Element Brochures - White River High School
advertisement

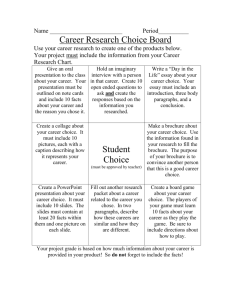
Element Brochures CHARLES BEYL by Daryl Kuhn hen I introduce the periodic table to my seventh graders, I want them to learn how to use this valuable resource, but I don’t expect them to memorize the entire chart. To help familiarize my students with the information available from the table, I have them create element brochures. This activity teaches students how to navigate the periodic table and gives them a chance to sharpen their research skills. The nature of this activity allows students of all ability levels to achieve spectacular results. W Teacher preparation Make a two-sided photocopy of the brochure page that accompanies this article for each student. Write the names of different elements, one for each member of your class, S C I E N C E on small slips of paper and put them in a fishbowl. Before you begin the activity, explain how to use the periodic table to determine an element’s symbol, atomic number and weight, physical state at room temperature, and group name (metal, nonmetal, metalloid, or noble gas). As students enter the room, hand them a folded instructional brochure and a folded blank brochure to work on and ask them to select an element from the fishbowl. The brochure explains in detail what information they will need to research and where to place that information on the brochure. After students read the brochure, answer questions that they may have about the project. Then, take your class to the library to give them time to research their S C O P E 22 O C T O B E R elements. Students are only allowed to conduct research at this time— brochure construction is done at home. When the brochures are complete, create a scavenger hunt based on the information that students gathered (see Figure 1 for a sample based on the elements I assign my students). The scavenger hunt encourages students to review their classmates’ brochures and, in the process, learn about the other elements. Display the brochures around the room to show off your students’ creativity and to give everyone easy access to each other’s brochures. Daryl Kuhn is a science teacher at Edmunds Middle School in Burlington, Vermont. 1 9 9 6 Chemistry Assignment On the back cover, include • Your name and the assignment due date • The name of your printer/publisher (use your imagination) For this assignment, you will be creating a brochure about an element that is assigned to you. Your element is __________________. • Is the brochure neat and easy to read? • Is all the required information there? • Are spelling and grammar correct? • Is the brochure colorful? Will the reader want to pick up your brochure and read it? • Can you add any finishing touches to your brochure to improve it? Use your textbook and resources from the library to locate information about your element. Refer to this sample brochure for the information you need to include. Set up your brochure in the format described. The assignment is worth three grades, so include all the required information and do your best! Your brochure should be fun and interesting to read. The assignment is due ___________ fold line fold line The cover should include • The element name • The element symbol 23 Remember to check over your brochure after you have finished it. Check for the following: O C T O B E R 1 9 9 6 ELEMENT BROCHURE S C O P E • Describe the uses of your element. • Draw or provide a color picture of it. (You may need to be creative here. Show a picture of its use if you cannot draw how the element occurs in nature.) S C I E N C E This page should include introductory information: On the middle and right-hand pages, include information about your element: On this page, include • An electron shell diagram. • The number of electrons are in each shell around the nucleus. S C I E N C E • The atomic mass • The atomic number • Who discovered the element • When it was discovered • How it was named • What its natural state is • Its melting point • Its boiling point • The group it belongs to in the periodic table (noble gas, metal, and so forth) • Some of its characteristics, such as color, odor, whether it’s dangerous—explosive, radioactive, or noxious • Any interesting facts about the element S C O P E 24 O C T O B E R Remember to check all your facts. Is there anything you can add to improve your brochure? Be creative . . . Have fun! 1 9 9 6 fold line fold line Element scavenger hunt Name _____________________ Period _____ Use the element brochures around the room to identify each element described. Answers may be used more than once. I am an element that . . . 1. has 27 electrons in its orbital diagram. 2. is used in swimming pools to keep out bacteria. 3. is used in incandescent lamps. 4. is used in aircraft gas turbine engines. 5. was discovered by Charles Wood in South America. 6. melts at 1455°C and boils at 1499°C. 7. is a dangerous gas released by rocks and soil. 8. is a shiny black solid, but gives off a purple vapor when heated. 9. has a W for its symbol. 10. is represented by Thomas Answers 1. Cobalt 2. Chlorine 3. Tungsten 4. Cobalt 5. Platinum 6. Nickel 7. Radon 8. Iodine 9. Tungsten 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. Jefferson on a coin. 11. can be used in surgical instruments. 12. is a solid found in marble, limestone, chalk, and milk. 13. has a name that means lazy in Greek. 14. is a silvery-white metal used in flares, fireworks, and photographic light bulbs. 15. was isolated in 1808 by Sir Humphry Davy. 16. is used in fluorescent lights with argon. 17. is a silvery metal and more than 4 million tons are produced in the United States each year. 18. supports all plant and animal life. 19. was isolated by Martin Henrich Klaproth in 1789. 20. has a name that means hidden one in Greek. 21. is used in nuclear fission and is radioactive, poisonous, and explosive. Nickel Platinum Calcium Argon Magnesium Calcium Krypton Aluminum Oxygen Zirconium S C I E N C E S C O P E 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 25 22. is a silvery metal used for food storage, roofs, and poles. 23. is a noble gas used in light bulbs and photographic pictures. 24. is a black solid contained in every organism. 25. has a name that comes from a Greek word meaning stone. 26. can be found formed as diamond. 27. is an alkali metal that can be cut with a knife. 28. is a metal used to make silverware, jewelry, and coins. 29. is explosive and is the lightest of all metals. 30. has a name that comes from the goddess of moon, Luna or Diana. 31. is the lightest of all elements (atomic number = 1) and is a flammable gas. 32. is used to make batteries, enamels, dyes, and in its carbonate form is used in a drug to treat depression. 33. is found in aircrafts, automobiles, welding, and cooking utensils, and is used to transmit electricity. 34. is a solid and forms rust when reacted with air. 35. is a halogen gas that is yellow and explosive. 36. can be dangerous if inhaled or absorbed through the skin. 37. is a yellow solid and has a gas form that smells like rotten eggs. Krypton Plutonium Tin Xenon Carbon Lithium Carbon Lithium Silver Lithium O C T O B E R 30. 31. 32. 33. 34. 35. 36. 37. 1 9 9 6 Silver Hydrogen Lithium Aluminum Iron Fluorine Lead Sulfur