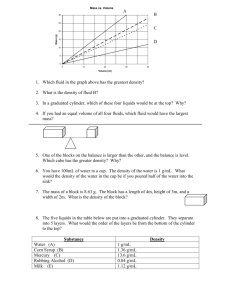
Packet 2 Density - Holy Family Catholic Schools
advertisement

Chemistry Name:__________________ Period:____ Chemistry Skills Packet 2 Day In Class Work 1 2 3 4 5 6 7 8 Sig Figs and Uncertainty Density Density of Liquids A Penny for your Thoughts Lab Density Freezing of PDCB Graphing Technique Density Practicum Outcomes Given two of the three variables in the density formula the student will calculate the third. Given the experiment and accepted values for density of an object the student will calculate the percent error. Given an experimental situation the student will determine the independent and dependent variable and two other factors that must be held constant. Given data from an experiment the student will graph the data. All five parts of the graph must be labeled. Students will perform a laboratory test including knowledge from Packet 1&2. 1 SIGNIFICANT FIGURES A. When taking measurements all certain digits plus the first uncertain number are significant. Example: Your bathroom scale weighs in 10 Newton increments and when you step onto it, the pointer stops between 550 and 560. Your look at the scale and determine your weight to 557 N. You are certain of the first two places, 55, but not the last place 7. The last place is a guess and if it is your best guess it also is significant. B. When given measurements, the numbers that are significant are the digits 1-9 and the 0 when it is not merely a place holder. 1. When 0’s are between sig. fig., 0’s are always significant. Example: 101 has 3 sig. fig. and 34055 has 5 sig. fig. 2. When the measurement is a whole number ending with 0’s, the 0’s are never significant. Example: 210 has 2 sig. fig. and 71,000,000 also has 2 sig. fig. 3. When the measurement is less than a whole number, the 0’s between the decimal and other significant numbers are never significant (they are place holders). Example: .0021 has 2 sig. fig. and .0000332 has 3 sig. fig. 4. When the measurement is less than a whole number and the 0’s fall after the other significant numbers, the 0’s are always significant. Example: .310 has 3 sig. fig. and .3400 has 4 sig. fig. 5. When the measurement is less than a whole and there is a 0 to the left of the decimal, the 0 is not significant. Example: 0.02 has only 1 sig. fig. and 0.110 has 3 sig. fig. 6. When the measurement is a whole number but ends with 0’s to the right of the decimal, the 0’s are significant. Example: 20.0 has 3 sig. fig., 18876.000 has 8 sig. fig. In case 4 and 6 the 0’s have no effect on the value (size) of the measurement. Therefore, these 0’s must have been included for another reason and that reason is to show precision of the measurement. Since these 0’s show precision they must therefore be significant. In cases 2 and 3 removal of the 0’s DO change the value (size) of the measurement, the 0’s are place holders and are thus not significant. In case 5 the 0 is completely unnecessary, it is neither a place holder nor adds to the accuracy of the measurement. 2 UNCERTAINTIES IN CALCULATIONS 1. When adding or subtracting numbers written with the notation, always add the uncertainties and then round off the value to the largest significant digit. Round off the answer to match. Example: (22.4 .5) + (14.76 .25) = 37.16 .75 = 37.2 .8 The uncertainty begins in the tenths place… it is the last significant digit. 2. When adding or subtracting numbers written in significant figures, show the uncertainty by rounding the answer to match the largest place with uncertainty. Example: 267 + 11.8 = 278.8 = 279 The least accurate original measurement is only accurate to the ones place. 3. When multiplying or dividing with numbers written with , do a base calculation and a maximum calculation. Find the difference between the base calculation and the maximum calculation to give you your calculated value. Round the calculated value to the largest significant digit and the answer to match the value. Example: (20.4 .6) x (17.70 .25) base calculation (20.4 x 17.70 = 361.08) maximum calculation, add the value ----- (21.0 x 17.95 = 376.95) difference = 15.87, initial answer = 361.08 15.87 final answer = 360 20. The tens place is the first uncertain digit and therefore the last significant figure. 4. When multiplying or dividing measurements written in significant figures, show the uncertainty of your calculations by rounding off your answer to match the same number of significant figures as your least precise measurement (the measurement with the least number of significant figures). Example: 477.85 32.6 = 14.657975 = 14.7 32.6 is the least accurate measurement with only 3 significant figures. NOTE: There are two types of precision: “absolute precision” and “relative precision.” Example: 322.45 x 12.75 x 3.92 = 16116.051 = 16100 All the measurements are accurate to the hundredth place (absolute precision) but the answer is rounded to 3 significant figures because 3.92 has only 3 significant figures (relative precision). In Summary: Adding and Subtracting Multiplying and dividing #’s with notation Rule 1 Rule 3 #’s with significant figures Rule 2 Rule 4 3 SIGNIFICANT FIGURES & ROUNDING A. Indicate the number of significant figures then round each to the number of significant figures indicated. For example: 1.234 has ______4___ significant figures and, rounded to 2 significant figures, is ___1.2____ 1. 0.6034 has __________ significant figures and, rounded to 2 significant figures, is __________ 2. 12,700 has __________ significant figures and, rounded to 2 significant figures, is __________ 3. 12,700.00 has __________ significant figures and, rounded to 1 significant figures, is __________ 4. 0.000983 has __________ significant figures and, rounded to 2 significant figures, is __________ 5. 123342.9 has __________ significant figures and, rounded to 5 significant figures, is __________ 6. 6.023 x 1023has __________ significant figures and, rounded to 2 significant figures, is __________ 7. .005600 has __________ significant figures and, rounded to 1 significant figures, is __________ 8. 10000.5006 has __________ significant figures and, rounded to 5 significant figures, is __________ 9. 2.0 x 10-3 has __________ significant figures and, rounded to 1 significant figures, is __________ 10. 3.456110 has __________ significant figures and, rounded to 3 significant figures, is __________ B. Given calculations with the calculator answer, write the answers with the appropriate number of significant figures. Example: 6.00 x 3.00 = 18 The answer should be ______18.0_____ 1. 23 + 46 = 69 The answer should be _______________ 2. 23.0 + 46.0 = 69 The answer should be _______________ 3. 253 + 345.8 = 598.8 The answer should be _______________ 4. 56 – 35 = 21 The answer should be _______________ 5. 56.00 – 35.0 = 21 The answer should be _______________ 6. 46 x 12 = 552 The answer should be _______________ 7. 3.24 x 5.63 = 18.2412 The answer should be _______________ (2.355 + 2.645) x 10.00 = 50 The answer should be _______________ 8 Use the rules for significant figures to report uncertainty for the following: Answer before rounding Final Answer 4 Example 134.8 + 27.67 = 162.47 162.5 16. 109.5 + 24.02 = ______________ _______________ 17. 66.341 + 902.10 = ______________ _______________ 18. 88.001 – 56.7 = ______________ _______________ 19. 5696.10 – 34.0010 = ______________ _______________ 20. 32.1 + 354.98 – 224.775 = ______________ _______________ 21. 412.667 + 22.11 + 15.6 = ______________ _______________ 22. 5286.445 – 26.0005 – 13.112 = ______________ _______________ 23. 3.468 – 2.4 + 13.67 – 117.11119 = ______________ _______________ Example 34.8 x 27.67 = Answer before rounding Final Answer 962.916 963 24. 109.5 x 24.02 = ______________ _______________ 25. 66.341 x 902.10 = ______________ _______________ 26. 88.001 56.7 = ______________ _______________ 27. 5696.10 x 34.0010 = ______________ _______________ 28. 32.1 x 354.98 224.775 = ______________ _______________ 29. 1.68 x 2.45 5.912 = ______________ _______________ 30. 432.721 13.2467 21.44 = ______________ _______________ 5 Density = mass / volume 1. Define mass? 2. Define volume? 3. Define density and show the formula for calculating density. 4. Why does changing the shape of an object have no effect on the density of that object? 5. Aluminum is used to make airplanes. Cast iron is used to make weightlifting equipment. Explain why the densities of these metals make them useful for these purposes? 6. What is the density of water? Remember for water 1g=1ml=1cm3 7. Why does an air bubble rise to the surface of a glass of water? 8. Calculate the densities of the following objects. Remember to place units after each number. Object A length = 6cm width = 3cm height = 1cm mass = 36g volume = _____ density = _____ Object B length = 10cm width = 5cm height = 2cm mass = 300g volume = _____ density = _____ Object C Use the water displacement method to determine the density of object C (silly putty). initial water level in graduated cylinder = 25ml final water level after placing silly putty into graduated cylinder = 29ml mass of silly putty=8g volume = _____ density = _____ 9. Which of the following materials will float on water (density 1 g/ml)? air = .001 g/cm3 corn oil = .93 g/ cm3 glycerine = 1.26 g/ cm3 corn syrup = 1.38 g/ cm3 wood = .85 g/ cm3 steel = 7.81 g/ cm3 rubber = 1.34 g/ cm3 ice = .92 g/c cm3 water = 1.00 g/ cm3 10. Assuming the materials don’t mix, show how the materials would "stack up" in a graduated cylinder. 6 Measure the Density of a Liquid **Note: Use a sheet of vinyl to protect the table from the chemicals in this expereiment. Procedure 1. Determine the mass of a clean dry 10ml graduated cylinder. Record its mass. 2. Add 10.0ml of distilled water to the graduated cylinder. Determine the mass of the water and record it. 3. Discard the water. Clean the cylinder. 4. Add 10.0ml of ethanol to the cylinder. Determine the mass of the ethanol and record it. 5. Place the ethanol back into its original bottle. Clean the cylinder. 6. Add 10.0ml of ethylene glycol to the cylinder. Determine the mass of the ethanol and record it. 7. Place the ethylene glycol back into the bottle. 8. Clean the cylinder. Data & Calculation Table Liquids Water Ethanol Ethylene Glycol Mass of Liquids Volume of Liquids Percent Error = Experimental Value – Accepted Value Experimental Density x 100 Accepted Value *Accepted density values for the three liquids can be found in Handbook if Chemistry and Physics. Liquids Water Ethanol Ethylene Glycol Experimental Density Accepted Density Percent Error 1. Working in the laboratory, a student find the density of a piece of pure aluminum to be 2.85 g/cm3. The accepted value for the density of aluminum is 2.699 g/cm3. What is the student's percent error? 2. A student experimentally determines the specific heat of water to be 4.29 J/g x Co. He then looks up the specific heat of water on a reference table and finds that is is 4.18 J/g x Co. What is his percent error? 3. A student takes an object with an accepted mass of 200.00 grams and masses it on his own balance. He records the mass of the object as 196.5 g. What is his percent error? 7 Density Worksheet Density is the ratio of the mass of the substance to the volume of the substance at agiven temperature. Density has units of g/ cm3 or g/c.c. or g/mL for liquids and solids, and g/L for gases. 1. A gold-colored ring has a mass of 18.9 grams and a volume of 1.12 mL. Is the ring pure gold? (The density of gold is 19.3 g/mL.) 2. What volume would a 0.871 gram sample of air occupy if the density of air is 1.29 g/L? 3. Pumice is volcanic rock that contains many trapped air bubbles. A 225 gram sample occupied 236.6 mL. What is the density of pumice? (Answer is 0.951 g/mL) Will pumice float on water? The density of water is 1.0 g/mL.) 4. A cup of sugar has a volume of 237 mL. What is the mass of the cup of sugar if the density is 1.59 g/mL? (Ans. is 377 grams) 5. Which has the greater mass, 1 liter of water or l liter of gasoline? The density of water is 1.00 g/mL and that of gasoline is appoximately 0.68 g/mL. 6. A crumpet recipe calls for 175 grams of flour. According to Julia Child's data, the density of flour is 0.620 g/mL. How many mL of flour are needed for this recipe? (Ans. is 282 mL) 7. From their density values, decide whether each of the following substances will sink or float when placed in sea water, which has a density of 1.025 g/mL. Gasoline 0.66 g/mL Asphalt l.2 g/mL Mercury 13.6 g/mL Cork 0.26 g/mL 8. Mercury is a liquid metal having a density of 13.6 g/mL. What is the volume of 1.00 lb of mercury metal? (33.4 mL) 2.2lb=1kg 9. A sample of lead is found to have a mass of 32.6 g. A graduated cylinder contains 2.8 mL of water. After the lead sample is added to the cylinder the water level reads 5.7 mL. Calculate the density of the lead sample. (11g/mL) 10. A piece of magnesium is in the shape of a cylinder with a height of 5.62 cm and a diameter of 1.34 cm. If the magnesium sample has a mass of 14.1 g, what is the density of the sample? (1.78 g/mL) V = hr2п A Penny for your Thoughts 8 Penny for Your Thoughts 1. What do you think is the chemical composition of pennies? 2. Have pennies changed over time? Inspect the collection of pennies provided by the instructor (you may augment our collection with some of your own pennies if you care to make a contribution). Consider the following hypotheses: a. Perhaps, because of wear, the older pennies are lighter that the newer ones. b. Possibility the pennies have changed in chemical composition over the years. 3. Devise an experiment to test the first hypothesis. You have access to an electronic scale that can measure the weight of a single penny to a precision of 0.01 gram. Devise and execute a data collection scheme to present your findings. Organize your group to divide the labor. Use as many pennies that you feel is necessary to come to some conclusion. If you need graph paper, ask for it. 4. Before beginning the measurements, make your own personal prediction as to what you will find: 5. Write the procedure that your group developed, the results that you obtained, and the conclusions your reached. 6. Where there any surprises? Speculate on the origin of any discrepancies that you observed. . 9 7. Why would differences in chemical composition of coins produce differences in mass? In other words, what property of the metals that makes up coins might explain why the mass of modern pennies (after 1983) would be less than older pennies, when their sizes (volumes) seem to have always been the same? 8. Assume, for simplicity, that new pennies are 100% zinc (actually they are 97.5% zinc, which is pretty close) and that old pennies are 100% copper. What is the density of pure copper and pure zinc? 9. You have no doubt worked with density and its measurement in previous schooling. Using common laboratory equipment, how could you measure the density of a coin experimentally? Of any irregularly shaped object? 10. Measure the average density of 5 old pennies and 5 new pennies as accurately as possible, using the equipment in the lab. Report your measurements and calculations below. 11. What is the percent error of your findings? New Penny = 7.13g/ml Old Penny = 8.96g/ml 12. Which measurement, mass or volume, is most difficult to make precisely and accurately? Why? 13. When measuring density, does it make any difference how accurately the volume is measured, as long as the mass is measured accurately enough? Explain. 14. Describe some ways of doing the experiment that would increase the accuracy of the measurement of the average density of coins. 10 Density (SHOW ALL WORK) 1. A 13.2 mL rock weighs 47.6 g. Determine its density. 2. 138.42 g of salt water has a volume of 117.0 mL. Determine its density. 3. 0.446 g of hydrogen gas fills a 5.0 L bag. Determine hydrogen's density. 4. 25.2 mL of water are placed in a graduated cylinder. A 22.6 g stone is dropped in, and the water level rises to 32.4 mL. Find the stone's density. Substance: Density: osmium 22.6 g/mL gold 19.3 g/mL mercury 13.6 g/mL lead 11.4 g/mL copper 8.96 g/mL aluminum 2.70 g/mL water 1.00 g/mL alcohol 0.781 g/mL styrofoam 0.145 g/mL air 1.28 g/L helium 0.179 g/L ________________________ Volume Equations block: V=lwh cylinder: V=πr2h sphere: V=4πr3 5. A graduated cylinder is placed on an electronic balance, and the scale reads 78.32 g. 10.0 mL of glycerine are added, and the scale reads 91.78 g. What is the density of glycerine? 6. A 3.0 cm x 4.5 cm x 6.7 cm brick as a mass of 985 g. a> What is its density, and...b> from what material is it most likely made? 7. A cylinder has a mass of 528.6 g, a length of 14.2 cm, and a diameter of 2.30 cm. Of what is the cylinder most likely made of? 8. A ball has a mass of 753 g and a radius of 5.62 cm. a> Will the ball float or sink in water? b> Will it float or sink in salt water? 9. a> How much would a 15.2 mL chunk of styrofoam weigh? b> How much would the same size chunk of osmium weigh? 10. How much would a 15.9 cm x 11.6 cm x 7.3 cm block of aluminum weigh? 11. 16. A rectangular piece of aluminum foil measures 13.72 cm x 8.63 cm and has a mass of 3.1 g. Find how thick it is. (remember V = l•w•h) 11 Freezing of PDCB In this experiment, you will examine the cooling behavior of the pure substance, paradichlorobenzene, otherwise known as PDCB but best known as Moth Nuggets. By measuring the temperature of PDCB as it goes from a LIQUID state to a SOLID state, you will discover a property of all materials as they pass through a phase change. PROCEDURE: 1. Obtain a test tube filled with Paradichlorobenzene (PDCB) with a thermometer in it. Heat the PDCB by placing in into a hot water bath to a temperature of 70° C. If the water begins to boil shut of the Bunsen burner. 2. Using tap water, prepare 100 ml 30-35 °C in a 250ml beaker. Keep a thermometer in the beaker. 3. Remove the test tube with the PDCB from the hot water bath and immediately place it in the 250ml beaker once the PDCB is melted. Immediately begin recording the temperature of the water in the beaker and the PDCB every 30 seconds until the two temperatures are within 5 degrees of each other. Be sure to record any visual observations you may notice, such as when crystallization first begins and ends. 4. Return the test tube with PDCB and thermometer back to its container. Time Temp (min) PDCB ° C Temp Water ° C Time Temp (min) PDCB ° C Temp Water ° C Time Temp (min) PDCB ° C 0.0 6.5 13.0 0.5 7.0 13.5 1.0 7.5 14.0 1.5 8.0 14.5 2.0 8.5 15.0 2.5 9.0 15.5 3.0 9.5 16.0 3.5 10.0 16.5 4.0 10.5 17.0 4.5 11.0 17.5 5.0 11.5 18.0 5.5 12.0 18.5 6.0 12.5 19.0 12 Temp Water ° C PROCESSING THE DATA: 1. Construct a graph of Temperature vs. Time for both PDCB and Water. 2. Assuming that you have good data that extended from when the PDCB was totally liquid to when it was totally solid, your graph of the PDCB should clearly show its freezing temperature. It is the temperature at which the curve plateaus (levels) out. What was the freezing temperature of the PDCB? 3. If the temperature of a material is related to the average kinetic energy of the molecules in the material, is the PDCB losing kinetic energy as it changes from a liquid to solid at its freezing temperature? Explain. 4. Thus, if the PDCB is constantly losing energy as it goes from a liquid to a solid and it is not kinetic energy, what form of energy can it be? 5. On a piece of graph paper, graph the data below if you performed a similar experiment to with a substance that went from a gaseous state to a frozen state. Remember to place and label the independent and dependent variable. Time (min) Temp (°C) Time (min) Temp (°C) Time (min) Temp (°C) 0.5 99.0 5.5 61.0 10.5 36.0 1.0 98.2 6.0 60.3 11.0 36.0 1.5 97.4 6.5 60.3 11.5 36.0 2.0 93.0 7.0 60.3 12.0 36.0 2.5 91.5 7.5 60.0 12.5 33.9 3.0 86.0 8.0 55.6 13.0 31.1 3.5 82.1 8.5 52.0 13.5 28.3 4.0 74.5 9.0 48.0 14.0 25.5 4.5 69.9 9.5 42.6 14.5 23.6 5.0 65.0 10.0 36.7 15.0 22.1 Divide the graph into 5 sections and describe what is happening in each section. Section 1Section 2Section 3Section 4Section 5- 13 Packet 2 Density 1. If a substance contracts when it freezes, its A. B. C. D. density will remain the same. density will increase. density will decrease. change in density cannot be predicted 2. A graduated cylinder contains 44.2 mL of water. A 48.6-g piece of metal is carefully dropped into the cylinder. When the metal is completely covered with water, the water rises to the 51.3-mL mark. What is the density of the metal? Use the atomic windows below to answer Questions 3 and 4. The atomic windows represent particles of the same gas occupying the same volume at the same temperature. The systems differ only in the number of gas particles per unit volume. 3. List the windows in order of decreasing density. 4. Compare the density of the gas in window (a) to the density of the gas in window (b). 5. What equation is used to determine the density of an object? 6. Would the density of a person be the same on the surface of Earth and on the surface of the moon? Explain. 3 7. A shiny, gold-colored bar of metal weighing 57.3 g has a volume of 4.7 cm . Is the bar of metal pure gold? The density of gold is 19.3 g/cm3? 8. The density of dry air measured at 25°C is 1.19 × 10−3 g/cm3. What is the volume of 50.0 g of air? 9. A flask that can hold 158 g of water at 4°C can hold only 127 g of ethanol at the same temperature. What is the density of ethanol? 10. Graph the following data: 14 Practice Graphing Graph the following sets of data on graph paper. Label the independent and dependent variables and their units. Graph 1 River Velocity (cm/sec) 25 50 75 100 125 150 175 200 225 Particle Diameter (cm) .0004 .004 .07 .09 .3 .09 3.0 5.0 6.5 Graph 2 Temperature °C 10 20 30 40 50 60 70 80 90 Amount of bacteria (g) 8.8 18.2 37.0 60.2 72.5 43.4 27.4 15.3 4.8 The volume of a liquid is measured to be 18.5 ml and the mass is 2.66g. What is the density of the liquid? What is the mass of a liquid if the density if 1.54g/ml and the volume is 2.2 ml? The experimental density of a liquid is o.98g/ml and the accepted value is 2.02g/ml. What is the percent error? 15 In this lab the density of and unknown metal object, marble and block of wood will be determined in Part A. The density of 2 unknown liquids will be determined in Part B. It is known that an objects density is a ratio of its mass and volume. Be sure to use the correct number of sig. figs. and units. Density of a solid. Box_______ Metal Marble Wood mass: mass: mass: volume: volume: volume: Density: Density: Density: Density of a liquid. Liquid 1 Liquid 2 mass: mass: volume: volume: Density: Density: Clean-up: dry solid A and return B to original containers. Questions Which part, A or B, do you feel more error was involves? Why? Suppose on part B, a student read the top of the meniscus. Would this cause the calculated value of density to com out too higher, too lower, or have no effect? Suppose on Part A, there was a rock stuck in the bottom of the graduated cylinder the entire lab. Would this cause the calculated value for density to come out too high, too low, or no effect? Describe what each object in Part A would do if it was placed in a container of water. Why? Explain why an ice cube is less dense than water even though they are made of the same material. 16 17