¼ with a Hydrate. Mass and record. Record the name of the Hydrate that you are using. 3. Heat gently over Bunsen Burner for 5 minutes to check for violent dehydration. After that heating can be severe for 10 minutes. Allow time for crucible cooling then mass and record. The material is now an Anhydrous salt. 4. Heat again severely for 5 more minutes to check for additional water removal. Mass and record as second heating. If no more mass is lost, you are ready for calculations. If more heat was lost, repeat heating, cooling and massing until no more mass is lost. Use the lowest mass in your calculations. 5 Discard cool Anhydrous salt in the trash. Put crucible back into desicator. Calculations: Don’t forget to remove empty crucible mass from all calculations. 1. Calculate the mass of the Anhydrous Salt from its name. 2. Calculate the mass of the water driven away from the Hydrate by heating. 3. Calculate the number of moles of water driven off. 4. Calculate the number of moles of the Anhydrous salt left at the end. 5. Calculate the mole ratio: moles of water / moles of Anhydrous Salt in whole number ratio. Ex: 3 moles water / 1 mole Salt 6. State the formula of the Hydrate in this fashion: Salt . 3 H20 Replace the word salt with your formula and the 3 with your ratio. Formula of our hydrate: _______________________________________________ Types of Chemical Reactions Record your results on a separate sheet. Write the complete chemical reaction that you observed. I. Synthesis 1. Clean a piece of Cu wire with sand paper. Note the appearance of the wire. Using crucible tongs, hold the wire in a hot flame for 1-2 minutes. Compare the appearance before and after flame. 2. Using crucible tongs, hold a piece of Magnesium ribbon in the hottest part of a flame. When major changes occur, DO NOT LOOK DIRECTLY AT THE FLAME!!!! Catch the MgO in an evaporating dish and observe. II. Decomposition 3. Place 2 scoops of Calcium Carbonate into a dry test tube. Using a test tube holder, heat the tube over a hot flame for 2 minutes. Remove from flame. Light a splint and insert a burning splint into the test tube. Carbon Dioxide is known for its ability to put out flames. III. Single Replacement 4. Put 3 ml of 6M Hydrochloric Acid into each of 3 test tubes. Put a small piece of Magnesium, Aluminum and Zinc into the 3 tubes Note the differences and similarity of reaction. respectively. 5. Put 3 ml of Copper(II)Sulfate solution into a test tube. Add a small piece of zinc. Note any changes of appearance of metal or solution. 6. Put 1 ml of Potassium Iodide into a test tube. Add 15 drops of Hexane. Add 10 drops of Chlorine water. Shake vigorously. Record results. IV. Double Replacement 7. Add 2 ml of Lead Nitrate to a test tube. Add 2 ml of Sodium Iodide to the same test tube. Observe and record any changes. 8. Add 2 ml of Silver Nitrate to a test tube. Add 2 ml of Potassium tube. Observe and record any changes. 9. Chromate to the same test Repeat #7 using Potassium Dichromate. V. Neutralization 10. Add 2 ml of 6M H2SO4 to 2 ml 6M Sodium Hydroxide into a test tube. Feel VI. Miscellaneous 11. Ignite a Bunsen Burner with a striker. The flame you see is the result of a Hydrocarbon combustion of Methane (CH4). 12. Add 1 scoop of metallic carbonate to 2 ml of acid. Record the results. 13. Add 1 scoop of metallic sulfite to 2 ml of acid. Record the results. the outside of the test tube. PRECIPITATION, SOLUBILITY AND FORMULA LAB NAME___________________________ PERIOD______________________ NAME___________________________ DATE________________________ NAME___________________________ GRADE_______________________ USING THE FOLLOWING CHART, MIX THE REQUESTED COMBINATIONS. RECORD THE RESULTS OF THE MIXTURE. IF A PRECIPITATE FORMS, USE THE SOLUBILITY CHART AND THE COMMON ION CHART TO DETERMINE THE FORMULA OF THE PRECIPITATE. Pb(NO3)2 AgNO3 Ba(NO3)2 Sr(NO3)2 Co(NO3)2 Mg(NO3)2 ________________________________________________________________________________________ Na3PO4 1. 2. 3. 4. 5. 6. ________________________________________________________________________________________ K2CrO4 7. 8. 9. 10. 11. 12. ________________________________________________________________________________________ K2Cr2O7 13. 14. 15. 16. 17. 18. _______________________________________________________________________________________ K2CO3 19. 20. 21. 22. 23. 24. _______________________________________________________________________________________ Na2SO4 25. 26. 27. 28. 29. 30. _______________________________________________________________________________________ Precipitation Lab Name______________________ , ___________________, ______________________ Period______ Write the reactions for each precipitate using Solubility Chart and Common Ion Chart. 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 30. MOLAR VOLUME LAB PURPOSE: To find the volume of ONE MOLE OF HYDROGEN GAS from Magnesium ribbon and Hydrochloric Acid under known conditions of temperature and pressure. PROCEDURE: 1. CAUTION: HYDROCHLORIC ACID WILL BURN YOUR EYES AND SKIN. DO NOT TOUCH ACID. Measure 10 ml of 6M Hydrochloric Acid (HCl) in a graduated cylinder and pour into a clean Eudiometer or Buret. 3. Fill Eudiometer to bulging with Deionized water. 4. The instructor will give you some limits for the Magnesium length based upon the current Barometric Pressure. Measure the exact length of Magnesium ribbon and record. Magnesium ribbon has a mass of 1.1 grams/meter. 5. Fold the ribbon tightly and wrap with a thin copper wire until a cage is covering most of the Magnesium. DON'T MAKE THE CAGE TOO BIG BECAUSE IT WILL BLOCK THE OPENING OF THE EUDIOMETER. 7. Fill a 400-600ml beaker with tap water. 8. Obtain a 00 rubber stopper with a hole through the middle. ***NEXT STEP MUST BE DONE QUICKLY TO AVOID REACTION BEFORE INVERSION.*** 9. Insert Mg and Cu cage 1 inch into the mouth of the Eudiometer and stopper tightly so water is purged out of the hole in the stopper. The acid will still be at the bottom of the eudiometer as you invert it so it will not touch you. Cover the hole with your thumb, invert the tube and quickly submerge the stopper and tube into the 400-600 beaker. Remove your thumb ONLY after the stopper is submerged in the water. . 10. Mount a clamp and stand to hold the Eudiometer upright and allow the reaction to take place. Note the Concentration waves of acid as they flow down the tube. 11. After the stream of bubbles ceases, allow 5 minutes for the apparatus to come to thermal equilibrium. 12. Now acid is mixed with a large volume of water and diluted so it will not hurt you. Reach your thumb into the beaker and cover hole. Remove the tube from the beaker. . 13. Take the tube to the large cylinder and insert the stoppered end into the water in the cylinder and remove your thumb. Move the tube up and down in the cylinder until the two levels of water are equal. Measure and record the volume of wet Hydrogen gas that you collected. 14. Clean up all equipment. MOLAR VOLUME LAB (Done without Eudiometers) PURPOSE: To find the volume of 1MOLE of HYDROGEN GAS from Magnesium ribbon and Hydrochloric Acid under known conditions of temperature and pressure. PROCEDURE: 1. Fill a buret to bulging with water. Place a “00” stopper with 1 or 2 holes into the mouth of the tube and push in. Some water will purge out. Remove stopper. Pour all to the water in the buret into a 100ml Graduated cylinder. Measure and Record accurately the volume of the water in the cylinder. ______________________________________________ml 2. Pour 10 ml of 6M Hydrochloric Acid (HCl) into the buret. Slowly, so as not to mix the acid and water, finish filling the buret to bulging with distilled water. The acid should remain mostly at the bottom of the buret at this point. 3. The instructor will give you a measurement of Magnesium ribbon to use for this step. The amount depends on the current Barometric Pressure which we will Record as _____________ inches. The length of Magnesium ribbon measured should be less than _______cm. Record the exact length as _______________cm The mass of 1 meter of Mg ribbon is 1.1g. 4. Fold the Magnesium ribbon tightly twice and wrap with 1 arms length of thin Cu wire or thread until a cage is formed which covers the Magnesium. Leave a 5-10 cm tail of Cu wire free. 5. Fill a 400ml or 600ml beaker with tap water. 6. ****DO THIS STEP QUICKLY TO INSURE ACCURACY OF YOUR DATA**** Insert the Cu cage into the buret w/ 3 cm of Cu wire folded over the lip of the tube. Place the “00” stopper tightly into the buret. Some water should purge out of the hole(s) of the stopper. Use your thumb to cover the holes in the stopper and invert the buret into the large beaker filled with water. Remove your thumb. Use your stand to mount the buret in an upright position while standing in the beaker. 7. The acid will begin to fall through the tube because it is more dense than water. When it reaches the Magnesium a reaction will begin releasing H2(g) which will rise as bubbles. 8. Allow the bubbling until they stop then allow reaching thermal equilibrium by sitting undisturbed for 5 minutes in the stand. 9. The acid that fell through the stopper holes is very diluted and therefore will not hurt you unless you have open wounds. Stick your finger into the beaker and stop up the holes in the stopper before removing the tube from the water. 10. Carry the buret (stoppered by your finger) to the large cylinder. Place the end of the tube into the water in the cylinder before removing your fingers from the stopper holes. Move the buret up or down in the water until the level of the water in the buret and the level of the water in the cylinder are even. This is equalizing the Hydrogen pressure with the Barometric pressure. 11. Measure and Record the volume of the equalized, wet Hydrogen Gas. Remove the buret from the tall cylinder by replacing your finger under stopper and sealing. 12. Pour the volume of liquid remaining in the buret into a Graduated cylinder. Measure and Record the remaining volume of liquid. _________________________________ml 13. Record the temperature of the room where the tall cylinder of water has been sitting. ___________________________˚C 14. Rinse out buret, stopper, graduated cylinder and beaker. Return them to storage. 15. Go answer the questions on the Evaluation Sheet MOLAR VOLUME of HYDROGEN GAS LAB—Evaluation Sheet NAME____________________________________ NAME____________________________________ Section___________ NAME____________________________________ NAME____________________________________ DATA: Mass of Mg ribbon/meter: ___________________g/meter (from Instructor) Barometric pressure: ________________________inHg (from Instructor) converted to mmHg: __________________mmHg Room Temperature: _________________________˚C Length OF Mg ribbon actually used :_______________________cm 1. Calculate the volume of H2 (wet) gas produced by subtracting the answer of step 12 from the answer from step 1. 2. Use Dimensional Analysis to calculate the mass of the Mg ribbon you used from the length that you measured. 3. Calculate the MOLES OF Mg ribbon used. 4. Write the balanced equation for Mg metal reacting with HCl forming H2 and MgCl2. (Types of Chemical Reactions) 5. What is the mole ratio between H2 and Mg in the balanced equation in problem 4?. 6. Using the Vapor Pressure of water chart and the room temperature, find the Vapor pressure supplied by the water in the gas sample. 7. Find the pressure of the H2 (dry) by subtracting the water pressure from the total pressure in the flask. (Dalton’s) 8. Using the formula: NEW H2 vol.= ( H2 pressure(dry))(vol H2(wet))/(760mmHg) calculate the volume of H2 that would have been produced at Standard Pressure. 9. Calculate the volume of dry H2 that would have been produced using 1 mole of Mg at Standard Pressure and the temperature of the room. Set up a proportion. This is called MOLAR VOLUME of Hydrogen Gas at Standard Pressure and room temp. Molarity--Solution Making Lab Per______ Name___________________________________________________ Name___________________________________________________ Name___________________________________________________ Purpose: To learn how to prepare solutions of specific quantities of desired Molarity. Safety Precautions: See specific chemical warnings in MSDS or catalogs and fill in below. ___________________________________________________ _______________________________________________________ __________________________________________________________________________________________________________ __________________________________________________________________________________________________________ __________________________________________________________________________________________________________ __________________________________________________________________________________________________________ __________________________________________________________________________________________________________ Each group is to use a 250ml Volumetric flask to make a solution whose content and Molarity are listed on the storage bottle. Check chemical bottle for information about the hydrate formula if any. Name of chemical: __________________________________________ Molarity from the bottle: __________________________________________ Formula from bottle: __________________________________________ Calculations for making 250 ml of the Molarity listed on the storage bottle: Molar mass of formula: ________________________________________________ Moles of formula needed(show all work): __________moles Grams of formula needed(show all work): __________grams 1. 2. 3. 4. 5. 6. 7. 8. Use weighting boat or paper to weigh out the calculated amount of your chemical. Tare the mass of the boat or paper so you see only the chemical mass. Use a scoop to get the dry chemical into the volumetric flask. Caution is needed when putting chemicals into neck of volumetric flask. It is difficult to get chemicals into neck and spills will cause results to be incorrect and solutions to be unusable. Discard used weighing boat in trash. Add DISTILLED WATER to the volumetric flask to HALF FULL. Swirl until dissolved. Add DISTILLED WATER to the volumetric flask up to the fill line on the neck of the flask. Caution when adding water not to go above the fill line. This would invalidate your solution. Show the final product to your instructor. Pour the final product into the storage bottle. Rinse the scoop and volumetric flask with lots of tap water and final rinse with Distilled water. Return to proper locations. Formula of a Precipitate Lab Precipitate: a solid substance formed when ions from soluble solutions are mixed in a way that violates natures solubility rules. Their combinations are specific to the ions present. We will determine the formula. Wear goggles as always for all steps of the lab. (*** Lead toxicity ***) PROCEDURE: 1. Clean and label 8 test tubes (1-7) 2. Using coded burets, fill test tubes with the following contents: ------------------------------------------------------------------------------------------------------------------CODE\ Tube 1 2 3 4 5 6 7 8 ------------------------------------------------------------------------------------------------------------------I .5M Pb(NO3)2 1 2 3 4 5 6 7 8 ml ------------------------------------------------------------------------------------------------------------------II .5M NaI 8 7 6 5 4 3 2 1 ml ------------------------------------------------------------------------------------------------------------------3. Premass, fold & label (visible from outside) 8 sheets of filter paper. 4. Filter each test tube into the proper filter paper. Squeeze top of filter paper together when finished filtering. Stack the 8 filter papers from 1 to 8 into a labeled 150ml beaker and put into drying oven overnight. Questions: 1. Mass each of the 8 filter papers and precipitates individually and subtract the mass of the filter paper from each. Tube | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | --------------------------------------------------------------------------grams of precipitate | | | | | | | | | 2. Using the Molarity= mmoles / ml formula to calculate the following information and recreate the chart filled in on your paper. ----------------------------------------------------------------------------------------------------------------------------------------------------------------Tube | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 ------------------------------------------------------------------------------------------------------mmoles Pb(NO3)2 | | | | | | | | ------------------------------------------------------------------------------------------------------mmoles NaI | | | | | | | | ------------------------------------------------------------------------------------------------------2. 3. Draw a graph of tube # vs precipitate mass. For the maximum value on the graph, find the related number of mmoles of lead present. 4. For the maximum value on the graph, find the related number of mmoles of iodide present. _______________________ 5. What is the ratio of lead to iodide implied in #3 & #4. _______________________ 7. From the above ratio, what is the formula of the precipitate? _______________________ _______________________ Formula of a Precipitate Lab (Quick Version) Precipitate: a solid substance formed when ions from soluble solutions are mixed in a way that violates natures solubility rules. Their combinations are specific to the ions present. We will determine the formula. Wear goggles as always for all steps of the lab. (*** TOXIC CHEMICALS ***) PROCEDURE: 1. 2. 3. Clean and label 8 test tubes (1-8) Clean and label(A and B) 2 beakers that will hold 50 ml of chemicals. Obtain 50 ml of A and 50 ml of B. Use measurements on the beakers. These are not accurate but that will be fine for this step. Using 2 separate droppers, count the following drops of each chemical into the 8 test tubes. ------------------------------------------------------------------------------------------------------------------CODE\ Tube 1 2 3 4 5 6 7 8 ------------------------------------------------------------------------------------------------------------------A 80 70 60 50 40 30 20 10 Drops ------------------------------------------------------------------------------------------------------------------B 10 20 30 40 50 60 70 80 Drops ------------------------------------------------------------------------------------------------------------------4. Gently shake each test tube to mix the chemicals and allow to sit undisturbed for 5-10 minutes. 5. Observe for the maximum height of the yellow precipitate in the tubes. 6. The drop ratio in that tube is the same as the MOLE RATIO. Questions: 1. Which tube is the highest? ___________________________________________| 2. For the maximum height, find the related number of moles of A present. ___________________________________________ 3. For the maximum height, find the related number of moles of B present. ___________________________________________ 4. What is the ratio of A:B implied in #2 & #3. (use reduced whole number ratio)_____________________________________ (Take the answer of this question to the instructor and he will provide you with information needed to answer the next question.) 5. From the above ratio and the information from the instructor, what is the formula of the precipitate? ______________________ Dry Ice Lab Name: _______________________________________ Name: _______________________________________ Name: _______________________________________ Section: _____ Introduction: Dry Ice is not ice. It is solidified Carbon Dioxide. The term dry is used because it has no water present. Density(s)= 1.35 g/ml. Density(g)= .001976 g/ml. Dry Ice sublimes at –78.48˚C. It forms Carbonic Acid (H2CO3) in the presence of water which decomposes to produce Carbon Dioxide gas which is released into the water. SAFETY: DO NOT HOLD DRY ICE IN YOU HANDS or allow contact with any other body part. Contact will feel like it is burning. However, it is really causing frost bite on you skin because the water in the cells is freezing instantly and expanding killing the cells of your skin. Use metal tongs, several layers of paper or gloves to protect your skin while touching the Dry Ice. Procedures: 1. Put several small pieces of dry ice into balloon and tie. Let sit for 5 minutes. Record observations. ________________ __________________________________________________________________________________________________ What is happening in the balloon? How big would it eventually become? What is the relationship of the size to the mass of the Dry Ice? ________________________________________________________________________________________ __________________________________________________________________________________________________ 2. Push small flat piece of dry ice along the black top table. Notice the ease needed to push the piece and the length of glide path compared to a similar size and weight of another material. Record observations. ____________________________ ___________________________________________________________________________________________________ Can you predict the reason for the Dry Ice sliding so easily? ___________________________________________________ ___________________________________________________________________________________________________ 3. Touch the dry ice with a metal object and hold there loosely for several seconds. Record observations. _______________ ___________________________________________________________________________________________________ Can you come up with a cause of the metal object’s “Squeal”? ________________________________________________ ___________________________________________________________________________________________________ 4. 5. Half fill a 250 ml beaker with tap water. Touch the water with a torn piece of pH paper and record observations of the color of the paper. __________________________________________________________________________________ Which direction is the Dry Ice Vapor going as it exits the beaker? Why? (Be sure to read the Introduction) ___________________________________________________________________________________________________ Put several small pieces of Dry Ice into the water. After a few minutes of bubbling, you may notice a change in the beaker. Record these observations. ___________________________________________________________________________ Touch the water with another piece of pH paper. Record observations again. ____________________________________ Was there a difference in the pH paper color in water compared to Dry Ice? What could be the cause? (Be sure to read the Introduction)_______________________________________________________________________________________ ___________________________________________________________________________________________________ 6. Have one group member volunteer to put a pea sized piece of Dry Ice into a small cup ½ filled with water. Too big of a piece will not work as well. Allow to sit until all vapors stop rising. Sip the liquid. Describe the taste or feel in your mouth. _____________________________________________________________________________________________ Did the taste of the water/Dry Ice mixture match anything you have tasted before? _________________________________ HEAT OF FUSION OF ICE LAB NAME______________________________________ NAME______________________________________ NAME______________________________________ Section ___________ USE ONLY INFORMATION ON THIS PAGE AND DATA FROM YOUR LAB MEASUREMENTS. PROCEDURE: 1. GET A CALORIMETER AND A THERMOMETER FROM INSTRUCTOR. 2. OBTAIN 100ml OF TAP WATER BETWEEN THE TEMPERATURES OF 35˚C AND 70˚C. 3. POUR INTO BASE OF CALORIMETER AND MEASURE THE TEMPERATURE ACCURATELY. RECORD THE TEMPERATURE:_______________________________________ 4. GRAB A SMALL HANDFUL OF ICE FROM THE COOLER, SHAKE OFF ANY WATER. QUICKLY PUT THE ICE INTO THE BASE OF THE CALORIMETER (INSTRUCTOR MAY ADJUST AMOUNT OF ICE USED). 5. PLACE TOP AND THERMOMETER ON THE BASE AND STIR SLOWLY UNTIL THE MINIMUM TEMPERATURE IS REACHED AND ALL SOLID ICE IS GONE. RECORD THE LOWEST TEMPERATURE:______________________________________ 6. MEASURE AND RECORD THE FINAL VOLUME OF THE CALORIMETER:____________________________ml CALCULATIONS: SHOW ALL WORK 1. CALCULATE THE MASS IN GRAMS OF THE ORIGINAL WATER. 2. CALCULATE THE MASS IN GRAMS OF THE MELTED ICE. 3. CALCULATE THE DELTA T OF THE ORIGINAL WATER BY SUBTRACTING THE MINIMUM TEMPERATURE FROM THE ORIGINAL TEMPERATURE. 4. CALCULATE THE CALORIES RELEASED BY THE ORIGINAL WATER SAMPLE BY MULTIPLYING THE DELTA T (FROM #3) TIMES THE GRAMS OF WATER (FROM #1) TIMES THE SPECIFIC HEAT OF LIQUID WATER (1 CALORIE/GRAM ˚C). 5. CONVERT THE CALORIES IN #4 INTO Kcals. 6. CALCULATE THE NUMBER OF MOLES OF ICE MELTED USING THE ANSWER FROM #2. 7. USING THE ANSWERS FROM #5 & #6, CALCULATE THE Kcals/MOLE OF ICE. *******THE ANSWER OF #7 IS CALLED THE MOLAR HEAT OF FUSION OF ICE******* WARMING AND COOLING OF A PURE SUBSTANCE (PDCB) Name____________________________________ Name____________________________________ Name____________________________________ Section_____ COOLING BEHAVIOR (day 1) 1. Fill a 400 or 600 ml beaker about 3/4 full of warm tap water that is between 30-35'C. The thermometer that is in the water should NOT come in contact with the PDCB. 3. Warm the test tube filled with ParaDiChloroBenzene (PDCB) over an open flame by moving the tube back and forth. As soon as the solid starts to melt, insert a DRY thermometer and start stirring and measuring the temperature until it reaches above 90˚C and all of the solid is melted. BE CAREFUL ABOUT THE TEMPERATURE RISING ABOVE THE LIMIT OF THE THERMOMETER(THEY WILL BREAK!). 4. Clamp the test tube into the beaker of water. Stir the contents of the tube CONSTANTLY. Every 30 seconds, RECORD the temperature of the PDCB and the water on the chart below. TIME (sec) 0 30 60 90 120 150 180 210 240 270 300 330 360 390 420 450 480 510 540 570 COOLING TEMP Day 1 PDCB WATER WARMING TEMP PDCB Day 2 WATER WARMING BEHAVIOR (day 2) 1. Fill a 400 or 600 ml beaker about ¾ full of warmed tap water that is between 65-70'C. Warm the tap water on a HOT PLATE. Caution using the HOT PLATE. Measure the temperature with a thermometer that will NOT be allowed to touch the PDCB. 2. The test tube filled with PDCB should have a thermometer frozen in it. 3. Clamp the test tube into the beaker of water. Stir the PDCB constantly. Every 30 seconds RECORD the temperature of the PDCB and the water in the last two columns on the chart above. 4. Continue stirring until the temperatures are within 5 degrees of each other. 5. Remove the thermometer from the PDCB and clean with paper towel. Turn in the PDCB test tube and the thermometers. Questions 1. DRAW A TIME vs TEMPERATURE GRAPH CONTAINING ALL 4 SETS OF DATA. TITLE, LABELS, 4 COLORS. 2. INTERPRET THE GRAPHS TO DETERMINE THE MELTING AND FREEZING POINTS OF PDCB. 3. WHAT WOULD HAPPEN TO THE LINES ON THE GRAPHS IF MORE PDCB HAD BEEN IN THE TUBE? Lab: Specific Heat of Substance Names _________________________________________________________________________ Section _______________ Procedure: 1. Place metal weight into boiling water for 5 minutes. 2. Measure 100ml of tap water in graduated cylinder and pour into calorimeter. Density of water is 1 g/ml. 3. Record temperature of the water and record. 4. Using tongs, place hot weight into calorimeter. 5. Immediately place top w/ thermometer onto calorimeter. 6. Stir constantly, avoiding the metal, and check temperature every 30 seconds. Do not remove the thermometer when checking. Just pull it out slightly and read. 7. When the thermometer has the same reading for 3 times, record the temperature. __________˚C 8. Find and record the mass of the metal object. Use a weighing boat to prevent rolling. __________ grams Calculations: calories = (grams)(specific heat)(change of temperature) __________˚C (Use this formula for both questions) 1. The specific heat of liquid water is 1 cal/g’C. Use that number to find the number of calories absorbed by the 100 grams of water. 2. The calories absorbed by the water is the energy (calories) released by the metal, Assume the starting temperature of the metal was 100˚C because it was in boiling water and the ending temperature was recorded by you above. Calculate the Specific Heat of the metal. Heat of Reaction Lab Purpose: To prove the energy in Chemical Reactions is mathematically related. Reaction A: Solid Sodium Hydroxide dissolving in water to form aqueous ions. NaOH(s) Na+(aq) + OH-(aq) + x1 kj Reaction B: Solid Sodium Hydroxide dissolves and reacts with aqueous Hydrochloric Acid forming water and an aqueous solution of Salt NaOH(s) + H+(aq) + Cl-(aq) H20 + Na+(aq) + Cl-(aq) + x2 kj Reaction C: Aqueous Sodium Hydroxide react with aqueous Hydrochloric Acid forming water and an aqueous solution of Salt. Na+(aq) + OH-(aq) + H+(aq) + Cl-(aq) H20 + Na+(aq) + Cl-(aq) + x3 kj Procedure: Reaction 1: a. b. c. Measure 100 ml of deionized water into a calorimeter. Mass and record about 2 grams of solid Sodium Hydroxide. [[CAUTION: DANGER TO SKIN]] This chemical absorbs moisture from the air so measurement must be quick and close the top of the bottle and start the next step quickly. Record the starting temperature of the water and add pellets. Close calorimeter and stir with thermometer until all Solid is gone and a high temperature has been reached and record. Dispose with down the drain. Reaction 2: d. Repeat steps a, b and c using 100 ml of .5M Hydrochloric Acid instead of water. Record beginning and final temperature and the mass of the Sodium Hydroxide. . Reaction 3: e. Measure 50 ml of 1.0M Hydrochloric acid and pour into calorimeter and measure 50 ml of 1.0M Sodium Hydroxide solution into a clean beaker. Measure and Record the temperature of both solutions rinsing the thermometer between solutions. Pour beaker into calorimeter, close, stir and Record the highest temperature. Questions: Use this conversion factor: kj = 4.18 kcal 1. Complete the following table from your data. Reaction number Delta T Heat Released Moles of NaOH A. B. C. 2. Using equation B as the net equation, see if A and B can act as sub-equations. 3.Find the calculated Net Delta H for the Net equation using Delta H’s from the other 2 reactions. 4. Find the percent error of the calculated Delta H using your experimental Delta H as being correct. Experimentally derived Energy Released/mole (Delta H) How the answer questions on Heat of Reaction Lab Delta T is the change of temperature they measured yesterday. Question 1: filling out the last 3 columns Heat released: Steps needed 1. Using cal = (g)(SH)(Delta T) grams will be the mass of the liquid using DH2O (1 g/ml) and the specific heat of liquid water (1 cal/g’C) and the Delta T’s from Tuesday. 2. Skip 2. Convert cals to Kcals using DA and definition of Kilo 3. Convert the Kcals to Kj using the conversion factor on the lab page. Moles of NaOH: 1 & 2 Use molar mass and DA from 2 grams to find moles. 3. Use Molarity formula to find moles Energy Released (kj/mole NaOH) also known as Delta H: divide Heat Released(kj) by Moles of NaOH to get kJ/mole. Question 2: Self explanatory, do like we have been doing on the worksheets. Question 3: Also like the worksheets, add the Delta H’s from reactions 1 & 3 to solve for Net Delta H for reaction 2. Question 4: (note absolute value signs) | 1 - ( Net Delta H for reaction 2 from question 3 / Reaction 2 Energy released from chart)| x 100 ORGANIC FAMILIES MODEL BUILDING LAB PERIOD___________ NAMES______________________, _______________________, _______________________ BUILD, DRAW COMPLETE MOLECULAR STRUCTURE, NAME AND WRITE THE FORMULA OF EACH REQUIRED STRUCTURE. BRING ALL WORK TO INSTRUCTOR FOR SIGNATURE. A STRUCTURE THAT FULFILLS ONE REQUIREMENT WILL NOT BE ACCEPTABLE FOR ANOTHER. 1. PROPYL GROUP:________________ BUTYL GROUP:______________ (FORMULA) (FORMULA) 2. 2 DIFFERENT 6 CARBON ALKANE ISOMERS: a. NAME______________________________ ___________ ____ (FORMULA) b. NAME______________________________ ___________ ____ (FORMULA) 3. 2 DIFFERENT 6 CARBON ALKENE ISOMERS: a. NAME______________________________ ___________ ____ (FORMULA) b. NAME______________________________ ___________ ____ (FORMULA) 4. 3 DIFFERENT 6 CARBON ALCOHOL ISOMERS: a. NAME______________________________ ___________ ____ (P) (FORMULA) b. NAME______________________________ ___________ ____ (S) (FORMULA) c. NAME______________________________ ___________ ____ (T) (FORMULA) 5. 2 DIFFERENT 6 CARBON ALDEHYDE ISOMERS: a. NAME______________________________ ___________ ____ (STRAIGHT CHAIN) (FORMULA) b. NAME______________________________ ___________ ____ (BRANCHED CHAIN) (FORMULA) 6. 2 DIFFERENT 7 CARBON KETONE ISOMERS: a. NAME______________________________ ___________ ____ (SYMMETRICAL) (FORMULA) b. NAME______________________________ ___________ ____ (ASYMMETRICAL) (FORMULA) 7. 2 DIFFERENT 6 CARBON ETHER ISOMERS: a. NAME______________________________ ___________ ____ (SYMMETRICAL) (FORMULA) b. NAME______________________________ ___________ ____ (ASYMMETRICAL) (FORMULA) 8. 2 DIFFERENT 6 CARBON ALKYNE ISOMERS: a. NAME______________________________ ___________ ____ (SYMMETRICAL) (FORMULA) b. NAME______________________________ ___________ ____ (ASYMMETRICAL) (FORMULA) 9. 2 DIFFERENT ISOMERS OF C6H12O: a. NAME______________________________ ___________ ____ (NOT A KETONE ) (FORMULA) b. NAME______________________________ ___________ ____ (NOT AN ALDEHYDE) (FORMULA) 10. 2 DIFFERENT ISOMERS OF C6H12: a. NAME______________________________ ___________ ____ (FORMULA) b. NAME______________________________ ___________ ____ (NO DOUBLE BONDS ALLOWED) (FORMULA) RATE OF REACTION LAB – INSTRUCTIONS 2I- + S2O8-2 I2 + I2 + 2SO4-2 (SLOW) 2 S2O3-2 2I- You will be mixing: + S406-2 (FAST) A. .2M Sodium or Potassium Iodide B. .005M Sodium Thiosulfate (4 drops of Starch added each time) C. .1M Ammonium Peroxydisulfate Table 1: Concentration -----------------------------------------------------------------------------Mixture Solution A Solution B Solution C -----------------------------------------------------------------------------1 20 ml 10 20 -----------------------------------------------------------------------------2 15 + 5ml H20 10 20 -----------------------------------------------------------------------------3 10 + 10ml 10 20 -----------------------------------------------------------------------------4 5 + 15ml 10 20 -----------------------------------------------------------------------------5 20 10 15 + 5ml H20 -----------------------------------------------------------------------------6 20 10 10 + 10ml -----------------------------------------------------------------------------7 20 10 5 + 15ml ------------------------------------------------------------------------------ Time (sec.) Table 2: Temperature (Add 4 drops of starch to each B). Use Ice water or hot water baths to reach the desired temperatures. ----------------------------------------------------------------------------------------Mixture Solution A Solution B Solution C Temp C’ Time(sec) ----------------------------------------------------------------------------------------8 20 ml 10 20 5 - 10 ----------------------------------------------------------------------------------------9 20 10 20 10 - 20 ----------------------------------------------------------------------------------------10 20 10 20 20 - 30 ----------------------------------------------------------------------------------------11 20 10 20 30 - 40 ----------------------------------------------------------------------------------------12 20 10 20 40 - 45 ----------------------------------------------------------------------------------------Table 3: Catalyst (Add 4 drops of Copper solution to each B along w/ starch). Use same water baths as above. ----------------------------------------------------------------------------------------Mixture Solution A Solution B Solution C Temp C’ Time(sec) ----------------------------------------------------------------------------------------13 20 ml 10 20 5 - 10 ----------------------------------------------------------------------------------------14 20 10 20 10 - 20 ----------------------------------------------------------------------------------------15 20 10 20 20 - 30 ----------------------------------------------------------------------------------------16 20 10 20 30 - 40 ----------------------------------------------------------------------------------------17 20 10 20 40 - 45 ----------------------------------------------------------------------------------------You will be assigned specific mixtures to prepare and time. You must repeat each within 10%. Average your times per reaction and report to the nearest second. Use glassware marked for specific chemical only. Wash glassware that contacts multiple solutions. Use test tubes in Hot or Cold water baths to obtain desired temperature midpoint. Stir!!! Rate of Reaction Lab – Data Group numbers 1 2 3 4 5 6 -----------------------------------------------------------------REACTION 1 -----------------------------------------------------------------REACTION 2 -----------------------------------------------------------------REACTION 3 -----------------------------------------------------------------REACTION 4 -----------------------------------------------------------------REACTION 5 -----------------------------------------------------------------REACTION 6 -----------------------------------------------------------------REACTION 7 -----------------------------------------------------------------REACTION 8 -----------------------------------------------------------------REACTION 9 -----------------------------------------------------------------REACTION 10 -----------------------------------------------------------------REACTION 11 -----------------------------------------------------------------REACTION 12 -----------------------------------------------------------------REACTION 13 -----------------------------------------------------------------REACTION 14 -----------------------------------------------------------------REACTION 15 -----------------------------------------------------------------REACTION 16 -----------------------------------------------------------------REACTION 17 ------------------------------------------------------------------ 7 8 9 10 Average for all groups RATE OF CHEMICAL REACTIONS LAB EVALUATION 1. DRAW A DATA TABLE SHOWING THE AVERAGE TIME FOR EACH REACTION AS AVERAGED BY YOU FROM THE CLASS DATA. 2. FILL IN THE FOLLOWING TABLE OF CONCENTRATIONS: MIXTURE ml .2M I[I-] ml .1M S2O8-2 [S2O8-2] TIME ____________________________________________________________________________________ 1 ____________________________________________________________________________________ 2 ____________________________________________________________________________________ 3 ____________________________________________________________________________________ 4 ____________________________________________________________________________________ 5 ____________________________________________________________________________________ 6 ____________________________________________________________________________________ 7 ____________________________________________________________________________________ 3. MAKE A GRAPH PLOTTING INITIAL CONCENTRATION OF I- Vs. ELAPSED TIME. FOR MIXTURES 1-4. PUT TIME ON THE HORIZONTAL AXIS. 4. ON THE SAME GRAPH PAPER USING THE SAME SCALES, PLOT THE PEROXYDISULFATE CONCENTRATION Vs. ELAPSED TIME FOR MIXTURES 1,5,6,7. 5. HOW DOES CHANGING THE CONCENTRATION OF REACTANTS AFFECT THE ELAPSED TIME REACTION? 6. HOW MANY MOLES OF S2O8-2 ARE NOT USED UP IF ALL OF THE I- IN MIXTURE 3 IS COMPLETELY CHANGED TO I2? 7. DRAW A TIME Vs TEMPERATURE GRAPH ON A SECOND PAPER OF REACTIONS 8-12. USE THE MIDPOINT OF THE REQUESTED TEMPERATURE RANGES. PLOT TIME ALONG THE HORIZONTAL AXIS. 8 5-10 9 10-20 10 20-30 11 30-40 12 40-45 8. WHAT GENERALIZATIONS CAN BE DEDUCED FROM GRAPH 7? 9. PREPARE ANOTHER GRAPH ON THE SAME SHEET AS 7 OF TIME Vs TEMPERATURE OF 13-17. LABEL THIS CURVE "CATALYZED". 10. WHAT GENERALIZATION CAN BE DEDUCED BY COMPARING THE TWO CURVES OF GRAPH 7 AND GRAPH 9. FOR THE EQUILIBRIUM LAB PURPOSE: TO FIND A MATHEMATICAL APPROACH TO DEAL WITH CHEMICAL EQUILIBRIUM SERIAL DILUTION INSTRUCTIONS: -YOU NEED 5 CLEAN TEST TUBES ALL THE SAME SIZE -AFTER TUBES 1 AND 2 ARE MIXED, ONE PARTNER CAN START VIEWING. -TIME IS VERY IMPORTANT IN THIS LAB, DON'T SIT AND WATCH OTHERS WORK. TUBE 1: 5 ml .2M Fe(NO3)3 + 5 ml .002M KSCN TUBES 2-5 BEAKER A: 10 ml .2M Fe(NO3)3 + 15 ml H2O TAKE 5 ml FROM BEAKER A, AND PUT INTO TUBE 2 ADD 5 ml .002M KSCN TO TUBE 2 BEAKER B: TAKE 10 ml FROM BEAKER A + 15 ml H2O TAKE 5 ml FROM BEAKER B, AND PUT INTO TUBE 3 ADD 5 ml .002M KSCN TO TUBE 3 BEAKER C: TAKE 10 ml FROM BEAKER B + 15 ml H2O TAKE 5 ml FROM BEAKER C, AND PUT INTO TUBE 4 ADD 5 ml .002M KSCN TO TUBE 4 BEAKER D: TAKE 10 ml FROM BEAKER C + 15 ml H2O TAKE 5 ml FROM BEAKER D AND PUT INTO TUBE 5 ADD 5 ml .002M KSCN TO TUBE 5 -------------------------------------------------------------------------VIEWING FILL OUT THE FOLLOWING CHART OF HEIGHTS OF TEST TUBES AFTER COMPARING THE COLORS AFTER FOLLOWING THE SERIAL DILUTION INSTRUCTIONS IN PREPARING THE FIVE TUBES. TUBE 1 IS THE ONLY TUBE THAT HAS SOLUTION POURED IN OR OUT. POUR 1/2 OF TUBE 1 INTO CATCH CONTAINER. COMPARE COLORS OF TWO TUBES. USE DROPPER TO ADD SOLUTION 1 BACK UNTIL COLORS MATCH. MEASURE HEIGHTS OF BOTH TUBE AND RECORD IN THE FOLLOWING CHART. HEIGHT OF TUBES WHEN COLORS MATCH ALL MEASURED IN MILLIMETERS ----------------------------------------------------------------HEIGHT OF HEIGHT OF TUBE 1 (mm) OTHER TUBE (mm) --------------------------------------------------------------| | 2 | | --------------------------------------------------------------| | 3 | | --------------------------------------------------------------| | 4 | | --------------------------------------------------------------| | 5 | | --------------------------------------------------------------(HEIGHTS IN THIS COLUMN SHOULD EQUAL) EQUILIBRIUM LAB -- CONCENTRATION CHART Column VIAL # A B INITIAL ION CONCENTRATIONS [Fe+3] [SCN-] 1 C HEIGHT RATIO (1/OTHER) Do not use this cell D E F EQUILIBRIUM ION CONCENTRATIONS [FeSCN+2] [Fe+3] [SCN-] 2 3 4 5 CHEMICAL REACTION: Fe+3 + SCN--> FeSCN+2 FERRIC THIOCYANATE THIOCYANO IRON(III) ION ION COMPLEX ION FOR TUBES 2-5, USE THE EQUILIBRIUM CONCENTRATION DATA FROM THE ABOVE CHART TO COMPLETE THE FOLLOWING TABLE USING THREE DIFFERENT MATH OPERATIONS. WE WILL FIND WHICH MATH OPERATION WILL PROVIDE US WITH THE LEAST VARIABLE ANSWER(CONSTANT). RELATIONSHIP EVALUATION TABLE A B [Fe+3][FeSCN+2] [FeSCN+2] TUBE -----------------------------------[SCN-] [Fe+3][SCN-] 1 Do not use this row C [FeSCN+2] -------------------[Fe+3] + [SCN-] 2 3 4 5 A RATIO OF 2-5 LARGEST 2-5 SMALLEST Answers: _________________ _________________ B __________________ __________________ C _________________ _________________ Le Chatelier’s Principle Lab Introduction The purpose of this lab is to develop a greater understanding of le Chatelier’s principle. In this lab you will observe the way that changing the concentration of products, the concentration of reactants, and temperature affect the equilibrium position of a reaction. You will be using the following reaction of the cobalt ion, which has a pink product and a blue reactant. The beginning solution will be at equilibrium with half product and half reactant, resulting in a purple solution. Co(H2O)62+ (aq) + 4 Cl- (aq) CoCl42- (aq) + 6 H2O (in solution, not pure) WARNING!!!!! CONCENTRATED HCl IS EXTREMELY DANGEROUS!!!!!! Procedure 1. Use a 250-mL beaker to make a hot water bath (use about 100 mL water). Do not let the water boil, but get it hot. 2. Obtain 2 well plates and place them side by side so you have six columns and four rows of wells. Number the rows A-D and the Columns 1-6 as shown at the end of the procedure. 3. Obtain four small beakers and 4 droppers and obtain a small sample of cobalt solution (47-g CoCl2 in 1000-mL solution), silver solution (8.5-g AgNO3 in 500 mL solution) , deionized water, and concentrated hydrochloric acid. Label all beakers and droppers! 4. Place five drops of the cobalt solution in all 24 wells on the plates. 5. Follow the chart below and add the appropriate number of drops in each well of the correct solution in the correct order. Do one well at a time and stir with a toothpick after adding the drops. Record the color of the solution after stirring in the data table. 1 2 HCl 3 HCl 2 HCl then 5 H2O 2 HCl then 5 Ag A B C D 2 4 HCl 5 HCl 4 HCl then 5 H2O 4 HCl then 5 Ag 3 6 HCl 7 HCl 6 HCl then 5 H2O 6 HCl then 5 Ag 4 8 HCl 9 HCl 8 HCl then 5 H2O 8 HCl then 5 Ag 5 10 HCl 11 HCl 10 HCl then 5 H2O 10 HCl then 5 Ag 6 12 HCl 13 HCl 12 HCl then 5 H2O 12 HCl then 5 Ag 6. Obtain a test tube and fill it about ¼ of the way with purple solution. Place the test tube in the hot water bath. Record the color change in the data table. 7. Place the test tube in an ice bath and record the color change in the data table. 8. All chemicals may be dumped down the drain with plenty of water. Data 1. Fill in the final color of each well in the data table below. 1 2 3 4 5 A B C D 2. What was the color of the solution after it was placed in the hot water bath? 3. What was the color of the solution after it was placed in the ice bath? 6 Discussion 1. When HCl is added to the solution, what ions are being added? Which of these two ions is important to the equilibrium position? 2. Which direction should the reaction shift when HCl is added? 3. When H2O is added to the solution, which direction should the reaction shift? 4. When silver nitrate is added to the solution what happens to the concentration of chloride ions in the solution? How do you know this? 5. Which direction should the reaction shift when silver nitrate is added? 6. Did all of your data agree with the answers to numbers 1-5? 7. Which direction did the reaction shift when it was placed in hot water? What does this tell you about the reaction? 8. Which direction would the reaction shift if sodium chloride were added? ANALYTICAL LAB SILVER, LEAD AND MERCURY THE PURPOSE OF THIS LAB IS TO DETERMINE THE PRESENCE OF THESE HEAVY METALS. PARTNER 1:___________________ PARTNER 2:___________________ PARTNER 3:___________________ PERIOD #:__________________ I. TEST FOR LEAD A. ADD 1 ml OF LEAD NITRATE TO T.T. B. ADD 5 DROPS OF HCl TO T.T. C. THE WHITE PPT. IS AN INDICATOR THAT LEAD IONS MIGHT BE PRESENT. D. DECANT AWAY MOST OF THE WATER SO THAT THE PPT REMAINS IN THE T.T. E. ADD 2 ml DEIONIZED H2O. F. HEAT T.T. IN A BOILING WATER BATH FOR 1 MIN. E. DECANT AWAY MOST OF THE LIQUID INTO A CLEAN T.T. F. TO THE LIQUID ADD 5 DROPS OF K2CrO4. G. IF THE A YELLOW PPT APPEARS, THEN LEAD WAS PRESENT. II. TEST OF MERCURY A. ADD 1 ml OF MERCURY NITRATE TO T.T. B. ADD 5 DROPS OF HCl TO T.T. C. THE WHITE PPT. IS AN INDICATOR THAT MERCURY IONS MIGHT BE PRESENT. D. DECANT AWAY MOST OF THE WATER SO THAT THE PPT REMAINS IN THE T.T. F. ADD 3 ml OF 6M NH4OH. G. IF THE A BLACK PPT APPEARS, THEN MERCURY WAS PRESENT. III. TEST FOR SILVER A. ADD 1 ml OF SILVER NITRATE TO T.T. B. ADD 5 DROPS OF HCl TO T.T. C. THE WHITE PPT. IS AN INDICATOR THAT SILVER IONS MIGHT BE PRESENT. D. DECANT AWAY MOST OF THE WATER SO THAT THE PPT REMAINS IN THE T.T. E. ADD 2 ml DEIONIZED H2O. F. ADD 2 ml OF NH4OH TO THE T.T. G. IF THE PPT DISAPPEARS, THEN SILVER MIGHT BE PRESENT. H. ADD 3 ml OF 6 M HNO3 TO THE SOLUTION. I. IF CLOUDY OR WHITE PPT RETURNS, THEN SILVER IONS WERE PRESENT. IV. DETERMINATION OF AN UNKNOWN EACH GROUP WILL TAKE A CLEAN T.T. TO THE INSTRUCTOR TO RECEIVE A SOLUTION WHICH WILL CONTAIN A MIXTURE OF THESE IONS. IT COULD CONTAIN ANY MULTIPLE COMBINATION OF THE THREE IONS. IT WILL BE YOUR JOB TO FOLLOW THE FLOW CHART PROVIDED TO MAKE YOUR ANALYTICAL DECISION ABOUT THE CONTENTS OF THE UNKNOWN. WHEN YOU HAVE MADE A DECISION ABOUT THE CONTENTS, FILL IN THE FOLLOWING CHART AND SHOW TO YOUR INSTRUCTOR. IF ANY PART OF THE CHART IS INCORRECT, THE CHART WILL BE RETURNED TO YOU FOR CORRECTION WITH A TEN POINT REDUCTION. UNKNOWN #________ CONTAINS: LEAD_______________ MERCURY__________ SILVER_____________ NEGATIVE ION TESTING LAB -- Unknown YOUR UNKNOWN WILL CONTAIN SEVERAL OF THE FOLLOWING IONS. YOU WILL HAVE TO TEST FOR EACH TO DECIDE WHICH OF THE IONS ARE PRESENT. BRING YOUR LIST OF POSITIVE TESTS TO THE COMPUTER AND PUT THE DATA INTO THE COMPUTER AND SEE IF YOU ARE CORRECT. TEST 1: HYDROGEN IONS (H+) AND HYDROXIDE IONS (OH-) A. PUT 1 ML OF UNKNOWN INTO CLEAN TEST TUBE. B. DIP CLEAN STIRRING ROD INTO SOLUTION. C. TOUCH THE TIP OF THE ROD TO A SMALL SQUARE OF LITMUS PAPER. D. IF PAPER TURNS:PINK----------POSITIVE TEST FOR ACID. BLUE----------POSITIVE TEST FOR BASE. NO CHANGE WITH EITHER RED OR BLUE THEN BOTH NEGATIVE. TEST 2: BROMIDE ION (Br-) AND IODIDE ION (I-) A. PUT 1 ML OF UNKNOWN INTO TEST TUBE. B. ADD 5 DROPS OF CHLORINE WATER (******* CLOSE TOP QUICKLY ******) C. ADD 1 ML OF Hexane ;SHAKE VIGOROUSLY; OBSERVE THE COLOR OF THE BOTTOM IF THE TOP IS: PURPLE TO PINK----------POSITIVE IODINE RUST TO GOLD------------POSITIVE BROMINE OTHER COLORS------------NEGATIVE (****** DISCARD Hexane IN THE PROPER WASTE CONTAINER ******) TEST 3: CHLORIDE IONS (Cl -) A. PUT 1 ML OF UNKNOWN INTO TEST TUBE. B. ADD 5 DROPS OF .1 M AgNO3 SOLUTION. C. IF NO PPT. OCCURS THEN CHLORIDE IS NEGATIVE-----GO ON TO TEST 4. D. IF WHITE PPT. DOES OCCUR, THEN CHLORIDE IS POSSIBLE. PROCEED BY ADDING 3 ML OF 6 M AMMONIUM HYDROXIDE. IF ANY PPT. REMAINS, ALLOW SETTLING TO OCCUR, AND POUR OUT 1 ML OF LIQUID(ONLY) INTO ANOTHER TEST TUBE. TO THE SECOND TEST TUBE, ADD 6 M NITRIC ACID UNTIL ACIDIC (TEST WITH LITMUS). WHEN ACIDIC, A WHITE PPT. IS A POSITIVE TEST FOR CHLORIDE (OFTEN ONLY SHOWS AS INCREASED CLOUDINESS). TEST 4: SULFIDE ION (S -2) A. PUT 1 ML OF UNKNOWN INTO TEST TUBE. B. DIP A STRIP OF FILTER PAPER INTO SAT. LEAD ACETATE SOLN. C. ADD ENOUGH 2M HYDROCHLORIC ACID TO TEST FOR ACIDITY. (DO NEXT STEP VERY QUICKLY) QUICKLY INSERT THE STRIP INTO THE MOUTH OF THE TEST TUBE SIDE OF THE TUBE). D. IF STRIP TURNS DARK----------------POSITIVE SULFIDE. (DON'T TOUCH THE PAGE TWO TEST V. A. B. C. D. E. SULFATE ION (SO 4-2) PUT 1 ml OF UNKNOWN INTO TEST TUBE. ADD 5 DROPS OF .1 M BARIUM NITRATE IF NO PPT FORMS THEN SULFATE IS NEGATIVE(GO TO TEST VI) ADD 3 ml OF 2 M HCl TO THE TEST TUBE. IF PPT. REMAINS-----------------------SULFATE = POSITIVE TEST VI. SULFITE ION (SO3-2) A. PUT 1 ml OF UNKNOWN INTO THE TEST TUBE. B. ACIDIFY WITH .2 M HCL. (USE TEST PAPER) C. PUT 1 ml OF POTASSIUM PERMANGANATE SOLUTION INTO SECOND TESTTUBE. D. CONNECT TWO TUBES WITH GLASS U-TUBE WITH GLASS DOWN INTO THE PERMANGANATE SOLUTION. E. HEAT THE UNKNOWN TUBE OVER BURNER SLOWLY BUT CONSISTENTLY UNTIL BUBBLES BEGIN TO MOVE INTO SECOND TUBE FOR THIRTY SECONDS. F. IF THE POTASSIUM PERMANGANATE IS DISCOLORED THEN -- SULFITE = POSITIVE TEST VII. NITRATE ION (NO 3-) A. PUT 1 ml OF THE UNKNOWN INTO THE TEST TUBE B. ADD 2 ml OF FERROUS SULFATE (FeSO4) TO THE TEST TUBE. ****** PROCEDURE THAT FOLLOWS IS DANGEROUS--CAUTION ******* C. TILT TEST TUBE 45 DEGREES AND SLOWLY ADD 20 DROPS OF 18 M SULFURIC ACID. WATCH FOR VIBRATIONS, LIGHT, NOISE OR EXCESS HEAT COMING FROM TUBE. D. IF A BROWN RING APPEARS BETWEEN THE TWO LAYERS ---NITRATE = POSITIVE TEST VIII. A. B. C. D. E. PHOSPHATE (PO 4-3) PUT 1 ml OF UNKNOWN INTO THE TEST TUBE. ADD 2 M NITRIC ACID UNTIL ACIDIC WHEN TESTED. ADD 1 OR 2 SMALL CRYSTALS OF AMMONIUM MOLYBDATE. WAIT 5 MINUTES (YELLOW COLOR MIGHT OCCUR SOONER). IF YELLOW FORMS AROUND CRYSTALS---PHOSPHATE IS POSITIVE LIMITING FACTOR LAB PRECIPITATE FORMATION NAME__________________________ NAME__________________________ NAME__________________________ PERIOD____ PURPOSE: TO VERIFY THE QUANTITATIVE NATURE OF PRECIPITATE FORMATION BETWEEN TWO INSOLUBLE IONS SAFETY PRECAUTIONS: WILL VARY DEPENDING ON THE CHEMICALS USED. PROCEDURE: (ALL QUANTITATIVE MEASURMENTS REQUIRE EXTREME ACCURACY) 1. MEASURE OUT 100 DROPS OF CHEMICAL "A" INTO A SMALL CLEAN TEST TUBE. 2. COUNT THE NUMBER OF DROPS OF CHEMICAL "B" THAT WILL CAUSE PRECIPITATION TO OCCUR BEFORE PRECIPITATION STOPS. NEAR THE END OF THE DROPPING, YOU MAY HAVE TO WAIT SEVERAL MINUTES BETWEEN DROPS TO CHECK FOR ADDITIONAL PRECIPITATE FORMATION. DROPS________ 3. COUNT THE NUMBER OF DROPS OF TAP WATER NEEDED TO SUPPLY THE VOLUME OF 5 ml. IN A GRADUATED CYLINDER. DROPS________ 4. REPEAT STEP 3 FOR ACCURACY UNTIL THE RESULTS ARE WITHIN 5 DROPS. 5. FROM THE RESULTS OF STEP 4, DETERMINE THE DROPS/ml. DROPS/ml__________ 6. USING THE ANSWER OF STEP 5, DETERMINE THE NUMBER OF ml USED OF CHEMICAL "A" AND OF CHEMICAL "B". ml "A"=___________ ml "B"=___________ 7. USING THE ORIGINAL CONCENTRATION LISTED ON EACH STOCK BOTTLE, CALCULATE THE NUMBER OF MOLES OF "A" AND "B" USED. moles "A"=________ moles "B"=_________ 8. SOLUBILITY CHART: CHECK THE CHART TO DETERMINE WHICH OF THE AVAILABLE IONS WILL PRECIPITATE. WRITE THE CHEMICAL REACTION FOR THE FORMATION OF THE PRECIPITATE. 9. CHECK THE RATIO OF REACTION OF THE IONS IN THE FORMATION EQUATION. MOLES OF "A"/MOLES OF "B" _________/_________ 10. CHECK THE RATIO OF MOLES USED IN YOUR PRECIPITATE FORMATION FROM QUESTION 7. MOLES OF "A"/MOLES OF "B" _________/_________ 11. HOW DO THE ANSWERS OF QUESTIONS 9 AND 10 COMPARE. Colloid Lab Period________ GAK Making Name__________________________________ Name__________________________________ Name__________________________________ Purpose: To prepare and observe the properties of a Colloid Procedure: 1. Use master cup to mark your cup. 2. Measure 40 ml of water in a beaker and pour into cup. 3. Measure glue into the cup up to the pre-measured line. Add color to suit. Stir with rod. 4. Measure 60 ml of water into same beaker, add 7 ml of Borax and mix until dissolved. 5. Combine all into cup and stir. Eventually you can thoroughly mix with hands. Data and Observations: Property Observations Interpretation ----------------------------------------------------------------------------------------1. Ability to flow --------------------------------------------------------------------------------------- -2. Shape change ---------------------------------------------------------------------------------------3. Volume change ---------------------------------------------------------------------------------------Analyze: 1. Is the new material more like a Liquid, Solid or a Gas and give supporting evidence? __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ 2. What other materials have you seen that have similar properties? __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ 3. Considering the flowablity, how would you rate strength of attraction among particles? __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ Conclude: 1. How can the Kinetic Theory be used to explain the closeness of the particles? __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ 2. How can the properties be used to classify this product? __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ TITRATION LAB Names: ___________________________________________________________________________________ Per: ____ DAY 1: PRACTICE DAY (Stock acid and Stock base) 1. Prepare titration setup with buret, stand and flasks. 2. Fill buret with stock base solution to the zero mark. Record the concentration of base. 3. Measure 25ml of stock acid and pour into a clean Erlenmeyer flask. 4. Add 4 drops of phenolphthalein to the acid flask. (DO THIS EVERY TIME YOU PREPARE A NEW TITRATION) 5. Add base from buret into the acid flask until pink color begins to form. Swirl until pink color goes away. Add more base and swirl going with smaller additions as you approach the End Point. 6. Continue adding and swirling until the last drop of base causes a pink color that will not go away with swirling. 7. Record the volume of base solution that was used. 8. Calculate the Molarity of the acid (SHOW WORK ON BACK) 9. Repeat steps 1-8 several times for accuracy. DAY 2: UNKNOWN ACID TITRATION Titrations are most accurate if done in 3 step increments: 1. Ball Park Run: Unknown acid + Stock base to find approximate acid concentration. A base of the same concentration as the acid is then prepared. (Step 5) 2. Approximation Run: Using Unknown acid and your new, more accurate base, now in the buret. 3. Pin Point Run: Using Unknown and your base to find the [ACID] accurate to one drop of base. Lab Procedure to determine the [Acid] of the Unknown Acid: Ball Park Run 1. Give a marked, clean 125ml Erlenmeyer flask to the instructor. 2. Obtain and record the Unknown Acid number at the bottom of this page. 3. Set up buret for titration and repeat steps using Unknown Acid and stock Base. 4. Calculate the [ACID] using your data. (SHOW WORK HERE) 5. Calculate the grams of Sodium Hydroxide needed to make 250 ml of a Sodium Hydroxide solution with the same concentration as your Unknown acid. (SHOW WORK HERE) Approximation Run 6. Make 250ml of a new base solution using the calculated data above. CAUTION: DO NOT ALLOW PELLETS TO COME IN CONTACT WITH YOUR SKIN. REPLACE THE TOP ON THE CONTAINER IMMEDIATELY BECAUSE IT ABSORBS WATER FROM THE ATMOSPHERE. 7. Purge buret with 10 ml of your new base solution to flush out old base. Refill the buret with new base and use to titrate your Unknown acid. 9. Calculate the [ACID]. (SHOW WORK in the space below). 10. It is recommended to repeat the titration for a third time (Pin Point Run) using your new base solution to reach the 1 drop accuracy for your last run. Titration Lab Key 200753707550104 753570119259010 930740220609030 802012139808500 012202240100128 901026080501008 102006179758025 050854505950354 Unknown Acid Number: ____________________________ [Unknown Acid]: _________________________________ Titration Lab Day 1 Practice Instructions 1. 2. 3. 4. Fill the buret with the .5M NaOH stock solution. Fill above the “0” line and drain out the excess to rinse the buret. Catch and discard the waste base. Very accurately measure out 25ml of the stock acid( BE VERY CAREFUL WITH HYDROCHLORIC ACID, IT BURNS SKIN BADLY AND CAUSES BLINDNESS) that is supplied using a 25ml graduated cylinder. Pour into a 125ml or larger Erlenmeyer flask. Put 4 drops of Phenolphthalein into the acid flask. Begin titration by delivering base quickly. Periodically stop the titration and swirl the pink color away. As the pink color increases begin to reduce the amount added before the next swirling. When the pink color develops and remains for 30 seconds while swirling, stop and calculate the [ACID]. 5. Calculate the grams of Sodium Hydroxide solid needed to make a 250ml solution of base that is the same concentration as the acid in the previous problem. BE VERY CAREFUL WITH NaOH. IT BURNS SKIN BADLY AND CAUSES BLINDNESS. Show these calculations to the instructor. 6. (If time is short skip this step)Repeat steps 1-5 for practice and trying increase accuracy, speed and competency. 7. Have one partner, without the knowledge of the other group members, create an UNKNOWN [ACID] by diluting some of the .5M HCl stock acid with distilled water using the formula: M1•V1=M2•V2 where M2 is the 1M stock acid and M2 is the new UNKNOWN [ACID]. Repeat steps 1-5 using this UNKNOWN as the acid in step 2 and see if the other partners can determine accurately the UNKNOWN [ACID]. Next lab day you will actually determining the concentration of an acid whose concentration is unknown to you. GOOD LUCK!!!! Microscale Acid-Base Titration : This process is used to find the concentration of a hydrochloric acid solution by acid-base titration Introduction: In a neutralization process of an acid by a base (or a base by an acid), the hydronium ion donates protons to the hydroxide ions to form water according to the equation: H3O+ + OH- <--> 2H2O. A neutralization process provides a means of determining chemically equivalent volumes of acids or bases. In a neutralization reactions, moles of H3O+ ions equal moles of OHions. This relationship is the basis of a procedure called titration. In a titration, you will add drops of a standard (0.200M NaOH) to a quantity of acid. Phenolphthalein, an indicator, is added to the acid at the beginning. The indicator is clear in the acid, but will change color when the pH of the solution reaches 8. (Remember the pH of the acid will be less than 7 at the start). The pH of the solution will increase as the NaOH is added. When the indicator changes color, you have reached the end point of the titration. This means to stop adding the NaOH base. At this point the moles of acid are equivalent to the moles of base (equivalence point). Using the equation: MaVa = MbVb, you can determine the molarity of the acid used. Procedure: 1. Place 10 drops of vinegar into three wells in a well plate. 2. Add 1 drop of phenolphthalein to each of the wells containing vinegar. 3. Titrate your sample of vinegar (in well #1) by adding a 0.200M solution of NaOH dropwise, counting drops, until the end point is reached (color of the solution changes from clear to pink). Record the number of drops of sodium hydroxide used. 4. Repeat step 3 with wells #2 and #3. Data: Measurements Trial One Trial Two Trial Three Average 10 drops 10 drops 10 drops 10 drops Concentration of NaOH Volume acid (vinegar) Volume base Analysis: Now use the equation MaVa = MbVb to determine the concentration of the acid. (The unit for volume is drops) Eggshell Titration Lab Name: __________________________________ Period: ________ Procedure: 1. Place a crumbled eggshell into a marked beaker and into the oven to dry until the next day. 2. Take the eggshell out of the oven and cool to room temperature. 3. Place the eggshell in a mortar and grind it into a powder with the pestle. 4. Mass out about 0.2 g of the eggshell and place into a clean 250 mL beaker. 5. Pipet 50 mL of HCl (1.0 M) and add this to the beaker. 6. Stir or swirl for 5 minutes 7. Add 3 drops of Phenolphthalein and titrate this with 1.0 M NaOH. Record the results of the titration. Data: Molarity of NaOH Mass of eggshell used Amount of 1.0 M HCl used Reading on buret of NaOH (initial) Reading on buret of NaOH (final) Volume of NaOH used 0 M g mL mL mL mL Calculations: 1. Use your Types of Chemical Reactions Chart to determine what happens with CaCO3 reacts with Hydrochloric Acid. Write and balance the entire equation. 2. Calculate the original number of moles of HCl added to the eggshell (use molarity formula – HCl is 1.0M) 3. Calculate the number of moles of HCl left over after it reacts with the calcium carbonate in the eggshell. (number of moles of HCl left over is equal to number of moles of NaOH used in titration) 4. Calculate the number of moles of HCl consumed in the reaction with calcium carbonate in eggshell (subtract the number of moles of base used in titration from the number of moles of acid you had originally) 5. Calculate the number of grams of calcium carbonate contained in the eggshell (use stoichiometry, starting with the number of moles of HCl used up before the titration and the mole ratio from the balanced equation) 6. Calculate the percent of calcium carbonate in the eggshell (mass of calcium carbonate divided by the mass of eggshell used times 100) pH Titration Lab Name______________________________ Name______________________________ Name_____________________________ period_______________ Purpose:____________________________________________________________ Safety Precautions:_________________________________________________________ _____________________________________________________________________ _____________________________________________________________________ _____________________________________________________________________ _____________________________________________________________________ _____________________________________________________________________ 1. Put 25 ml of Hydrochloric Acid of Unknown concentration into a 250ml beaker. 2. Fill buret with stock base. 3. While pH meter is inserted into the yellow pH7 buffer solution use the left screw to adjust the reading to close to 7. 4. Remove the pH meter from the buffer and rinse tip with deionized water. Insert the pH meter into the acid and read and record the pH 5. Add .5 ml of base to the acid and stir with stirring rod. Read and record the pH as the .5 ml reading. 6. Repeat step 5 for every .5 ml until the pH is between 11 & 12. 7. Graph pH(horizontal) vs. ml of base. 8. You will notice a very distinct motion on the graph. 9. Each group is to interpret the signifigance of that motion and use it to determine the acid concentration. reading as the 0 reading. OXIDATION-REDUCTION LAB NAME__________________________ NAME__________________________ NAME__________________________ PERIOD________________________ PURPOSE:___________________________________________________________________ ___________________________________________________________________________ ___________________________________________________________________________ SAFETY:____________________________________________________________________ ___________________________________________________________________________ ___________________________________________________________________________ ___________________________________________________________________________ PART I: METAL-METAL ION PAIRS A. GET 6 CLEAN TEST TUBES AND LABEL I-VI. B. USING THE FOLLOWING CHART, PLACE 1 ml OF CORRECT IONIC SOLUTION AND SMALL PIECE OF CORRECT METAL INTO EACH TUBE. METAL SAMPLE MAY NEED CLEANING w/ STEEL WOOL TO CLEAN BEFORE USE. PLEASE NOT LOCATIONS OF ROMAN NUMERALS AND USE PROPER COMBINATIONS IN PROPER TUBES. METAL\SOLUTION Cu+2 Pb+2 Zn+2 ------------------------------------------------Cu | N.R. |III |V | | | _________________________________________________ Pb |I | N.R. |VI | | | _________________________________________________ Zn |II |IV | N.R. | | | _________________________________________________ C. AFTER 5 MIN., RECORD OBSERVATIONS IN THE BLANKS ABOVE INDICATING WHICH METAL/METAL ION PAIRS REACTED. USE N.R. FOR NO REACTION. D. DISCARD SOLUTIONS INTO WASTE SOLUTION JUG. CLEAN AND DRY METALS AND RETURN THEM TO THE PROPER METAL CONTAINING CONTAINER. RINSE OUT THE 6 TEST TUBES. PART II--HALOGEN/HALIDE ION PAIRS A. IN EACH OF THE 6 NUMBERED TUBES PUT THE FOLLOWING COMBINATIONS: RECORD COLORS OF Hexane (top) LAYER I. 1 ml Cl- + 1 ml Hexane_______________________ II. 1 ml Br- + 1 ml Hexane_______________________ III. 1 ml I- + 1 ml Hexane_______________________ IV. 1 ml Cl2 + 1 ml Hexane_______________________ V. 1 ml Br2 + 1 ml Hexane_______________________ VI. 1 ml I2 + 1 ml Hexane_______________________ B. DISCARD ALL OF THESE SOLUTIONS INTO TTE WASTE CONTAINER AND RINSE OUT ALL TEST TUBES WITH DEIONIZED WATER. C. IN EACH OF THE 6 NUMBERED TUBES PUT: 2 ml OF HALIDE ION SOLUTION 10 DROPS OF HALOGEN WATER 1 ml OF Hexane HALOGEN\HALIDE ClBrI------------------------------------------------Cl2(aq) | N.R. | III |V | | | _________________________________________________ Br2(aq) |I | N.R. | VI | | | _________________________________________________ I2(aq) | II | 1V | N.R. | | | _________________________________________________ D. OBSERVE THE COLOR OF THE TTE LAYER IN EACH TEST TUBE. COMPARE THE COLOR PRESENT WITH THE EXPECTED COLOR OF THE HALOGEN THAT IS PRESENT. IF THE COLOR IS DIFFERENT THAN EXPECTED, THAT MEANS THAT A REACTION HAS OCCURED CHANGING THE INSERTED CHEMICAL INTO ANOTHER CHEMICAL THAT WAS NOT PRESENT UNTIL IT WAS THE PRODUCT OF THE REACTION. RECORD COLOR AND DECIDE IF INDICATES A REACTION OR NOT. I_________________________________________________________ II________________________________________________________ III_______________________________________________________ IV________________________________________________________ V_________________________________________________________ VI________________________________________________________ THAT OXIDATION-REDUCTION LAB NAME__________________________ NAME__________________________ NAME__________________________ PERIOD________________________ QUESTIONS: ANALYZING THE DATA 1. RANK THE METAL IONS ABILITY TO BE REDUCED BY DETERMINING THE FOLLOWING, WHICH METAL ION REACTED: TWICE_______ ONCE________ NONE_______ 2. USING THIS GENERIC EQUATION: X+2 + 2e- --> X , WRITE EACH OF THE METAL IONS BEING REDUCED TO METAL ATOM. MOST ACTIVE:_______________________________________________ (strongest) ACTIVE:_______________________________________________ LEAST ACTIVE:_______________________________________________ 3. RANK THE HALOGENS ABILITY TO BE REDUCED BY DETERMINING THE FOLLOWING, WHICH HALOGEN REACTED: TWICE_______ ONCE________ NONE_______ 4. USING THIS GENERIC EQUATION: X2 + 2e- --> 2X- , WRITE EACH OF THE METAL IONS BEING REDUCED TO METAL ATOM. MOST ACTIVE:_______________________________________________ (strongest) ACTIVE:_______________________________________________ LEAST ACTIVE:_______________________________________________ 5. YOU WILL REMEMBER THAT IN THE SILVER NITRATE LAB, THE Ag+ ION CAUSED Cu METAL TO DISSOLVE AND FORM BLUE Cu+2 IONS WHILE THE SILVER IONS PRODUCED Ag METAL CRYSTALS. a. WHICH OF THE REACTANTS IS REDUCING (Ag+ or Cu):_________________ b. WRITE THE REDUCING EQUATION FOR a.______________________________ (careful with e-'s) c. WHERE WOULD THIS REDUCER RANK WITH ITS GROUP OF METAL ION/METALS? _____________________________________________________________ OXIDATION-REDUCTION LAB NAME__________________________ NAME__________________________ NAME__________________________ PERIOD________________________ 6. OXIDIZING AGENTS ALLOW OTHER CHEMICALS TO OXIDIZE BY REDUCING THEMSELVES. REDUCING AGENTS ALLOW OTHER CHEMICALS TO REDUCE BY OXIDIZING THEMSELVES. USING THE FOLLOWING CLUES, WE ARE NOW GOING TO COMBINE THE LIST OF 4 METAL ION/METAL REDUCERS WITH THE LIST OF THREE HALOGEN/HALIDE REDUCERS. a. Ag+ IS A STRONGER OXIDIZING AGENT THAN I2, BUT WEAKER THAN Br2. b. I- IS A WEAKER REDUCING AGENT THAN IS Cu, BUT IS STRONGER THAN Ag. strongest reducer:________________________________________________ R A N K I N G :________________________________________________ :________________________________________________ :________________________________________________ :________________________________________________ :________________________________________________ weakest reducer :________________________________________________ 7. USING INFORMATION FROM QUESTION 6, DETERMINE WHICH OF THE FOLLOWING ARE USEFUL OR COUNTER PRODUCTIVE AND WHY: a. COPPER SULFATE IS TO BE STORED IN A ZINC CONTAINER __________________________________________________________ b. COPPER SULFATE IS TO BE STORED IN A SILVER CONTAINER __________________________________________________________ c. A SILVER PLATED SERVING DISH FALLS INTO A LEAD SULFIDE SOLUTION. __________________________________________________________ d. A SILVER RING IS SPLASHED BY SOME BROMINE(aq). __________________________________________________________ Re-dox Lab Preparation Purpose of this lab is to rank chemicals in their ability to reduce. These are the needs for a class of 28 students working in pairs. Part I: Copper, Zinc and Lead strips — 28(2”x ½ “)each 300 ml each of .1 Molar Cu(NO3)2, Zn(NO3)2, Pb(NO3)2 Don’t mix a metal with the same ion. They are automatically No Reactions. Part II: 100ml of Hexane 100ml of .1 Molar NaCl, NaBr, NaI 100ml of water saturated solutions of Cl2, Br2, I2 Disposal of copper and lead solutions and TTE should be done with proper procedures.