OIE?????????????????????
advertisement

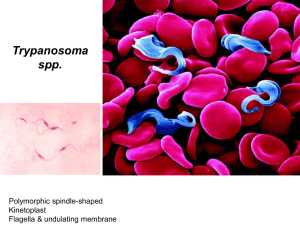
CHAPTER 1 2.2.18. 3 TRYPANOSOMA EVANSI INFECTIONS (INCLUDING SURRA) 4 SUMMARY 5 6 7 8 9 10 Definition of the disease: Trypanosoma evansi causes a disease known as Trypanosomosis 1 (‘surra’). It affects a number of domesticated animals species in Asia, Africa and Central and South America. The principal host species affected varies geographically, but buffalo, cattle, camels and horses are particularly affected, although other animals, including wildlife, are also susceptible. It is an arthropod-borne disease and several species of haematophagous flies, including Tabanus spp. and Musca spp., are implicated as vectors. 11 12 13 14 15 16 Description of the disease: The disease in susceptible animals is manifested by pyrexia, directly associated with parasitaemia, together with a progressive anaemia, loss of condition and lassitude. Recurrent episodes of fever and parasitaemia occur during the course of the disease. Oedema, particularly of the lower parts of the body, urticarial plaques and petechial haemorrhages of the serous membranes are often observed. Abortions have been reported in buffaloes and camels. There are indications that the disease causes immunodeficiencies. 17 18 19 20 21 22 23 Identification of the agent: The general clinical signs of T. evansi infection are not sufficiently pathognomonic for diagnosis and laboratory methods for detecting the parasite are required. Examination of the host blood is problematic as trypanosomes can be detected only when there is a high parasitaemia. Under these circumstances examination of wet blood films, stained blood smears or lymph node material might reveal the trypanosomes. In other, more chronic cases, such as the carrier state, the examination of thick blood smears, as well as methods of parasite concentration and the inoculation of laboratory animals, are required. 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 Serological tests: Infection gives rise to specific antibody responses and a variety of antibody detection tests have been introduced for laboratory use. Some have been partially validated, but await large-scale evaluation and standardisation. Among those that are used regularly are immunoenzyme assays, card agglutination tests and latex agglutination tests. For field use, relatively simple tests have yet to be developed although obvious candidates might be indirect agglutination tests using antigen-coated latex particles. Assays for detection of circulating antibodies have high measures of validity. Estimates of predictive values of different serological tests indicate that enzyme linked immunosorbent assays (ELISA) for detecting IgG antibodies are more likely to classify correctly uninfected animals, and card agglutination tests (CATT) are more likely to classify correctly truly infected animals. An IgG ELISA would thus be suitable for verifying that animals are free from infection, prior to movement or during quarantine. In situations where there is overt disease, CATTs can be used to target individual animals for treatment with trypanocidal drugs. For declaring a disease-free status, serial testing – ELISA followed by retesting of suspect samples by CATT – is recommended. It must be stressed however, that there are considerable antigenic similarities among the different species of pathogenic trypanosomes, hence in areas where tsetse-transmitted trypanosomoses occur cross-reactions will occur with any serological test employed. 41 42 Requirements for vaccines and diagnostic biologicals: No vaccines are available for the disease. 2 1 Nomenclature of parasitic diseases: see the note in Chapter. 2.3.17. Trypanosomosis (Tsetse-borne). OIE Terrestrial Manual 2008 1 Chapter 2.2.18. – Trypanosoma evansi infections (including surra) A. 43 INTRODUCTION 44 45 46 47 48 49 50 51 The clinical signs of surra, the disease caused by Trypansoma evansi, are indicative but are not sufficiently pathognomonic and diagnosis must be confirmed by laboratory methods. The disease in susceptible animals, including cattle, buffalo, camels (dromedary and bactrian), horses, pigs, sheep and goats, is manifested by pyrexia, directly associated with parasitaemia, together with a progressive anaemia, loss of condition and lassitude. Recurrent episodes of fever and parasitaemia occur during the course of the disease. Oedema, particularly of the lower parts of the body, urticarial plaques and petechial haemorrhages of the serous membranes are often observed. Abortions have been reported in buffaloes and camels (7, 15) and there are indications that the disease causes immunodeficiency (4, 19). 52 53 54 55 56 There is considerable variation in the pathogenicity of different strains and the susceptibility of different host species to disease. Disease may manifest as an acute or chronic condition, and in the latter case may persist for many months. The disease is often rapidly fatal in camels, buffaloes, horses, cattle, llama and dogs, but mild and subclinical infections can also occur in these species. Wild animals such as deer and capybara can become infected. Animals subjected to stress – malnutrition, pregnancy, work – are more susceptible to disease. 57 58 59 60 61 62 63 64 Biologically T. evansi is very similar to T. equiperdum, the causative agent of dourine (2), and morphologically resembles the slender forms of the tsetse-transmitted trypanosomes, T. brucei, T. gambiense and T. rhodesiense. Molecular characterisation indicates that various strains of T. evansi isolated form Asia, Africa and South America have a single origin. Moreover, it is also likely that T. evansi and T. equiperdum belong to the same species. Like all pathogenic trypanosomes, T. evansi is covered by a dense protein layer consisting of a single protein called the variable surface glycoprotein. This acts as a major immunogen and elicits the formation of specific antibodies. The parasites are able to evade the consequences of these immune reactions by switching the variant surface glycoprotein, the phenomenon known as antigenic variation . B. 65 DIAGNOSTIC TECHNIQUES 66 1. Identification of the agent 67 68 69 70 The classical direct parasitological methods for the diagnosis of trypanosomosis, namely examining blood or lymph node material, are not highly sensitive. In regions where other Trypanozoon spp. occur in addition to T. evansi, specific identification by microscopy is not possible. Specific DNA probes (17, 22) may enable identification of trypanosome species by nonradioactive DNA hybridisation. 71 • 72 a) Usual field methods 73 Direct methods i) Blood sampling 74 75 76 77 Trypansoma evansi is a parasite of the blood and tissues often inhabiting the deep blood vessels in cases of low parasitaemia. For this reason, it is recommended that blood for diagnosis be obtained from both the peripheral and deep blood vessels. However it should be realised that less than 50% of infected animals may be identified by examination of peripheral blood. 78 79 80 81 82 Peripheral blood is obtained by puncturing a small vein in the ear or tail. Deeper samples are taken from a larger vein by syringe. Cleanse an area of the ear margin or tip of the tail with alcohol and, when dry, puncture a vein is with a suitable instrument. Ensure that instruments are sterilised or disposable instruments are used between individual animals, so that infection cannot be transmitted by residual blood. 83 ii) 84 85 Wet blood films Place a small drop of blood on to a clean glass slide and cover with a cover-slip to spread the blood as a monolayer of cells. Examine by light microscopy (×200) to detect any motile trypanosomes. 86 iii) 87 88 89 90 91 92 93 Stained thick smears Place a large drop of blood on the centre of a microscope slide and spread with a toothpick or the corner of another slide so that an area of approximately 1.0–1.25 cm in diameter is covered. Air-dry for 1 hour or longer, while protecting the slide from insects. Stain the unfixed smear with Giemsa’s Stain (one drop of commercial Giemsa + 1 ml of phosphate buffered saline [PBS, 2.4 g Na2HPO4.2H2O, 0.54 g NaH2PO4.2H2O, 0.34 g NaCl], pH 7.2), for 25 minutes. After washing, examine the smears by light microscopy at high magnification (×500–1000). The advantage of the thick smear technique is that it concentrates the drop of blood into a small area, and thus less time is required to detect the 2 OIE Terrestrial Manual 2008 Chapter 2.2.18. – Trypanosoma evansi infections (including surra) 94 95 parasites. The disadvantage is that the trypanosomes may be damaged in the process, and the method is therefore not suited for species identification in case of mixed infections. 96 iv) 97 98 99 100 101 102 103 104 105 Place a drop of blood 20 mm from one end of a clean microscope slide and draw out a thin film in the usual way. Air-dry briefly and fix in methyl alcohol for 2 minutes and allow to dry. Stain the smears in Giemsa (one drop Giemsa + 1 ml PBS, pH 7.2) for 25 minutes. Pour off, stain and wash the slide in tap water and dry. Unfixed smears can be stained by covering them with May–Grünwald stain for 2 minutes, then adding an equal volume of PBS, pH 7.2, and leaving the slides for a further 3 minutes. Pour off and add diluted Giemsa for 25 minutes. Pour off, wash the slides with tap water, and dry. Examine at high magnification (×400–1000). This technique permits detailed morphological studies and identification of the trypanosome species. Rapid staining techniques also exist (Field’s stain, Diff Quick®). 106 v) 107 108 109 110 111 112 Stained thin smears Lymph node biopsies Samples are usually obtained from the prescapular or precrural (subiliac) lymph nodes. Select a suitable node by palpation and cleanse the site with alcohol. Puncture the node with a suitable gauge needle, and draw lymph node material into a syringe attached to the needle. Expel lymph on to a slide, cover with a cover-slip and examine as for the fresh blood preparations. Fixed thin or thick smears can also be stored for later examination. b) Concentration methods 113 114 115 In most hosts T. evansi can induce mild clinical or subclinical carrier state infections with low parasitaemia in which it is difficult to demonstrate the parasites. In these circumstances, concentration methods become necessary. 116 i) 117 118 119 120 121 122 123 Haematocrit centrifugation Collect blood (70 µl) into at least two heparinised capillary tubes (75 × 1.5 mm). Seal at the dry end by heat and centrifuge, sealed end down, at 3000 g for 10 minutes. Place the capillary tube between two pieces of glass (25 × 10 × 1.2 mm) glued to a slide. Place a cover-slip on top at the level of the buffy coat junction where the trypanosomes will be concentrated. Flood the space around this part of the tube with water or immersion oil, and examine the buffy coat area under the microscope (×100–200). A simpler alternative is to examine the centrifuged capillary tube by placing a drop of immersion oil on the tube and ensuring that there is contact between the objective lens and the immersion oil. 124 ii) Dark-ground/phase-contrast buffy coat technique 125 126 127 Collect blood into heparinised capillary tubes and centrifuge as above. Scratch the tube with a glasscutting diamond and break it 1 mm below the buffy coat layer – the upper part thus contains the top layer of red blood cells (RBCs), the buffy coat (white blood cells) and some plasma. 128 129 Partially expel the contents of this piece on to a slide, cover with a cover-slip and examine under darkground, phase-contrast or ordinary illumination. 130 131 As an alternative to the electrically powered haematocrit centrifuge, hand-powered micro-centrifuges have been used successfully for detection of trypanosomosis in cattle and camels (8, 13). 132 iii) 133 134 135 136 137 Haemolysis techniques Sodium dodecyl sulphate (SDS) can be used as a reagent to haemolyse RBCs to facilitate detection of motile trypanosomes in parasitised blood samples. As SDS is toxic, contact with skin, inhalation and ingestion should be avoided. SDS solution can be stored for several months at ambient temperature. Both the SDS solution and the blood samples should be used at a temperature above 15°C. At lower temperatures the trypanosomes may be destroyed. 138 Two general procedures, namely wet blood film clarification and haemolysis centrifugation, can be used 2. 139 140 141 142 143 Wet blood film clarification method This method uses the partial lysis of RBCs to facilitate the detection of motile trypanosomes. The method requires an SDS solution: 1% SDS dissolved in Tris/glucose/saline, pH 7.5, inoculating loops (10 µl), slides and cover-slips (24 × 24 mm), and a drop of fresh neat or heparinised blood. Dissolve 100 mg of SDS in 100 ml of isotonic Tris/NaCl/glucose buffer, pH 7.5 (Trizma base 14.0 g, NaCl 3.8 g, 2 All necessary materials and instructions can be obtained from the Institute of Tropical Medicine, Laboratory of Serology, Nationalestraat 155, B-2000 Antwerp, Belgium. OIE Terrestrial Manual 2008 3 Chapter 2.2.18. – Trypanosoma evansi infections (including surra) 144 145 146 147 148 glucose 10.8 g; dissolve chemicals in 750 ml distilled H2O, add 90–100 ml 1 N HCl and adjust pH to 7.5 then make up to final volume of 1000 ml with distilled H2O). This buffer can be stored in small vials at ambient temperature for several months. The test does not give a significantly higher sensitivity than wet film technique. In addition, the SDS can cause problems when focusing the microscope and the movements of trypanosomes can be severely curtailed owing to the high viscosity of the SDS. 149 150 151 Put approximately 10 µl of blood on a slide. Add 10 µl of SDS solution using a dip inoculation loop and mix gently. Apply a cover-slip and examine the preparation without delay at low magnification (×100 or ×200). 152 Haemolysis centrifugation technique 153 154 155 156 Nearly complete lysis of RBCs is required for this procedure. The materials needed include: SDS solution (0.1% SDS dissolved in Tris/glucose/saline, pH 7.5), conical centrifuge tubes, ordinary test tubes, large and fine plastic tapering pipettes with attached bulb, slides, cover-slips (24 × 24 mm or 24 × 32 mm), and heparinised blood. 157 158 159 160 161 Using a pipette or syringe, transfer nine volumes (maximum 6.3 ml) of SDS solution into an ordinary test tube. Aspirate one volume (maximum 0.7 ml) of heparinised blood into a bulb pipette and expel it just above the surface of the SDS solution; mix quickly and thoroughly. Avoid foam formation, which may result in destruction of the trypanosomes. Wait for 10 minutes so as to achieve complete haemolysis. 162 163 164 165 Pour the haemolysed suspension into a conical centrifuge tube and spin at approximately 500 g for 10 minutes. With a clean bulb pipette, remove as much supernatant as possible without disturbing the sediment. Using a fine tapering bulb pipette, remove more supernatant, leaving 10–20 µl of undisturbed sediment at the bottom. 166 167 Very carefully collect the entire sediment and put on to a microscope slide. Apply a cover-slip and examine the preparation without delay at low magnification (×100 or ×200). 168 iv) 169 170 171 172 173 174 Mini-anion exchange centrifugation technique When a blood sample from animals infected with salivarian trypanosomes is passed through an appropriate anion-exchange column, the host blood cells, being more negatively charged than trypanosomes, are adsorbed onto the anion-exchanger, while the trypanosomes are eluted, retaining viability and infectivity. A simplified field method for detection of low parasitaemia has been developed (24). The sensitivity of this technique can be increased by approximately tenfold by the use of buffy coat preparations rather than whole blood (23). 175 Preparation of phosphate buffered saline glucose, pH 8 176 Na2HPO4 (anhydrous) (13.48 g); NaH2PO4.2H2O (0.78 g); NaCl (4.25 g); distilled water (1 litre). 177 178 179 180 181 Solutions of different ionic strength are made by diluting the stock PBS, pH 8, and adding glucose to maintain a suitable concentration. For blood of mice, domestic and wild ruminants and dog, add four parts PBS to six parts distilled water and adjust the final glucose concentration to 1%. For blood of pigs and rabbits, add three parts PBS to seven parts distilled water and adjust the final glucose concentration to 1.5%. The PBS/glucose solution (PSG) must be sterile. 182 183 184 185 186 Equilibration of DEAE-cellulose Suspend 500 g of DEAE-cellulose (diethylamino-ethylcellulose) in 2 litres of distilled water. Adjust the pH to 8 with phosphoric acid. Allow to settle for 30 minutes. Discard the supernatant fluid containing the fine granules. Repeat the procedure three times. Store the equilibrated concentrated suspension of DEAE-cellulose (slurry) at 4°C or in small aliquots at –20°C. 187 188 189 190 191 192 Packing of equilibrated DEAE-cellulose Place a 2 ml syringe without the plunger on a test-tube rack complete with needle (20 G × 1.5 inch). Put a disc of Whatman No. 41 filter paper at the bottom of the syringe and moisten by adding a few drops of PSG. Pour 2–2.5 ml of the slurry of equilibrated cellulose into the syringe and allow to pack by elution of the buffer. The height of the sediment should be approximately 3 cm. Wash and equilibrate the column with 2 ml of PSG without disturbing the surface. 193 194 195 196 197 Adsorption of blood eluate of the trypanosomes Gently place 100–300 µl of heparinised blood above the surface of the cellulose column.Add ten drops of PSG and discard the first ten drops of the eluate. Progressively add 1.5 ml of PSG and start collecting the eluate into a finely tapered Pasteur pipette with a sealed end. Put the filled pipette, protected by a conical plastic pipette tip, in a tube and centrifuge at 525 g (or up to 1000 g) for 4 OIE Terrestrial Manual 2008 Chapter 2.2.18. – Trypanosoma evansi infections (including surra) 198 199 200 10 minutes. Examine the bottom of the pipette under the microscope (×100 or ×200) using a special mounting device. Alternatively, the eluate could be collected into 50 ml plastic tubes, with conical bottoms, centrifuged at 1000 g and the sediment examined by dark-ground microscopy. 201 The cellulose column should remain wet throughout the procedure. 202 c) Animal inoculation 203 204 205 206 207 Laboratory animals may be used to reveal subclinical (nonpatent) infections in domesticated animals. Trypanosoma evansi has a broad spectrum of infectivity for small rodents, and so rats and mice are often used. Rodent inoculation is not 100% sensitive (16) but further improvement in its efficacy can be obtained by the use of buffy coat material. Such a procedure was able to detect as few as 1.25 T. evansi/ ml blood (23). 208 209 210 211 212 213 Inoculate heparinised blood intraperitoneally into rats (1–2 ml) or mice (0.25–0.5 ml). Inoculate a minimum of two animals. Bleed animals from the tail three times a week to detect parasitaemia. The incubation period before appearance of the parasites and their virulence depends on the strain of trypanosomes, their concentration in the inoculum, and the strain of laboratory animal used. Sensitivity of this in-vivo culture system may perhaps be increased by use of immunosuppressed laboratory animals. Drugs such as cyclophosphamide or hydrocortisone acetate, or X-ray irradiation or splenectomy are used for this purpose. 214 d) Recombinant DNA probes 215 216 217 218 Specific DNA probes have been used to detect trypanosomes in infected blood or tissue but are not routinely applied (26, 28). Although molecular methods have a potentially high analytical sensitivity there have been few convincing studies to critically evaluate the diagnostic sensitivity of these tests compared with other techniques, such as serology. 219 • Indirect methods 220 221 These methods involve tests that demonstrate the effects of the parasite on its host rather than directly detecting the parasite itself. 222 a) Haematology 223 224 Anaemia is usually a reliable indicator of trypanosome infection, although it is not in itself pathognomonic. However, animals with a mild subclinical infection can have parasitaemia without evidence of anaemia (3). 225 226 227 Anaemia can be estimated by measuring the packed cell volume and may be used in surveys of herds at risk. The technique is identical to that of haematocrit centrifugation. The capillary tube is examined and the results are expressed as a percentage of packed RBCs to total blood volume. 228 229 230 231 232 b) Detection of trypanosomal DNA Detection of minute amounts of trypanosomal DNA using a polymerase chain reaction (PCR) procedure is a possible means of identifying animals with active infections, and could have the sensitivity and specificity required (18, 29). Experimental studies in buffalo (9) showed the diagnostic sensitivity of a PCR was only 78%, similar to that of mouse inoculation. 233 2. Serological tests 234 235 236 237 238 239 240 241 242 243 244 Historically, many different methods have been used to detect specific humoral antibodies to trypanosomal antigens but, more recently, there has been a tendency to concentrate on more easily standardised techniques such as ELISA (5, 6, 11, 20, 21, 22, 25) card agglutination tests (CATT) (1, 18, 22) and latex agglutination tests (LAT) (10, 14). Extensive evaluation of ELISA and CATT has been carried out in buffaloes in Indonesia and Vietnam (5, 10, 27). The collection of samples can be simplified by using filter paper blood spots for later use in the ELISA, while for the CATT whole blood can be substituted for serum (10). Other innovative modifications that might be developed in the future are the use of a colloidal-dye dipstick test (12) that could enable the tests to be carried out under field conditions. It is vitally important that serological tests are validated and standardised if they are to be suitable for correctly identifying infected animals. This means that standard criteria for interpreting the tests might have to be developed for each animal species as well as each laboratory operating the procedure. 245 a) Enzyme-linked immunosorbent assay 246 247 248 249 250 The principle of this technique is that specific antibodies to trypanosomes can be detected by enzymelinked anti-immunoglobulins using solid-phase polystyrene plates coated with soluble antigen. The enzyme may be peroxidase, alkaline phosphatase or any other suitable enzyme. The enzyme conjugate binds to the antigen/antibody complex and then reacts with a suitable substrate to yield a characteristic colour change either of the substrate itself or of an added indicator (the chromogen). OIE Terrestrial Manual 2008 5 Chapter 2.2.18. – Trypanosoma evansi infections (including surra) 251 252 253 254 255 256 257 258 The antigen for coating the plates is derived from the blood of a heavily parasitaemic rat. The trypanosomes are separated on a DEAE-cellulose column and washed three times by centrifugation in cold PSG, pH 8 (PBS with 1% glucose). The final pellet is suspended in cold PSG to a concentration of 3–5%, and briefly ultrasonicated on ice for 30–120 seconds until disintegration of the organisms is complete. This preparation is centrifuged at 4°C and 40,000 g for 60 minutes. The supernatant is diluted in water so as to obtain a protein concentration of 1 mg/ml. The reagent thus obtained can be stored in small aliquots at –70°C for several months. It can also be freeze-dried and stored at –20°C. Various treatments of the antigen preparations have been applied to improve the accuracy of antibody detection (29). 259 Test procedure 260 261 262 i) Dilute or reconstitute the frozen or freeze-dried antigen with freshly prepared 0.01 M carbonate/bicarbonate buffer, pH 9.6. Add 100 µl to each well of a 96-well microtitre plate and incubate overnight at 4°C or for 1 hour at 37°C. 263 264 265 ii) Remove excess antigen by are wash plate with 0.01 M PBS containing 0.05% Tween 20 (PBST). Add test serum dilutions in PBST (100 µl). Include control negative and positive sera. Dilutions must be determined empirically, but are usually between 1/100 and 1/1000. 266 iii) Incubate plates at 37°C for 30 minutes. Eject contents and wash three times with PBST. 267 268 iv) Add a specific peroxidase conjugated species-specific anti-globulin (100 µl) appropriately diluted in PBST (usually between 1/1000 and 1/50,000). 269 v) Incubate the plates at 37°C for 30 minutes, eject contents and wash three times with PBST. 270 271 272 273 274 275 276 277 278 vi) For peroxidase conjugates a number of substrate/chromogen solutions can be used, consisting of hydrogen peroxide with a chromogen, such as tetramethylbenzidene (TMB), 2,2’-azino-bis-(3ethylbenzothiazoline-6-sulphonic acid) (ABTS) and ortho-diphenylenediamine (OPD). A suitable substrate/chromogen solution for peroxidase conjugates is 30% hydrogen peroxide (0.167 ml and 35 mg) in citrate buffer (100 ml), pH 6.0. The citrate buffer is made up as follows: Solution A (36.85 ml): (0.1 M citric acid [21.01 g/litre]); Solution B (65.15 ml): (0.2 M, Na2HPO4 [35.59 g/litre]); and distilled water (100 ml). Dissolve 10 mg TMB in 1 ml dimethyl suphoxide and add to 99 ml of the citrate buffer. A number of these combinations are available commercially in ready-to-use formulations that remain stable at 4°C for up to 1 year. 279 280 vii) Add the substrate chromogen (100 µl) to the plates and incubate at room temperature for 15– 20 minutes. 281 282 283 viii) Stop the reaction by adding 50 µl 1 M sulphuric acid. Read the absorbance of each well at 450 nm for TMB chromogen. Other chromogens may require the use of a different wavelength. All tests should include known high and medium positive control sera, a negative control serum, and a buffer control. 284 285 286 287 288 289 290 291 292 293 294 295 296 297 298 299 A large variety of other test procedures exists. For closely related animal species, cross-reacting reagents may often be used (e.g. anti-bovine immunoglobulin for buffaloes and the use of monospecific anti-IgG conjugates is generally recommended. There are a number of methods that can be used to determine a cut-off point to discriminate between positive and negative results. The simplest method is to base the cutoff on visual inspection of the test results from known positive and negative populations. These results are likely to show some overlap. The operator can choose the most appropriate point to modify the false negative or false positive results depending on the required application of the assay. An alternative is to base the cut-off on the mean +2 standard deviations (SD) or +3 SD values from a large sample of negative animals. Finally, if no suitable negative/positive samples are available a cut-off can be based on the analysis of the data from animals in an endemic situation. If a bimodal distribution separates infected from uninfected animals, then an appropriate value can be selected. The ELISA is likely to correctly identify uninfected animals. Equivocal results can be re-tested using CATT. Currently there are no readily available defined antigens. The Institute for Tropical Medicine in Antwerp has a number of antigens that are still undergoing evaluation. Diagnostic assays incorporating antigens derived from a single VAT should be interpreted with caution and fully evaluated in different hosts and different geographical regions to determine if their performance is consistent throughout the range of T. evansi. 300 b) Card agglutination tests 301 302 303 304 305 306 307 308 309 It is well known that certain predominant variable antigen types (VATs) are expressed in common in different strains of salivarian trypanosomes from different areas. This finding was used as a basis for a test for the diagnosis of T. evansi, the card agglutination test – CATT/T.evansi – was developed at the Laboratory of Serology, Institute of Tropical Medicine, Antwerp. The test makes use of fixed and stained trypanosomes of a defined VAT known as RoTat 1.2. Both variable and invariable surface antigens take part in the agglutination reaction. The CATT is available in kit form from ITM, Antwerp. It consists of lyophilised antigen, PBS, pH 7.4, plastic-coated cards, spatulas, positive and negative control sera and a rotator. The lyophilised antigen can be stored at 2–8°C for up to 1 year. Reconstituted antigen can be stored at 2–8°C for 2 days, but preferably should be used within 8 hours. 6 OIE Terrestrial Manual 2008 Chapter 2.2.18. – Trypanosoma evansi infections (including surra) 310 311 312 313 314 315 For screening, put 25 µl of test serum diluted 1/4 or 1/8 in PBS on to circles inscribed on the plastic cards. Add one drop (45 µl) of the prepared antigen suspension to the diluted serum sample. Mix and spread the reagents with a spatula and rotate the card for 5 minutes using the rotator provided in the kit. Compare the pattern of agglutination with the illustrations of different reactions provided in the kit. Blue granular deposits reveal a positive reaction visible to the naked eye 3. c) Latex agglutination tests 316 317 A kit is available from ITM, Antwerp. It comprises a lyophilised latex suspension coated with T. evansi RoTat 1.2 variable antigens, PBS, positive and negative controls, test cards, plastic spatulas and a rotator. 318 319 Reconstitute the antigen-coated latex particles using distilled, deionised water. Mix gently. Add 20 µl of latex suspension onto each black spot on the test cards. 320 321 Dilute test sera with PBS (1/2 to 1/4) and add 20 µl to the latex suspension. Include appropriate controls. Mix the reagents carefully with a plastic spatula. 322 323 Rotate test cards at 70 rotations/minutes for 5 minutes and view cards under a good light source at the end of the incubation. Positive sera will cause agglutination of the latex particles. C. 324 325 REQUIREMENTS FOR VACCINES AND DIAGNOSTIC BIOLOGICALS No vaccines are available for this disease. REFERENCES 326 327 328 1. BAJYANA SONGA E. & HAMERS R. (1988). A card agglutination test (CATT) for veterinary use based on an early VAT RoTat 1–2 of Trypanosoma evansi. Ann. Soc. Belg. Med. Trop., 68, 233–240. 329 330 331 2. CLAES F., AGBO E.C., RADWANSKA M., TE PAS M.F.W., BALTZ T., DE W AAL D.T., GODDEERIS B.M., CLASSEN E. & BUSCHER P. (2003). How does Trypanosoma equiperdum fit into the Trypanozoon group? A cluster analysis by RAPD and multiplex-endonuclease genotyping approach. Parasitology, 26, 425–431 332 333 3. DARGANTES A.P., REID S.A. & COPEMAN D B. (2005). Experimental Trypanosoma evansi infection in the goat. I Clinical signs and pathology. J. Comp. Pathol., 133, 261–266. 334 335 4. DARGANTES A.P., REID S.A. & COPEMAN D.B. (2005). Experimental Trypanosoma evansi infection in the goat. II Pathology. J. Comp. Pathol., 133, 267–276. 336 337 338 5. DAVISON H.C., THRUSFIELD M.V., MUHARSINI S., HUSEIN A., PARTOUTOMO S., MASAKE R. & LUCKINS A.G. (1999). Evaluation of antigen- and antibody-detection tests for Trypanosoma evansi infections of buffaloes in Indonesia. Epidemiol. Infect., 123, 149–155. 339 340 341 6. FRANKE C.R., GREINER M. & MEHLITZ D. (1994). Investigations on naturally occurring Trypanosoma evansi infections in horses, cattle, dogs and capybaras (Hydrochaeris hydrochaeris) in Pantanal de Pocone (Mato Grosso, Brazil). Acta Trop., 58, 159–169. 342 343 344 7. GUTIERREZ C., CORBERA J.A., JUSTE M.C., DORESTE F. & MORALES I. (2005). An outbreak of abortions and high neonatal mortality associated with Trypanosoma evansi infection in dromedary camels in the Canary Islands. Vet. Parasit., 130, 163–168. 345 8. HOLMSTEDT H. (1965). Simple microcentrifuge for use in the field. Science, 149, 977–978. 346 347 348 9. HOLLAND W.G., CLAES F., MY L.N., THANH N.G., TAM P.T., VERLOO D., BUSCHER P., GODDEERIS B. & VERCRUYSSE J. (2001). A comparative evaluation of parasitological tests and a PCR for Trypanosoma evansi diagnosis in experimentally infected water buffaloes. Vet. Parasit., 97, 23–33. 3 Experimental CATT/T. evansi kits for evaluation purposes are available at the Laboratory of Serology, Institute of Tropical Medicine, Nationalestraat 155, B-2000 Antwerp, Belgium. They are being evaluated in collaboration with the Institute of Molecular Biology of the Free University of Brussels, with financial support from the International Laboratory for Research on Animal Diseases (ILRAD), Nairobi, Kenya. OIE Terrestrial Manual 2008 7 Chapter 2.2.18. – Trypanosoma evansi infections (including surra) 349 350 351 10. HOLLAND W.G., THANH N.G., MY L.N., MAGNUS E., VERLOO D., BUSCHER P., GODDEERIS B. & VERCRUYSSE J. (2002). Evaluation of whole fresh blood and dried blood on filter paper discs in serological tests for Trypanosoma evansi in experimentally infected water buffaloes. Acta Trop., 81, 159–165. 352 353 354 11. INTERNATIONAL ATOMIC ENERGY AGENCY (IAEA) (1993). Improving the diagnosis and control of trypanosomiasis and other vector-borne diseases of African livestock using immunoassay methods. International Atomic Energy Agency, TECDOC, 707, 171 pp. 355 356 357 12. KASHIWAZAKI Y., KANITPUN R., SUTEERAPARP P. & BOONCHIT S. (2000). A preliminary comparative study of a dipstick colloidal dye immunoassay and two antigen-detection ELISAs for diagnosis of Trypanosoma evansi infection in cattle. Vet. Res. Commun., 92, 533–544. 358 359 13. KELLY S. & SCHILLINGER D. (1983). Improved field diagnostic technique for trypanosomiasis by use of a microcentrifuge. Vet. Rec., 113, 219. 360 361 362 14. LEJON V., CLAES F., VERLOO D., MAINA M., URAKAWA T., MAJIWA P.A. & BUSCHER P. (2005). Recombinant RoTat 1.2 variable surface glycoprotein as antigen for diagnosis of Trypanosoma evansi in dromedary camels. Int. J. Parasitol., 35, 455–460. 363 364 15. LOHR K.F., PHOLPARK S., SIRIWAN P., LEESIRIKUL N., SRIKITJAKARN & STAAK C. (1986). Trypanosoma evansi infection in buffaloes in North-East Thailand. II. Abortions. Trop. Anim. Health Prod., 18, 103–108. 365 366 16. MONZON C.M., MANCEBO O.A. & ROUX J.P. (1990). Comparison between 6 parasitological methods for diagnosis of Trypanosoma evansi in the subtropical area of Argentina. Vet. Parasitol., 36, 141–146. 367 368 17. MASIGA D.K. & GIBSON W.C. (1990). Specific probes for Trypanosoma (Trypanozoon) evansi based on kinetoplast DNA minicircles. Mol. Biochem. Parasitol., 40, 279–284. 369 370 371 18. NJIRU Z.K., CONSTANTINE C.C., NDUNG’U J.M., ROBERTSON I., OKAYE S., THOMPSON R.C. & REID S.M. (2004). Detection of Trypanosoma evansi in camels using PCR and CATT/T. evansi tests in Kenya. Vet. Parasitol., 124, 187–199. 372 373 19. ONAH D.N., HOPKINS J.A. & LUCKINS A.G. (1998). Increase in CD5 B cells and depression of immune responses in sheep infected with Trypanosoma evansi. Vet. Immunol. Immunopathol., 63, 209–222. 374 375 376 20. RAE P.F., THRUSFIELD M.V., HIGGINS A., AITKEN C.G.G., JONES T.W. & LUCKINS A.G. (1989). Evaluation of enzyme immunoassays in the diagnosis of camel (Camelus dromedarius) trypanosomiasis: a preliminary investigation. Epidemiol. Infect., 102, 297–307. 377 378 21. REID S.A. & COPEMAN D.B. (2002). Evaluation of an antibody-ELISA using five crude antigen preparations for the diagnosis of Trypansoma evansi infection in cattle. Vet. Parasitol., 104, 79–84. 379 380 22. REID S.A. & COPEMAN D.B. (2003). The development and validation of an antibody-ELISA to detect Trypanosoma evansi infection in cattle in Australia and Papua New Guinea. Prev. Vet. Med., 61, 195–208. 381 382 23. REID S.A., HUSEIN A. & COPEMAN D.B. (2001). Evaluation and improvement of parasitological tests for Trypanosoma evansi infection. Vet. Parasitol., 102, 291–297. 383 384 24. SACHS R. (1984). Improvements in the miniature anion exchange centrifugation technique for detecting trypanosomes in domestic pigs. Trans. R. Soc. Trop. Med. Hyg., 78, 561. 385 386 25. TUNTASUVAN D., CHOMPOOCHAN T., VONGPAKORN M. & MOHKAEW K. (1996). Detection of Trypanosoma evansi antibodies in pigs using an enzyme linked immunosorbent assay. J. Thai Vet. Med. Assoc., 47, 45–53. 387 388 389 26. VENTURA R.M., TAKEDA G.F., SILVA R.A., NUNES V.L., BUCK G.A. & TEIXEIRA M.M. (2002). Genetic relatedness among Trypanosoma evansi stocks by random amplification of polymorphic DNA and evaluation of a synapomorphic DNA fragment for species-specific diagnosis. Int. J. Parasitol., 32, 53–63. 390 391 392 27. VERLOO D., HOLLAND W., MY L.N., THANH N.G., TAM P.T., GODDEERIS B., VERCRUYSSE J. & BUSCHER P. (2000). Comparison of serological tests for Trypanosoma evansi natural infections in water buffaloes from north Vietnam. Vet. Parasitol., 92, 87–96. 393 394 28. VISESHAKUL N. & PANYIM S. (1990). Specific DNA probe for the sensitive detection of Trypanosoma evansi. Southeast Asian J. Trop. Med. Public Health, 21, 21–27. 8 OIE Terrestrial Manual 2008 Chapter 2.2.18. – Trypanosoma evansi infections (including surra) 395 396 29. W UYTS N., CHODESAJJAWATEE N. & PANYIM S. (1994). A simplified and highly sensitive detection of Trypanosoma evansi by DNA amplification. Southeast Asian J. Trop. Med. Public Health, 25, 266–271. 397 398 * * * 399 400 NB: There is an OIE Reference Laboratories for Trypanosoma evansi infections (including surra) (see Table in Part 3 of this Terrestrial Manual or consult the OIE Web site for the most up-to-date list: www.oie.int). 401 OIE Terrestrial Manual 2008 9