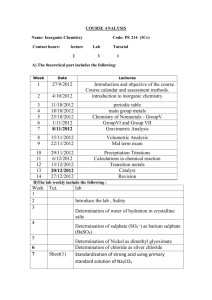
II YR SYLL
advertisement

GIRRAJ GOVT COLLEGE : NIZAMABAD (AUTONOMOUS) DEPARTMENT OF CHEMISTRY B.Sc. IInd YEAR SEMESTER-III PAPER-III Unit-I (Inorganic Chemistry) 1. d-block elements. (9h) Chemistry of elements of First Transition series- electronic configuration, metallic nature, atomic and ionic radii, ionization potential- Oxidation state-relative stability of various oxidation states, ionic and covalent character, acidic and basic nature, oxidizing and reducing nature of various oxidation states, redox potential-Frost and Latimer diagramsstability, disproportionation and comproportionation of different oxidation states. Colourd-d transition, colour and spectral behavior of transition metal ions with respect to d1-d2 configuration. Magnetic behavior-determination of magnetic moment, Gouy’s balance, paramagnetism, diamagnetism. Complexbehaviour stability of complexes-oxidation states, pi complexes. Class-a, class-b and class a/b acceptors. Catalytic properties – important examples. Chemistry of elements of second and third transition series – Comparative treatment with their 3d analogues with respect to oxidation state, magnetic behaviour, spectral properties. Study of Ti, Cr and Cu triads – Titanium triad-electronic configuration, reactivity of +III and +IV states-Oxides, halides. Chromiujm triad-reactivity of +III and +VI states. Copper triad-reactivity of +I, +II, +III states. 2. f-block elements (6h) Chemistry of Lanthanides-electronic structure, position in periodic table, oxidationstate, Atomic and ionic radii, lanthanides contraction – cause and consequences, anomalous behavior of post lanthanides, basicity, Complexation-type of donor ligands preferred. magnetic properties-paramagnetism.Colour and spectra-f-f transition. Occurrence and separation- ion exchange method, solvent extraction. Chemistry of Actinides – General features – electronic configuration, oxidation state, actinide contraction, and colour, magnetic properties and complex formation. Comparison with lanthanides. UNIT -II (Organic Chemistry) 1. Halogen compounds. (5h) Nomenclature and classification of alkyl (into primary, secondary, tertiary), aryl, aralkyl, allyl, vinyl, benzyl). Chemical reactivity – reduction, formation of RMgX, Nucleophylic substitution reaction – Classification into SN1 and SN2, Mechanism of, energy profile diagrams of SN1 and SN2 reactions. Stereochemistry of SN2 (Walden Inversion), SN1 (recemisation) explanation of both by taking the example of optically active alkyl halide – 2-bromo butane. Structure and reactivity – Ease of hydrolysis – comparison of allyl, benzyl, alkyl, vinyland aryl halides. 2. Hydroxy compounds (6h) Nomenclature and classification of hydroxy compounds.Alcohols: Preparation with hydroboration reaction,Grignard synthesis of alcohols.. Aryl carbinols by hydroxy methylation. Phenols – (a) by diazotisation (b)from sulfonic acid (c) from cumene (d) by hydrolysis of halo benzene. Physical properties – Hydrogen bonding (intermolecular and intermolecular) effect of hydrogen bonding on boiling point and water solubility. Chemical properties (a) acidic nature of Phenols (b) formation of alkoxides/phenoxides and their reaction with RX (c) replacement of OH by X using PCl5, PBr3, SOCl2 and with HX/ZnCl2.Esterification by (a) acid halides, anhydrides and acids (mechanism) (b) Esters of inorganic acids (c) dehydration of alcohols. Oxidation of alcohols by CrO3, KMnO4. Special reactions of Phenols – (a) Bromination, (b) Kolbe – Schmidt reaction (c) Riemer Tiemann (d) Azo coupling. Identification of alcohols by oxidation – KMnO4, Ceric ammonium nitrate – Lucas reagent; Phenols by reaction with FeCl3 and by the solubility in NaOH. Poly hydroxyl compounds –Pinacol –pinacolone rearrangement, Oxidative cleavage (Pb (OAc)4 &HIO4). 3. Synthesis based on carbanions. (4h) Acidity of α-Hydrogens, structure of carbanions. Preparation of Aceto acetic ester by Claisen condensation and synthetic application of Aceto acetic ester. (a) Acid hydrolysis and ketonic hydrolysis. Preparation of (i) monocarboxylic acids (ii) dicarboxylic acids (b) malonic ester – synthetic applications. Preparations of (i) substituted mono carboxylic acids(propionic acid and n-butyric acid) (ii) substituted dicarboxylic acids.(succinic acid and adipic acid (iii) α,β-unsaturated acids ( crotonic acid). Reaction with Urea. Unit –III (Physical Chemistry) 1. Phase rule (5h) Statement and meaning of the terms – Phase, Component and degrees of freedom, Gibb’s Phase rule, phase equilibria of one component system – water system. Phase equilibria of two component system – Solid-liquid equilibria, simple eutectic – Pb-Ag system, desilverization of lead. Solid solutions – compound with congruent melting point (MgZn) system and incongruent melting point – (NaCl-H2O) system. Freezing mixtures. 2. Dilute solutions & Colligative Properties. (10h) Dilute solutions and Colligative properties, ideal and non-ideal solutions. Raoult’s law, relative lowering vapour pressure, molecular weight determination. Osmosis laws of osmotic pressure. Elevation of boiling point and depression of freezing point. Derivation of relation between molecular weight and elevation in boiling point and depression in freezing point. Experimental methods for determining various colligative properties. Abnormal molar mass, Van’t Hoff factor, degree of dissociation and association of solutes. Unit –IV (General Chemistry) 1..Moleculr Symmetry (5h) Symmetry elements and symmetry operations in molecules. Rotational axis of symmetry and types of rotational axis. Plane of symmetry and types of planes. Improper rotational axis of symmetry. Inversion center. Identity element. Symmetry operations of a molecule form a group. Flow chart for the identification of molecular point group. 2 Theory of quantitative analysis (6h) Principles of volumetric analysis: Introduction, standard solution, indicators, end point, titration error, and titration curves. Types of titrations- (i) Neutralization titration – Principle, titration curves and selection of indicators-strong acid-strong base, strong acidweak base, weak acid –strong base, weak acid-weak base, pH indicators (ii) Redoxtitrations-Principles, detection of endpoint, redox indicators. (iii) Precipitation titrations-Principle, detection of end point, indicators, and titration curves. (iv) Complexation titrations – Principle, metal ion indicators. Principles of Gravimetric analysis- Nucleation, precipitation, growth of precipitate, filtration and washing, drying and ignition of precipitate. – Co-precipitation and post-precipitation.Requirements of gravimetryic analysis. G-3.Evaluation of analytical data (4h) Accuracy & precision and their methods of expression. Errors – classification of errors. Determinate and Indeterminate errors, and relative error propagation of errors. Significant figures and their importance. GIRRAJ GOVT COLLEGE : NIZAMABAD (AUTONOMOUS) DEPARTMENT OF CHEMISTRY B.Sc. IInd YEAR SEMESTER-III PAPER-III Unit-I (Inorganic Chemistry) 1. Theories of bonding in metals and metallurgy. (9h) Theories of bonding in metals – Free electron theory – thermal and electrical conductivity of metals, drawbacks. Valance bond theory – explanation of metallic properties and its limitations. Band theory- explanation of metallic properties, conductors, semiconductors and insulators. General methods involved in extraction of metals – minerals and ores, ore concentration – electromagnetic separation, gravity separation – wilfley tables, hydraulic classifier, leaching froth flotation, calcinations and roasting. Acid and alkali digestion. Reduction of oxides, carbonates, halides, sulphides, sulphates – smelting, flux, auto reduction, alumino thermic reduction, hydrometallurgy, elelctrolytic reduction. Purification of impure metals- liquation, fractional distillation, zonerefining, oxidative processes – Cupellation, bessemerisation, puddling, poling, thermal decomposition, Amalgamation, Electrolysis. 2. Metal carbonyls and related compounds. (6h) EAN rule, classification of metal carbonyls, structures and shapes of metal carbonyls of V, Cr, Mn, Fe, Co and Ni. Metal nitrosyls and metallocenes (only ferrocene). Unit-II (Organic Chemistry) 1.Carbonyl compounds (9h) Nomenclature of aliphatic and aromatic carbonyl compounds and isomerism. Synthesis of aldehydes and ketones from acid chlorides by using 1,3-dithianes, nitriles, and from carboxylic acids. Special methods of preparing aromatic aldehydes and by (a) Oxidation of arenas (b) Hydrolysis of benzal halides physical properties – absence of hydrogen bonding. Keto-enol tautomerism, polarisability of carbonyl groups, reactivity of the carbonyl groups in aldehydes and ketones. Chemical reactivity- I. Addition of (a) NaHSO3 (b) HCN (c) RMgX (d) NH3 (e) RNH2 (f) NH2OH (g) PhNHNH2 (h) 2,4DNP Schiff bases, Addition of H2O to form hydrate (unstable), comparison with chloral hydrate (stable), addition of alcohols-hemiacetal and acetal formation, Halogenation using PCl5 with mechanism. Base catalyzed reactions – with particular emphasis on aldol, cannizaroreaction, perkinreaction, Benzion condensation, haloform reaction, Knoevengeal condensation. Oxidation rections-KMnO4 oxidation and auto- reductioncatalytic hydrogenation, clemenson’s reductionWolf-kishner reduction MPV reduction, reduction with LiAlH4, NaBH4.Analysis- 2,4-DNP test, Tollens test, Fehling test, Schiff’s test, haloform test (with equations). 2.Carboxylic acids and derivatives. (6h) Nomenclature, classification and methods of preparation a) Hydrolysis of Nitrites, amides and esters. B) Carbonation of Grignard reagents. Special methods of preparation of aromatic acids. Oxidation of side chain. Hydrolysis of benzotrichlorides. Kolbe reaction. Physical Properties – hydrogen bonding, dimeric association, acidity-strength of acids with the examples of trimethyl acetic acid and trichloro acetic acid, Relative differences in the acidity of aromatic and aliphatic acids. Chemical properties – Reactions involving H, OH, and COOH groups-salt formation, anhydride formation, acid halide formation, Esterification (mechanism) & amide formation. Reduction of acid to the corresponding primary alcohol-via ester or acid chlorides. Degradation of carboxylic acids by Hunsdiecker reaction, Schmidt reaction (Decarboxylation). Arndt-Eister synthesis, Halogenation by Hell-Volhard-Zelensky reaction. Carbolxylic acid Derivatives-reactions of acid halides, Acid anhydrides, acid amides and ester (Mechanism of ester hydrolysis by base and acid). Unit- III (Physical Chemistry) 1.Electrochemistry (15h) Electrical transport-conduction in metals and in electrolyte solutions, specific conductance and equivalent conductance, measurement of equivalent conductance, variation of specific and equivalent conductance with dilution. Migration of ions and Kholrausch’s law, Arrhenius theory of electrolyte dissociation and its limitations, weak and strong electrolytes, Ostwald’s dilution law, its uses and limitations. Debye-HuckleOnsagar’equation for strong electrolytes (elementary treatment only). Transport number, definition and determination by Hittorf method for attackable electrodes. Applications of conductivity measurements: Determination of degree of dissociation, determination of Ka of acids, determination of solubility product of sparingly soluble salt, conductrometric titrations. Types of reversible electrodes: gas-metal ion, metal-metal ion, metal-insoluble salt-anion and redox electrodes. Electrode reactions, Nernst equation, cell EMF and single electrode potential, standard Hydrogen electrode-reference electrodes-standard electrode potential, sign convention, electrochemical series and its significance. Electrolytic and galvanic cells-reversible and irreversible cells, conventional representation of electrochemical cells. EMF of a cell and its measurement. Computation of EMF. Applications of EMF measurements. Calculation of thermodynamic quantities of cell reaction (∆G, ∆H and K). Determination of pH using quinhydrone elelctrode. Solubility product of AgCl. Potentiometric titrations. Unit-IV (General Chemistry) 1. Introducatory treatment to : Pericyclic Reactions (5h) Concerted reactions, Molecular orbitals, symmetry properties HOMO, LUMO, Thermal and photochemical pericyclic reactions. Types of pericyclic reactionsElectrolytic,cycloaddition and sigmatropic ractions-one example each. Synthetic strategies (5h) Terminology-Disconnection (dix), Symbol ( ), synthon, synthetic equivalence (SE), Functional group interconcersion (FGI), linear, Convergent and Combinatorial, Target molecule ™ Retrosynthesis of the following molecules i) acetophenone ii)cyclohexene iii) phenyleathylbromide Asymmetric (Chiral) synthesis (5h) Definition, eneatiometric excess, diastereometric excess, stereo specific reactiondefinition-example-dehalogenation of 1, 2-dibromoides by I-, stereo selective reactionsdefinition-example-acid catalysed dehydration of 1-phenylproponal. GIRRAJ GOVT COLLEGE : NIZAMABAD (AUTONOMOUS) DEPARTMENT OF CHEMISTRY B.Sc. IInd YEAR Practical Syllabus I. Estimate the amount in the given following experiments. 1. 2. 3. 4. 5. 6. 7. 8. Determination of carbonate and bicarbonate in a mixture. Determination of Fe(II) using K2Cr2O7 Determination of Fe(II) usingKMnO4 with oxalic acid as primary standard Determination of Cu(II) using Na2S2O3 with K2Cr2O7as primary standard.. etermination of Zn(II) using EDTA Determination of Ni(II) using EDTA Determination of Hardness of water. Determination of Zn(II) by ferrocyanide II. Gravimetric analysis: 1. Determination of Nickel as Ni-DMG 2. Determination of Lead as Lead Chromate. 3. Determination of Magnesium as magnesium pyrophosphate