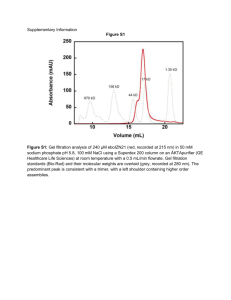
Homework 4: Mueckler
advertisement

Homework 4: Mueckler- Answer Key 1. Describe the basis of the hydrophobic effect and how it influences the structure of membranes and proteins. The hydrophobic effect causes hydrophobic surfaces such as fatty acyl chains to aggregate in water. Water molecules squeeze hydrophobic molecules into as compact a surface area as possible in order to minimize the free energy (ΔG) of the system by maximizing the entropy (ΔS) or degree of disorder of the water molecules. The formation of spherical bilayers is energetically favorable as it prevents edges of the phospholipid bilayer, and thus the nonpolar fatty acid chains, from being exposed to water. 2. What is the minimum number of residues required for a polypeptide domain to completely traverse the fatty acyl core of the lipid bilayer as part of a beta sheet structure? Speculate on why two completely different types of protein secondary structure have evolved to form channels through membranes. Membranes are approximately 30 angstroms thick. β sheets have a rise of 3.3 angstroms/residue. 30A / 3.3A/residue = 9-10 residues α helices: tend to form narrower channels than β sheets, making them ideal for highly selective ion channels. β sheets: tend to form wider, nonselective channels, allowing for the creation of aqueous pores through membranes. 3. What would be the molecular and cellular consequences of adding to cultured mammalian cells a reagent that specifically inhibited the interaction between SRP and its receptor? All protein transport through the secretory pathway would be inhibited. SRP would bind the signal sequences on the N-termini of free ribosomes, but never dock at the ER membrane, as its docking consists of binding to the SRP receptor. The free ribosome would remain bound to SRP with stalled translation at 70-80 residues. Thus, transmembrane and secreted proteins would be produced and ribosomes would become sequestered on ER targeting mRNA. This would eventually result in cell death. 4. Using your knowledge of the secretory pathway, describe one way in which a cytoplasmic protein could be engineered to make it easier to purify in large quantities from cultured cells. Add an 8-12 residue hydrophobic signal sequence to its N-terminus so that the cytoplasmic protein is now targeted to the secretory pathway and secreted to the cell media for easy collection. This 8-12 residue hydrophobic signal sequence will be cleaved during ER translocation, thus yielding the original, unmodified protein. 5. The first scientist to discover a “signal sequence” on a secreted protein subsequently won a nobel prize for a completely different discovery. Who was it? (Hint: it’s not the most obvious person.) Cesar Milstein, the inventor of hybridoma technology. The purpose of this question is to get you to look up and read Blobel’s original papers on the signal hypothesis, where Milstein is briefly mentioned. 6. Summarize the characteristics of mitochondria that support the concept that they represent the remnants of an ancient bacterial “invasion” of a primitive eukaryotic cell. Mitochondria contain their own genome and protein synthetic machinery (tRNAs, mRNAs, ribosomes, initiation and elongation factors, etc.), their DNA is circular, and they undergo binary fission like bacteria. 7. Briefly outline a procedure to separate free polysomes from membrane bound polysomes starting with rat liver tissue. Homogenize the tissue to lyse the cells, centrifuge the solution to remove unlysed cells, perform a sucrose centrifugation to separate the organelles by density. Free polysomes have lower density than membrane bound ribosomes, and thus settle to a lower sucrose concentration. That is, bound polysomes will settle towards the bottom of the tube while free polysomes will stay in lower density sucrose fractions higher in the tube. 8. Briefly outline an approach to identify a nuclear localization sequence in a novel protein that does not possess any of the currently recognized signals. How could you determine which karyopherin is involved in its translocation? Serial deletions or site directed mutagenesis followed by examination of cellular localization through immunoflorescence or centrifugation. The best place to start making deletions are stretches of polybasic residues as many NLSs consist of these residues. Co-immunoprecipitation and look at what binds the NLS by immunoblotting for known targets and/or mass spec for novel ones.