Week 1 - WebAssign
advertisement

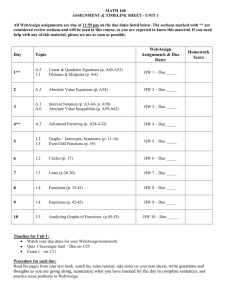
Weeks 1-2: Once you have enrolled at Webassign you have to get used to using it. The first assignment is an Introduction to Webassign. It’s very important to complete these first exercises before going to chapter 1. A traditional class starts with a lecture, followed by text assignments. Students try the assignment first and if they need help, they go over the chapter and their notes. We are going to duplicate this online. We will follow this pattern: Thinkwell Video Lessons Webassign assignments Text sections The thinkwell video lessons take the place of the lecture. You can match up the title of the video with the assignment which helps a lot. Just like a traditional class, sometimes the lecture is not enough. This is when you go to the text. Whether you start with the videos or the text, make sure you complete the Webassign exercises. These exercises are the same as the end of chapter questions in the text but with different data. Webassign remembers your answers so you can gradually complete the exercises for the week. Most exercises will allow up to 3 submissions of answers. If you get the first submission wrong, “Hints” may appear to help you. If you are still stuck on a question, post your question at the “Forum” on Webassign. The average of your webassign assignments will equal an exam grade. There is also a Chang text site for an earlier edition that contains animations, selfassessment quizzes, and an interactive study guide. Some will find the interactive study guide very useful. The only drawback is you have to use internet explorer to use the interactives. Here is the address: http://highered.mcgrawhill.com/sites/0072512644/student_view0/interactive_study_guid e.html Click on “interactive study guide” then choose chapter 1. Here is a breakdown by text section for the weeks1-2. Section 1.1-1.3: This week you will review how Chemistry helps explain the world around us by using the scientific method to study nature. The scientific method is a logical problem solving method we all use without realizing it. Section 1.3: We study nature by observing it, especially by taking and using measurements. The international system (SI) is used rather than the metric system for our measurement units. Familiarize yourself with table 1.1. The metric prefixes in 1.2 must be memorized for the first exam and will be used later this chapter in problem solving. 1.4-1.6: Classification of Matter-Matter can be classified in different ways, by state or by properties. You should know the basic classification of matter as is summarized in figure 1.14 on page 29 in the text. You will be asked to classify various examples of matter as elements, compounds, homogenous mixtures and heterogeneous mixtures. Matter can undergo physical and chemical changes. You should know the difference between these changes and be able to classify changes as either one of these. 1.7: Temperature- Converting between Celcius and Fahrenheit doesn’t happen that much anymore but you still have to be able to do it and we definitely still convert between Celcius and Kelvin. You should know the formulas: oF = 1.8xoC +32 and K = oC + 273 Density-You should know how to find and use density. You will be doing a lab on density so much of this will be seen there. There is a visualization on “properties of matter” that goes over density as well as the differences in physical and chemical changes. Here are the Thinkwell Video Lessons for weeks 1-2: An Introduction to Chemistry and the Scientific Method An Introduction to Chemistry The Scientific Method Properties of Matter States of Matter CIA Demonstration: Differences in Density Due to Temperature Properties of Matter