Avogadro and Gay-Lussac’s Laws
Kyle Baseden
Chemistry I
Gas Laws/ Avogadro’s and Gay-Lussac’s Law
2/20/15
CCSS.MATH.CONTENT.HSA.SSE.A.1.A-Interpret parts of an expression, such as terms, factors,
and coefficients.
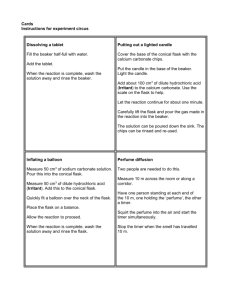
1. Materials
1 250 mL Erlenmeyer flask
about 10 mL of water
balloon
hot plate
2. Objective(s)
calculate the change in the number of moles or volume of a substance when one of the
factors stay the same.
Calculate the change in the pressure or temperature of a gas when one of the factors
remains constant throughout the change.
Differentiate between these laws and Boyle’s and Charles’ Laws
o Recognize when to apply which law to a situation.
Be able to convert between grams and moles
Convert between pressure units successfully
3. Motivation
Engagement
o Balloon in a flask
At the beginning of the lesson, students will see a balloon that is sucked
into a flask.
To get the balloon into the flask
o Take one 250 mL Erlenmeyer flask and fill with a small
(about 10 mL) amount of water
o Bring the water to a boil on a hot plate
o Quickly cover the mouth of the flask with a balloon while
the water is boiling.
o Remove the flask from the hot plate and allow to cool
o The balloon should be sucked into the flask
If this does not happen, the balloon may need to be
guided into the flask.
The instructor should ask the students if they have any idea how this could
happen using any knowledge they have.
The reason this does happen is because as the temperature of the
gas decreases the volume the gas occupies decreases, so the
pressure from the atmosphere pushes on the balloon forcing it into
the flask.
4. Goal for Learner
Students should be able to recognize the difference between these two laws and the gas
laws taught in the previous lesson while also being able to accurately apply Avogadro’s
and Gay-Lussac’s laws in problems.
Revised 4/11
5. Content and Procedures
The instructor will begin the lesson by having a short discussion over the demonstration
in the engagement section.
The instructor will then begin the lesson by giving a lecture over Avogadro’s and GayLussac’s laws.
o Avogadro’s Law
You should remember that Avogadro is most remembered for his work in
defining the number of molecules/atoms/formula units in a mole.
He also did work with gases.
He determined that under constant pressure and temperature, that
the number of moles of a gas is directly correlated to the volume
of a gas.
His law is summarized in the following equation:
o V1/n1 = V2/n2
o Working with Avogadro’s Law
Example
If the volume of 25.0 g of F2 is 15.4 L, what volume would the gas
have if the F2 was decreased to 15.0 g?
Example
If the volume of 30.0 g of methane (CH4) is decreased from 13.2 L
to 9.8 L was amount of mass was lost?
o Gay-Lussac’s Law
Gay-Lussac determined the relationship between the temperature and
pressure of a gas.
He determined that the pressure and temperature of a gas are
directly related.
The instructor should ask the students what they think the equation
will work on will look like.
His law can be represented mathematically by the following
equation:
o P1/T1 = P2/T2
Remember that the temperature has to be in Kelvin
when using this equation.
o Using Gay-Lussac’s Law
Example
What is the pressure of a gas that was originally at 1 atmosphere is
increased from 39 degrees Celsius to 100 degrees Celsius.
After going over the notes for this lesson students will be advised that an additional
review over manometer problems will be given
o This was because most of the students seemed to have difficulties after learning
the material the first time.
The instructor should give a quick review of the material before the class
ends.
To solve a manometer problem, the first step is to draw a picture of
the problem.
o Remember that a manometer is a U shaped tube filled with
mercury.
Revised 4/11
It is probably best to convert all of the pressure units to mmHg as
the height difference will be equal to the pressure difference
between the objects in mmHg.
o You just have to decide whether the pressures should be
added up or subtracted from one another.
To determine this you need to determine whether
the pressure you are solving for has a lower or
higher mercury level than the known side.
The side with the lower mercury level has
the higher pressure.
o This is because more force is being
pushed on the surface of the mercury
forcing it down the tube and up the
other side.
6. Practice/Application
Students will complete a worksheet over using Avogadro’s and Gay-Lussac’s laws
o The worksheet will also cover a review of Boyle’s and Charles’ laws
Students will have to differentiate between the possible correlations
between factors using gas laws.
7. Evaluation of student learning
The instructor should make use of both formal and informal evaluations to gauge student
learning throughout the lesson.
o Formal
The worksheets students are to complete will be collected and graded for
correctness.
Based on the results from the worksheets, adjustments will be
made if necessary.
o Informal
The instructor should make use of probing questions to continuously
monitor and assess student understanding of material presented in this
lesson.
8. Closure
At the end of the lesson, and before students begin to work on their assigned worksheet
o The instructor should be sure to note how the balloon got inside the flask.
o Before telling students however, the instructor should have students try to explain
how the balloon got inside the flask using knowledge they should have gained
through the past two lessons over gas laws.
The reason this does happen is because as the temperature of the gas
decreases the volume the gas occupies decreases, so the pressure from the
atmosphere pushes on the balloon forcing it into the flask.
NOTE: This lesson was modified by giving a review over manometer problems. This was done
due to the fact that many students struggled with answering problems involving a manometer
correctly.
Revised 4/11
 0
0