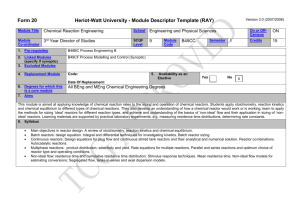
Course File - Chemical Engineering and Applied Chemistry
advertisement
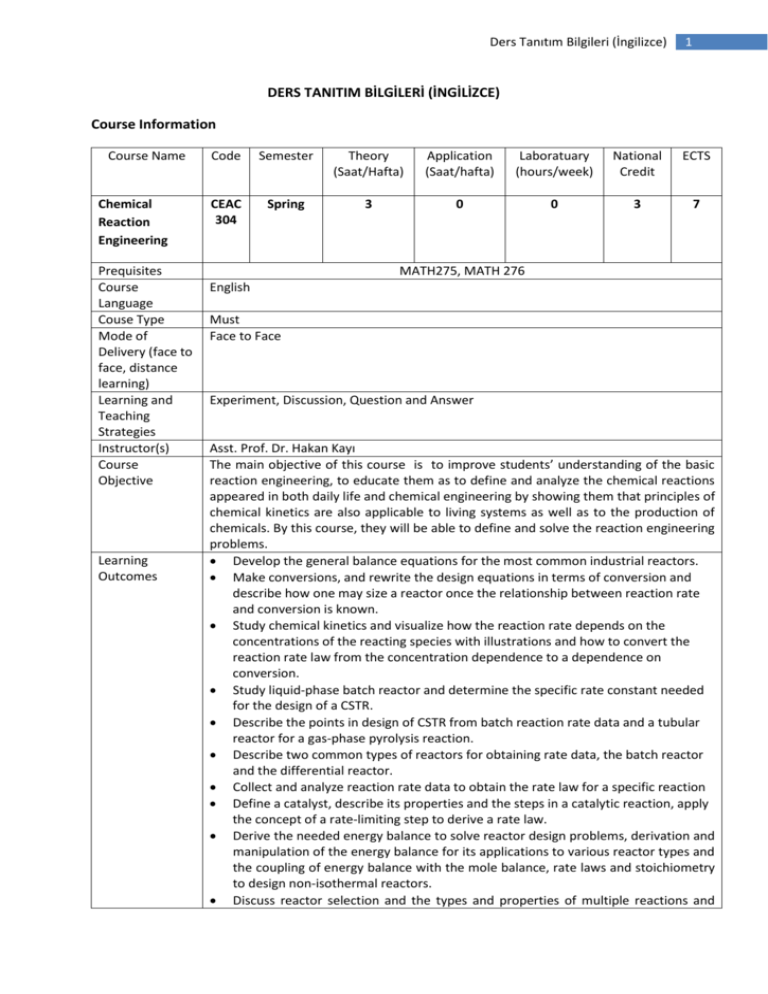
Ders Tanıtım Bilgileri (İngilizce) 1 DERS TANITIM BİLGİLERİ (İNGİLİZCE) Course Information Course Name Chemical Reaction Engineering Prequisites Course Language Couse Type Mode of Delivery (face to face, distance learning) Learning and Teaching Strategies Instructor(s) Course Objective Learning Outcomes Code Semester Theory (Saat/Hafta) Application (Saat/hafta) Laboratuary (hours/week) National Credit ECTS CEAC 304 Spring 3 0 0 3 7 MATH275, MATH 276 English Must Face to Face Experiment, Discussion, Question and Answer Asst. Prof. Dr. Hakan Kayı The main objective of this course is to improve students’ understanding of the basic reaction engineering, to educate them as to define and analyze the chemical reactions appeared in both daily life and chemical engineering by showing them that principles of chemical kinetics are also applicable to living systems as well as to the production of chemicals. By this course, they will be able to define and solve the reaction engineering problems. Develop the general balance equations for the most common industrial reactors. Make conversions, and rewrite the design equations in terms of conversion and describe how one may size a reactor once the relationship between reaction rate and conversion is known. Study chemical kinetics and visualize how the reaction rate depends on the concentrations of the reacting species with illustrations and how to convert the reaction rate law from the concentration dependence to a dependence on conversion. Study liquid-phase batch reactor and determine the specific rate constant needed for the design of a CSTR. Describe the points in design of CSTR from batch reaction rate data and a tubular reactor for a gas-phase pyrolysis reaction. Describe two common types of reactors for obtaining rate data, the batch reactor and the differential reactor. Collect and analyze reaction rate data to obtain the rate law for a specific reaction Define a catalyst, describe its properties and the steps in a catalytic reaction, apply the concept of a rate-limiting step to derive a rate law. Derive the needed energy balance to solve reactor design problems, derivation and manipulation of the energy balance for its applications to various reactor types and the coupling of energy balance with the mole balance, rate laws and stoichiometry to design non-isothermal reactors. Discuss reactor selection and the types and properties of multiple reactions and Ders Tanıtım Bilgileri (İngilizce) 2 derive general mole balances for simultaneous reactions. Describe types and properties of slurry and chemostat multiphase reactors and also of the growth and inhibition kinetics as well as the mass balance equations and stoichiometric relationships for bioreactors. Diagnose and handle chemical reactors whose performance deviates from the ideal and principles behind the analysis of non-ideal reactors. Model the reactor flow pattern using combinations and/or modifications of ideal reactors to represent real reactors, to classify a model as a one-parameter model or a two- parameter model and to use RTD for evaluation of parameter(s) in the model. Course Content More Balances, Conversion and Reactor Sizing, Rate Laws and Stoichiometry, Isothermal Reactor Design, Collection and Analysis of Rate Data, Catalysis and Catalytic Reactors, Nonisothermal Reactor Design, Multiple Reactions, Multiphase Reactors, Distributions of Residence Times for Chemical Reactors, Analysis of Nonideal Reactors. References Course Book: Fogler H. S., Elements of Chemical Reaction Engineering, 2nd Ed., P T R Prentice-Hall, Inc., 1992. Weekly Course outline Weeks Topics Pre-study 1. Week 2. Week 3. Week More Balances Conversion and Reactor Sizing Rate Laws and Stoichiometry 1-29 29-61 61-106 4. Week Isothermal Reactor Design 106-144 144-190 Isothermal Reactor Design 5. Week 6. Week Collection and Analysis of Rate Data 190-211 Collection and Analysis of Rate Data 214-241 MIDTERM EXAMINATION I 7. Week Catalysis and Catalytic Reactors 241-289 8. Week Catalysis and Catalytic Reactors 289-338 9. Week 10.Week 11.Week 12. Week Non-isothermal Reactor Design Non-isothermal Reactor Design Multiple Reactions 384-434 434-486 486-543 MIDTERM EXAMINATION II 13. Week Multiphase Reactors 660-708 14. Week Distributions of Residence Times for Chemical Reactors 708-759 Ders Tanıtım Bilgileri (İngilizce) 15. Week 759-802 Analysis of Nonideal Reactors 16. Week FINAL EXAMINATION Assessment methods Course Activities Number Percentage % 2 1 60 40 100 60 40 100 Attendance Laboratory Application Field Activities Specific Practical Training (if any) Assignments Presentation Projects Seminars Midterms Final Exam Total Percentage of semester activities contributing grade success Percentage of final exam contributing grade success Total Course Category Core Courses X Major Area Courses Supportive Courses Media and Management Skills Courses Transferable Skill Courses Workload and ECTS Calculation Activities Course Duration (x16) Laboratory Application (Quizzes) Specific practical training (if any) Field Activities Study Hours Out of Class (Preliminary work, Number Duration (Hours) Total Work Load 16 4 64 16 4 64 3 Ders Tanıtım Bilgileri (İngilizce) reinforcement, ect) Presentation / Seminar Preparation Projects Homework assignment Midterms ( Study duration ) Final ( Study duration ) Total Workload 2 1 25 35 4 50 35 213 Matrix of the Course Learning Outcomes Versus Program Outcomes Program Outcomes 1. An ability to apply knowledge of mathematics, science, and engineering to solve chemical engineering and applied chemistry problems. 2. An ability to analyze and model a domain specific problem, identify and define the appropriate requirements for its solution. 3. An ability to design, implement and evaluate a chemical engineering system or a system component to meet specified requirements. 4. An ability to use the modern techniques and engineering tools necessary for chemical engineering practices. 5. An ability to acquire, analyze and interpret data to understand chemical engineering and applied chemistry requirements. 6. The ability to demonstrate the necessary organizational and business skills to work effectively in inter/inner disciplinary teams or individually. 7. An ability to communicate effectively in Turkish and English. 8. Recognition of the need for, and the ability to access information, to follow recent developments in science and technology and to engage in life-long learning. 9. An understanding of professional, legal, ethical and social issues and responsibilities in chemical engineering and applied chemistry 10. Skills in project and risk management, awareness about importance of entrepreneurship, innovation and long-term development, and recognition of international standards and methodologies. 1: Lowest, 2: Low, 3: Average, 4: High, 5: Highest Contribution Level* 1 2 3 4 5 X X X X X X X X X X