The Biochemistry of Movement
advertisement

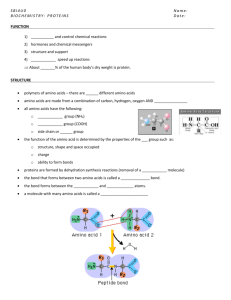
ATP and the structure of proteins page 1 The Biochemistry of Movement ATP is the energy currency of every living cell 1.1 Identify that adenosine triphosphate is used as an energy source for nearly all cellular metabolic processes Metabolism is the sum of the chemical processes through which living things, both plant and animal, gain and use the energy they require to support their physiological needs. The human body, along with other life forms, uses a very complex but highly effective set of chemical reactions to meet its energy needs. Obviously the energy provided by food is not converted into heat energy or light energy in the same way as occurs in the combustion of a heating fuel. Despite this, cellular respiration is very similar to combustion in many respects. In the cell, energy released by the oxidation of biochemical fuels such as glucose is converted into chemical potential energy stored in adenosine triphosphate (ATP). ATP occurs in all known living species. It is the major energy source for cellular metabolic processes. The process of cellular respiration, as it commonly occurs, can be represented by the equation: glucose + oxygen carbon dioxide + water + energy C6H12O6(aq) + 6O2(g) 6CO2(g) + 6H2O(l) + energy (ATP) 1.4 Explain that biochemical fuels are broken down to release energy for making ATP Food provides the chemical compounds that are involved in chemical reactions. Both glucose and fats and oils (lipids) are the main sources of energy in humans. Enzymes catalyse the hydrolysis of carbohydrates into glucose before energy production can start. When glucose is oxidized 2816kj mol-1 of energy is released. ATP is the chemical used to regulate this energy Look at the above reaction This is a very simplified view of a process that has about 140 separate steps each catalysed by an enzyme and, in some cases, requiring the action of coenzymes. The total process of oxidising one glucose molecule is capable of producing 38 ATP molecules. In addition to carbohydrates, other food types such as lipids and proteins are also utilised by the body during cellular respiration. Fats and proteins are converted into intermediate compounds such as acetyl-CoA and pyruvate before entering the process. 1.2 Explain that the biologically important part of the molecule contains three phosphate groups 1.6 solve problems and process information from a diagram or model of the structure of the adenosine triphosphate molecule to discuss the nature and organisation of the phosphate groups Adenosine triphosphate consists of an adenosine core and three attached phosphate groups. This structure is shown below NOTE – The adenosine part of this molecule is made up of the N containing base adenosine and the sugar ribose. When a diester bond is broken by hydrolysis 33kj of energy is released. This energy can be used for muscle contraction, synthesizing reactions, maintaining body temperature and active transport of substances in the body. ATP and the structure of proteins page 2 The phosphate groups are attached in a linear sequence by phosphodiester bonds. The phosphodiester bonds which link the phosphate groups are regarded as high energy bonds and the hydrolysis (reverse of condensation) of a phosphate group in ATP to form adenosine diphosphate (ADP) is a highly exothermic process. As a result, ATP is regarded as an ‘energy rich’ compound. In cellular respiration, the chemical reaction of glucose with oxygen produces carbon dioxide and water. This reaction releases large amounts of energy that are stored in the form of ATP. The ATP can later be used to provide the energy needed for various endothermic biochemical reactions. Most ATP is synthesised within the cellular organelles called mitochondria. The hydrolysis of ATP leads to the release of energy. In the hydrolysis reaction, ATP loses one of its attached phosphate groups (often represented by Pi) and forms the lower energy compound ADP. ADP can be recycled to reform ATP when energy becomes available. This process is called oxidative phosphorylation. These two reversible reactions are represented below. ATP + H2O ADP + Pi (exothermic: releasing energy for cellular processes) ADP + Pi ATP + H2O (endothermic: absorbing energy which is stored in ATP). The amount of ATP required by the human body varies considerably with the degree of physical activity. The body has a well coordinated control system to ensure ATP production matches the level of activity. 1.3 identify the role of enzymes as catalysts in the conversion of ATP to ADP with energy made available for metabolism, given a flow chart of the biochemical pathways Enzymes are biological catalysts. Enzymes provide a pathway of lower activation energy The various metabolic processes require the involvement of enzymes so that these reactions are able to take place under the relatively mild conditions (pH 6 to 8; temperatures 37 0C) that usually prevail in the human body. The interconversion reactions of ATP and ADP do not occur in isolation but couple with other biochemical reactions. Enzymes perform the role of ensuring that all reactions occur at a rapid rate when required. 1.5 and 1.7 Identify mitochondria as the cell organelles involved in aerobic respiration and the site of most ATP synthesis. Process information from secondary sources to locate the site of each step of respiration in the cell There are 3 main stages to aerobic cellular respiration 1) Glycolysis (2 ATP) 2) The Krebs Citric acid cycle (tricarboxylic acid cycle) 2 ATP 3) The cytochrome chain or oxidative phosphorylation (the making of 34 xATP TOTAL ATP = 38 molecules from 1 glucose molecule ATP and the structure of proteins page 3 Draw a diagram of a mitochondria identifying the sites for aerobic respiration Proteins are used as both structural molecules and as enzymes to catalyse metabolic reactions 4.1, 4.2 and 4.9 describe the composition and general formula for amino acids. Identify the major functional groups in an amino acid. Process information from secondary sources to draw the generalised structural formula for an amino acid Proteins, like polysaccharides, are condensation polymers. However the chemical nature of proteins is vastly different. The sub-units or monomers of proteins are amino acids. AMINO ACIDS Proteins within the human body are made up from twenty different amino acids listed in Table 28.6. Their chemical composition varies but all contain the elements carbon, hydrogen, oxygen and nitrogen and some also contain sulfur. Amino acids contain both the amine (–NH2) and alkanoic (carboxylic) acid (–COOH) functional groups. The general structure of an amino acid is: Humans, like most animals, cannot manufacture all of the amino acids we require. We are capable of synthesising some amino acids but must consume the others as part of our diet. The eight (nine for children) amino acids we must consume within our diet are known as essential amino acids and include histidine (for children), isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan and valine. ATP and the structure of proteins page 4 4.3 Outline the nature of a peptide bond and, using a specific example, describe the chemistry involved in the formation of a peptide bond Two amino acids can undergo a condensation reaction to form a peptide. In this reaction the amino group of one amino acid joins with the alkanoic acid group of another to form a peptide bond and eliminate a water molecule. This is illustrated in Figure 28.14. The process can be repeated to form a polypeptide chain which consists of a series of amino acids linked through peptide bonds. Proteins range in size from single polypeptide chains containing about 100 amino acids to giant polymers of about 2000 amino acids. Within a particular protein the amino acids link in a specific order. The long polypeptide chain folds into a relatively stable three-dimensional shape. It is this natural shape of the protein that often determines the role it performs. Draw in your own diagrams of the formation of a peptide bond ATP and the structure of proteins page 5 4.4 and 4.5 Explain, using a named example, the relationship between the chemical features of a protein and its shape using appropriate diagrams or models Account for the shape of a protein molecule in terms of – electrostatic forces – hydrogen bonding forces – hydrophobic forces – disulfide bonds The chemical structure of the –R side chain groups strongly influences the properties of the amino acids. If the side chain is composed almost entirely of carbon and hydrogen atoms then it will be non-polar, as is the case in alanine and isoleucine. If the side chain contains oxygen and nitrogen atoms, as in serine and cysteine, then it will be polar. Some side groups are also charged. The structure of the amino acid side chains greatly influences the threedimensional shape of the protein molecule. Amino acids can function as both acids and bases due to the nature of their two defining functional groups. The alkanoic acid group is capable of ionising and donating a proton, as is shown for glycine. Similarly, the amino group can act as a proton acceptor or base. These acidic and basic groups within an amino acid actually react with one another to form a dipolar (positively and negatively charged) ion. These are called zwitterions and give amino acids many ionic compound characteristics, such as higher than predicted melting points and increased solubilities in water. Two examples of zwitterions are illustrated in Figure 28.13. The shape is determined by a combination of factors and is usually described as having four levels of organisation: primary, secondary, tertiary and quaternary. The primary structure is the sequence of amino acids within the protein polypeptide. While there are only 20 amino acids, the variety of combinations that can be created from them leads to a huge number of proteins. For convenience, amino acids are often indicated by the three-letter codes listed in Table 28.6. The code for a very small protein consisting of only nine amino acids, the hormone vasopressin, is cys-tyr-phe-glnasn-cys-pro-arg-gly. The secondary structure of proteins relates to the spatial arrangement of the polypeptide chain. The rigid planar structure of the peptide group determines the ways in which it may fold. Several different folding patterns may result— spiral helices, pleated sheets and turns. The tertiary structure refers to the overall folding pattern of the polypeptide chain. The tertiary structure is the threedimensional folding superimposed on the secondary helical, folded or pleated structure. If the secondary structure of the protein is helical then it is analogous to the folding of a spring. The particular folding pattern depends on the formation of hydrogen bonds, covalent disulfide bonds and dispersion forces that stabilise a particular arrangement. The tertiary structure of proteins may result in long elastic proteins required for structural proteins such as keratin or collagen or the globular shape of hemoglobin. The quaternary structure is only relevant where a protein includes two or more polypeptide chains. It refers to the way in which individual polypeptide chains form an aggregated whole. An example of quaternary structure is the formation of haemoglobin from four separate polypeptide units illustrated in Figure 28.15. ATP and the structure of proteins page 6 The forces and bonds which determine the final shape of a protein molecule, apart from the peptide bonds linking the amino acids, are electrostatic forces, hydrogen bonding, hydrophobic forces, and covalent disulfide bonds. Electrostatic (ionic) forces These result from the ionic nature of the side chains in some amino acids. These forces are relatively strong. Hydrogen bonding forces Hydrogen bonds readily form between H atoms attached to an F, O or N atom and another F, O or N atom. In the case of a protein coil, hydrogen bonding between the oxygen of a carbonyl (–C=O) group and hydrogen attached to a nitrogen atom plays a major role in determining the secondary structure of proteins. In addition, the location of polar side groups and hydrogen bonding with water molecules is an important factor in the solubility of globular proteins. Hydrophobic (dispersion) forces The non-polar side chains of some amino acids result in them exhibiting hydrophobic (water hating) behaviour. In globular proteins these non-polar side chains minimise their contact with polar water molecules by folding in so as to turn all the hydrophobic side groups inside, away from contact with the polar solvent. The intermolecular forces between these hydrophobic groups are dispersion forces or hydrophobic forces. Disulfide (covalent) bonds Disulfide bonds form crosslinks between sections of a folded protein molecule. These covalent bonds result when two sections of protein containing the –S–H group align and react to produce a disulfide covalent –S–S–. These bonds give considerable stability to the three-dimensional protein shape (see Fig. 28.16). The history of our understanding of protein structure is relatively recent. The first protein sequence to be determined ox insulin in 1953. This is a very small protein containing only 51 amino (Fig. 28.16). We now know the structure of several thousand proteins. bond was acids ATP and the structure of proteins page 7 4.6 Account for the process of protein denaturation PROTEIN DENATURATION As previously mentioned, the three dimensional shape of a protein is critical in it performing its specific function within the body. Disruption to this shape renders the protein biologically inactive. This process is called denaturation. It involves disrupting the forces and bonds which determine secondary, tertiary and, if relevant, quaternary structure of the protein, while leaving the chain of amino acids intact. Various factors can denature proteins, most irreversibly. These include temperature, pH and various chemicals. Increasing temperature increases the thermal motion of protein molecules and may disrupt the forces maintaining their particular arrangement. The protein molecule may unravel and then coagulate into solid lumps. This is illustrated by the denaturing of the protein albumin when egg white is cooked. Changes in pH can interfere with electrostatic forces, hydrogen bonds and disulfide bonds, thus destabilising the protein structure. This is what occurs when milk curdles. Bacteria in milk produce lactic acid, causing a change in the pH of the milk and the denaturation of casein (milk protein). Chemicals such as detergents, salts and alcohols can also interfere with hydrophobic forces and destabilise protein structure. 4.7 Identify enzymes as a special class of proteins with a binding site that is substrate specific 4.8 Using a named example of an enzyme, explain why the enzyme’s binding site is substrate specific 28.4 ENZYMES A group of proteins known as enzymes is responsible for catalysing almost all of the chemical reactions which occur in our bodies. The major reactions which occur in a cell can be summarised in a complicated flowchart showing the major energy producing pathways (see chemistry syllabus) Enzymes are almost always specific to a particular reaction, so to catalyse the many reactions involved in body processes thousands of enzymes are required. Enzymes are similar to other catalysts in that they speed up reactions, are not consumed by the reactions and do not alter the equilibrium position of a reaction. It should be noted that very few reactions in the body are ever in a state of equilibrium, as products are constantly being removed from the reaction site. Enzyme-catalysed reactions occur in very mild conditions, unlike many of the catalysed reactions in industry. Enzymes are very efficient, speeding up reactions by much greater rates than most chemical catalysts, and they are also more specific than most chemical catalysts, acting upon specific molecules called substrates. For example, the enzyme zymase catalyses the fermentation of glucose to form ethanol, rennin is used in the manufacture of cheese from milk, and chymotrypsin is one of many enzymes that catalyses the digestion of foods. Most enzymes are effective over restricted pH and temperature ranges. At higher temperatures and outside the preferred pH range the enzyme is often denatured and therefore loses its catalytic activity. MECHANISM OF ENZYME ACTION Enzymes function as catalysts by bringing together the reactant molecules and providing an ideal environment for bond breaking and bond making. For an enzyme to catalyse a reaction it must come into contact with the particular reactant or substrate molecule(s). The substrate and the enzyme must interact in a specific manner at what is termed the active site of the enzyme. This interaction is illustrated in Figure 28.18. This mechanism of action explains why enzymes are so specific in their activity. For a substrate to bind effectively to an enzyme, bonds must form between the active site of the enzyme and the substrate. The active site is located within the folded protein structure. These bonds are similar to the various bonds and forces which determine the secondary and tertiary structure of proteins. If an incorrect substrate comes into contact with an enzyme these bonds will not form because the shapes of the enzyme and substrate are not complementary. Because of the need for a precise ‘fit’ between the enzyme and substrate molecules, the mechanism of enzyme catalysed reactions are often discussed in terms of a ‘lock and key’ mechanism. If the active site of the enzyme has a specific shape (similar to a lock) then only the right shaped substrate (key) will fit that site. The enzyme that performs the role of breaking potentially toxic hydrogen peroxide into oxygen and water is called catalase. It speeds up this decomposition reaction by a factor of 109. Hydrogen peroxide is a strong oxidising agent. It is produced as a by-product of some cellular ATP and the structure of proteins reactions and must constantly be removed. The active site of the catalase molecules bonds only with hydrogen peroxide and facilitates its decomposition into oxygen and water. This prevents a build up of cellular hydrogen peroxide that could be fatal. page 8 4. 11 Process and analyse information from secondary sources to discuss the use of models in the development of understanding of enzyme function Class discussion and notes FIRST HAND INVESTIGATIONS 4.10 Solve problems, identify data, plan, choose equipment and perform first-hand investigations to observe the effect of changes in pH and temperature on a named enzyme reaction and use the available evidence to relate this to changes in the primary, secondary and/or tertiary structure of the enzyme involved You are to perform these experiments in class and complete a formal report